Australian snake envenoming is rare but potentially life-threatening and challenging to manage.1,2 Issues to be considered include the natural history of clinical envenoming, appropriate investigation and exclusion of envenoming in suspected snakebite cases,1 choice and dosing of antivenom, and ancillary treatments.3
Most snakebites do not result in clinical envenoming1,2,4 because insufficient venom is injected (ie, a dry bite) or because the snake is non-venomous. Clinical envenoming may include local effects, systemic symptoms and major toxin syndromes — venom-induced consumption coagulopathy (VICC), neurotoxicity and myotoxicity (Box 1).2,10 Local effects are uncommon in Australian snakebite and are minimal for bites by brown snakes, which cause most major cases of systemic envenoming in Australia.2,11 However, some local pain, swelling and bruising may occur.5,12,13 Less commonly, prominent regional swelling is observed after bites by snakes that cause myotoxicity, including black and tiger snakes.2,4,5,12
VICC occurs in most patients who require antivenom and is usually present on arrival (86% of cases in one study), but may become evident later, within 6 hours of the bite.1,14 VICC is complete or severe in most cases, but partial VICC occurs in some cases where there is only partial consumption of the clotting factors (Box 1). Neurotoxicity and myotoxicity are uncommon and evolve over hours, making the timing of decisions to use antivenom problematic.2,5,6 Thrombotic microangiopathy is a more recently recognised condition that is always associated with VICC and is characterised by thrombocytopenia, microangiopathic haemolytic anaemia (with fragmented red blood cells) and acute renal impairment.7 Anticoagulant “coagulopathy”, characterised by an elevated activated partial thromboplastin time (aPTT), occurs with black snake envenoming but is not associated with clinically important coagulopathy or bleeding.2,4,5 Non-specific systemic symptoms (Box 1) may be an early and useful indication of systemic envenoming that requires antivenom.
Clinical assessment of patients with snakebite includes details of the bite and symptoms or signs occurring in the first hour. A clinically important feature of severe envenoming is early collapse, most common with brown snake envenoming (Box 2).11 Examination focuses on:
Bite site: fang marks, bruising or local necrosis; draining lymph nodes may be painful and support a diagnosis of systemic envenoming
Neurological: cranial nerves (ptosis, ophthalmoplegia, bulbar weakness), limb weakness and respiratory muscle weakness
Haematological: evidence of abnormal coagulation (bleeding from bite site, cannula site, oral cavity or occult sites, including gastrointestinal, urinary and intracranial sites).2,17
Box 2 provides a summary of major clinical effects of the clinically important groups of Australian snakes.4
Investigations for diagnosis and treatment of snake envenoming (as well as excluding envenoming in suspected snakebites) include coagulation studies, a full blood count and biochemical tests.
Laboratory measurement of the international normalised ratio (INR) and aPTT is imperative. Measurement of fibrinogen and D-dimer levels may help with diagnosis of VICC (eg, to differentiate between VICC and anticoagulant effect) but is not essential. The D-dimer level is usually elevated by 100–1000 times the assay cut-off in VICC, and modest increases, in the absence of other indications of envenoming, must be interpreted with caution. Point-of-care devices for measuring INR or D-dimer have been found to give false negative results in VICC and should not be used.18 Results of whole blood clotting tests are also frequently misleading in snakebite,19 and glass tubes required for this test are not available in Australia.
Thrombocytopenia and red cell fragmentation on a blood film indicate a diagnosis of thrombotic microangiopathy.7 A fall in the platelet count is usually evident within 24 hours of the bite.7 A non-specific leukocytosis and lymphopenia can occur with systemic envenoming,20 and the absence of these in death adder envenoming may assist in excluding systemic envenoming.6
Serial measurements of electrolyte, urea and creatinine levels may assist in assessing renal function.2 An elevated creatine kinase (CK) level is a clinically important indicator of myotoxicity but, compared with clinical muscle injury, lags by up to 24 hours.5 An elevated lactate dehydrogenase level may assist in diagnosis of thrombotic microangiopathy.7
The Snake Venom Detection Kit (SVDK; CSL Ltd) is designed to assist in determining the appropriate antivenom to use in envenomed patients.2 Its results must not be used to either diagnose or exclude envenoming.17 However, even for envenomed patients, the SVDK has shown high rates of inaccuracy. This includes misdiagnosis of brown snake envenoming in one in 10 cases of tiger snake envenoming;12 an incorrect result in 14% of bites of snake handlers where an expert identification was available;21 and false negatives when no venom is detected in around 25% of envenomed patients.12 The SVDK may assist in antivenom selection once a diagnosis of envenoming has been made on clinical or laboratory grounds. However, due to its potential for inaccuracy, the result must only be used in light of the clinical syndrome and knowledge of local snake geographical distributions (except for snakes that are captive outside their distribution). Clinical and laboratory features will often indicate which snake group is involved1,12 and, in turn, the appropriate antivenom, without the need for an SVDK.
To ensure test procedures are properly followed, the SVDK should be used by laboratory staff wherever possible. The best specimen is a bite-site swab.22 Urine can be used if a bite-site specimen is not available, but timing of the urine collection is crucial (eg, the first collection of urine, produced before envenoming, may be negative).2,22 Bite-site swabs can be taken on admission and kept for later testing if envenoming develops.4
A pressure bandage with immobilisation (PBI) is recommended first aid for suspected or definite snakebite.23 The bandage needs to be broad (15 cm) and preferably elasticised, rather than the previously recommended crepe bandage.24 The bandage is applied first over the bite and then to cover the whole limb at a pressure similar to that used for a sprained ankle.2,4,22 Immobilisation is essential; the limb and whole patient must remain immobilised from the time of the bite.25 The use of a PBI more than 4 hours after the bite is unlikely to be effective.25
The PBI can be removed when the initial clinical and laboratory assessment shows no evidence of envenoming and the patient is in a facility where antivenom is available (Box 3).1,17 For patients with envenoming, the PBI can be removed after administration of antivenom. As there are reports of cases where envenoming appears to be delayed by early application of a PBI but becomes evident soon after its removal,26 careful observation of the patient in the hour after PBI removal is essential.
Most patients present with a report of a definite or suspected snakebite, but it is often unclear whether a patient is envenomed and what type of snake is involved.1,4,26 Less commonly, patients present after a collapse or seizure but with no report of a snakebite, and are found to have a coagulopathy.
Investigation and treatment should follow a logical process to ensure envenoming is correctly diagnosed and the correct antivenom is used:
Establish the presence of envenoming on arrival
Patients with no clinical or laboratory evidence of envenoming on arrival can be observed for 12 hours, with appropriate repeat laboratory testing (INR, aPTT, CK) and neurological examination to exclude envenoming1
In patients with envenoming, determine which snake or group of snakes is most likely involved
Administer an appropriate antivenom (monovalent, combination of monovalent antivenoms, or polyvalent) that covers the likely snake(s)
Admit the patient for observation or adjunctive treatment (eg, mechanical ventilation) if required.
This process is summarised in Box 3. All patients presenting with a suspected snakebite require an initial full set of investigations (INR, aPTT, full blood count, biochemistry, CK level).1 Some investigations traditionally done for snakebite (ie, D-dimer, fibrinogen and lactate dehydrogenase levels) are not always available, and are not essential when managing uncomplicated cases.1
Most patients will have no clinical or laboratory features of envenoming when first assessed. Once this initial assessment is complete, these patients can have their PBI (if present) removed in a critical care area. If no clinical features of envenoming occur within an hour of PBI removal, patients should be moved to a general clinical area and observed.17 In one large prospective study, all cases of severe envenoming were evident by 12 hours after the bite, irrespective of the timing of PBI application or removal.1 In addition to a neurological examination looking for ptosis or bulbar, respiratory or distal paralysis, laboratory tests of INR, aPTT and CK level should be undertaken 1 hour after removal of the PBI, 6 hours after the bite and 12 hours after the bite.1
Evidence of envenoming exists if neurotoxicity develops or the INR, aPTT or CK level becomes abnormal, and antivenom treatment should be considered based on the timing, severity and specific abnormality. Investigations conducted 1 hour after removal of the PBI may coincide approximately with those required 6 hours after the bite; in this instance, only one set of blood tests are required. If results of 6-hour laboratory investigations are normal, 12-hour investigations can be delayed a few hours if necessary to avoid recall of overnight laboratory staff.17
For the rare circumstances in which a PBI is left on for more than 6 hours, a final set of blood tests and a neurological examination should be done 6 hours after removal of the PBI.
Accumulating data suggest that antivenom might prevent certain envenoming syndromes if used early,5,13,27 but may have little, if any, effect once major envenoming syndromes are established. However, there is no high-level evidence to guide us in this regard. In the absence of evidence from properly conducted trials, clinicians have to balance the risks of giving antivenom (anaphylaxis and serum sickness) against the potential benefit of giving it early without waiting for confirmation of envenoming. Any early evidence of envenoming, such as non-specific systemic symptoms or mild coagulopathy, may be an indication for antivenom. Discussion with a clinical toxicologist may be beneficial in these circumstances. Absolute and relative indications for antivenom are listed in Box 4.
Determination of the snake (or snake group) involved and therefore the appropriate antivenom to be administered requires:
Local knowledge of snakes found where the patient was bitten
Observation of the specific clinical syndrome characterised by the clinical and laboratory features at presentation or subsequently (Box 2).
In some cases, an expert may be available for snake identification, or the person bitten may be a snake handler who can identify the snake. Snake identification should only be performed by experts such as professional herpetologists and museum curators.2 Snake handlers may be bitten by snakes outside of their geographical distribution. Some professional snake handlers may be able to provide accurate identification of the snakes in their possession, which should be used rather than the SVDK.21 If the snake type remains unclear based on location and clinical syndrome, the SVDK may assist in regions where the range of possible snakes is too broad to allow the use of one or two monovalent antivenoms. If there is any doubt about the snake involved, it may be safer to administer polyvalent antivenom or two monovalent antivenoms according to species endemic to the region.
In most parts of southern and central–eastern Australia, one vial each of brown snake and tiger snake antivenom will cover the clinically important snakes in the local area, based on the clinical envenoming syndrome.11,12 These two monovalent antivenoms comprise a much smaller total volume of antivenom (5–10 mL) compared with polyvalent antivenom (45–50 mL) and cost much less.22 In some cases, the larger-volume polyvalent antivenom may be appropriate, but it is associated with a higher risk of anaphylaxis.14 Black snake, death adder and taipan antivenoms are also larger volume (> 10 mL) than tiger and brown snake antivenoms.22 As these snakes account for a small number of severe envenoming cases in most parts of Australia, it is preferable to stock and use polyvalent antivenom for treating envenoming by these three snakes rather than keeping three rarely used monovalent antivenoms.
One vial of relevant snake monovalent antivenom is required to treat both children and adults for all snake types.17 The use of more than one vial or repeat doses of antivenom is no longer recommended,5,6,8,12,13,15,16 based on in-vitro and clinical evidence from the Australian Snakebite Project (ASP). Recovery of most clinical syndromes of snake envenoming takes time because of their irreversibility or slow reversibility (eg, synthesis of new clotting factors required for VICC to resolve).28 Further doses of antivenom have not been found to speed this recovery.28
Some guidelines or previous studies have suggested more than one vial of antivenom should be used for some snakes or in some situations.10,22,29 However, in over 15 ASP studies to date, there is no evidence to support such recommendations.5,6,8,11,12,15,16 The ASP studies have found no evidence to suggest there is active venom present after administration of one vial of antivenom.5,6,8,11,12,15,16 No difference in time course, severity of effects or recovery has been shown to correlate positively with total antivenom dose.6,28 Moreover, one vial of antivenom is designed to provide sufficient antivenom to completely neutralise the total maximum venom load from one snake, which is extremely unlikely to be injected in any snakebite.22 Thus, the case for more antivenom is not supported by evidence and has no biologically plausible basis for benefit, but can be predicted to increase adverse effects.14,21
Antivenom must be given in a critical care area, and staff must be prepared to treat anaphylaxis. It is advisable to use a small-bore cannula (18–20 G in adults) for antivenom infusion and to have a second, large-bore cannula (16–14 G in adults) inserted ready for emergency resuscitation (Box 5). Antivenom is given diluted in 100–250 mL of isotonic saline (smaller dilutions should be used for children) over 15–30 minutes.22 Premedication with adrenaline, antihistamines and corticosteroids is no longer recommended in Australia. The rate of antivenom administration does not appear to be associated with increased reactions.32 Systemic hypersensitivity reactions are the most important adverse effects from antivenom administration and occur in 20%–25% of patients.14,21 Severe anaphylaxis with hypotension occurs in 3%–5% of patients receiving antivenom in Australia.21 Previous exposure to antivenom is not an important risk factor. Reactions appear to be more common with larger-volume antivenoms.14 Serum sickness occurs in about a third of patients given antivenom and is characterised by influenza-like symptoms, fever, myalgia, arthralgia and rash developing 4–14 days after administration of antivenom. Serum sickness can be treated with prednisolone (25 mg daily for 5–7 days).2,22
Patients who are given antivenom must be admitted for repeat laboratory testing and observation to determine when envenoming has resolved and to identify complications. Measurement of INR, aPTT, creatinine level and CK level (for myotoxic snakes) and a full blood count should be done 6 and 12 hours after administration of antivenom, and then once to twice daily until there is sustained improvement.17
The median time to recovery to an INR < 2.0 in VICC is about 15 hours, and the INR will normalise 24–36 hours after the bite, after which most patients can be discharged.9 Appropriate investigation for occult bleeding, such as a cerebral computed tomography scan, should be undertaken if clinically indicated. Thrombotic microangiopathy should be excluded in all patients with VICC by observing no change in creatinine level and platelet count over the first 24 hours.7 Patients with uncomplicated myotoxicity and mild neurotoxicity can be discharged once the clinical features are resolving.17
The role of clotting factor replacement in treating VICC remains contentious. A recent randomised controlled trial of fresh frozen plasma (FFP) versus no additional treatment within 4 hours of administration of antivenom for VICC found that FFP results in more rapid restoration of clotting function in most patients, but with no decrease in time to discharge.3 Early FFP (< 6–8 hours after the bite) was less likely to be effective, with evidence of ongoing factor consumption.3 Larger studies are required to confirm the safety and effectiveness of FFP, particularly when given early. In the meantime, it is reasonable to administer FFP after giving antivenom to patients who have active bleeding and an imminent threat to their life. FFP is the best factor replacement (rather than cryoprecipitate or Prothrombinex-VF [CSL Bioplasma]) because patients are deficient in fibrinogen, factor V and factor VIII.
Complications of snake envenoming are rare and generally occur in patients presenting late with severe neuromuscular paralysis, rhabdomyolysis or thrombotic microangiopathy with acute renal failure.6,12,16 Such patients will require admission to a tertiary hospital with high-dependency or intensive care support. Major bleeding may require clotting factor replacement, such as FFP, and supportive care. Thrombotic microangiopathy may require haemodialysis, but there is no evidence to support the use of plasmapheresis.7 Consultation with a haematologist and renal physician is recommended. Significant rhabdomyolysis with acute renal failure is rare but should be treated with generous fluid therapy and close monitoring for electrolyte imbalances (eg, hyperkalaemia). Severe neuromuscular paralysis may require intubation and mechanical ventilation for days or weeks.2,6 Some patients may report altered taste or smell after snake envenoming, which may persist for many weeks but usually resolves.2
Bites of snake handlers comprise 10% of snakebites in Australia, and implicated snakes include several uncommon snakes kept in captivity that rarely cause bites in the wild.21 Snake handlers are often reluctant to receive antivenom because of a belief that they are at greater risk of systemic hypersensitivity reactions to antivenom, but there is little evidence to suggest this is true. However, they may develop hypersensitivity reactions to venom, which must be considered in the differential diagnosis.21
1 Characteristics of clinical syndromes from snakebite in Australia1,5-9
Sudden collapse
Collapse or syncope occurring within an hour of the bite
Collapse is associated with hypotension and loss of consciousness
Spontaneous recovery usually occurs within minutes
Minority of patients (about 5%) have a cardiac arrest or seizure
Venom-induced consumption coagulopathy (VICC)
Activation of the clotting pathway by prothrombin activator toxins and consumption of clotting factors (fibrinogen, factor V and factor VIII) lead to a consumptive coagulopathy
INR is high or unrecordable and aPTT is prolonged
Fibrinogen level is low or undetectable and D-dimer level is very high
Complete or severe VICC is defined as:
Undetectable fibrinogen level, INR > 3.0 (most often unrecordable), abnormal aPTT (outside the laboratory’s reference interval), and very high D-dimer level (100–1000 × assay cut-off)
Partial VICC (less severe changes) is defined as:
Low but detectable fibrinogen level (< 1.5 g/L) and INR < 3.0
Neurotoxicity
A descending flaccid paralysis that classically first involves the eye muscles (ptosis, diplopia and blurred vision), followed by bulbar muscles, respiratory muscle paralysis and limb paralysis
Myotoxicity
Local or generalised myalgia and/or muscle tenderness
CK level is usually normal (within the laboratory’s reference interval) on admission and rapidly rises over 24–48 hours (peak ranges from 1000 U/L in mild cases to > 100 000 U/L in severe cases)
Potassium level may also be elevated (> 5.0 mmol/L) in severe cases, and renal impairment may develop
Anticoagulant coagulopathy
Provides a good marker of envenoming by all black snakes, including mulga snakes, but is not clinically important
aPTT is moderately abnormal (1.5–2.5 × laboratory’s reference interval), with or without mild elevation of INR (> 1.3)
D-dimer and fibrinogen levels are generally normal (D-dimer < 1.0 mg/L and fibrinogen > 1.5 g/L [or > 2.0 g/L in some laboratories])
Thrombotic microangiopathy
Presence of fragmented red blood cells on blood film (microangiopathic haemolytic anaemia), thrombocytopenia and a rising creatinine level (> 120 mmol/L), which may lead to acute renal failure requiring dialysis
Systemic symptoms
INR = international normalised ratio. aPTT = activated partial thromboplastin time. CK = creatine kinase.
2 Summary of effects of clinically important venomous Australian snakes, based on definite cases of systemic envenoming4-6,8,11-13,15,16
Snake |
Coagulopathy |
Neurotoxicity* |
Myotoxicity |
Systemic symptoms† |
Thrombotic microangiopathy† |
Cardiovascular effects† |
Antivenom |
||||||||
Brown snake |
VICC‡ |
Rare and mild |
— |
< 50% |
10% |
Collapse (33%), cardiac arrest (5%) |
Brown snake |
||||||||
Tiger snake group |
|
|
|
|
|
|
|
||||||||
Tiger snake |
VICC‡ |
Uncommon |
Uncommon |
Common |
5% |
Rare |
Tiger snake |
||||||||
Rough-scaled snake |
VICC‡ |
Uncommon |
Uncommon |
Common |
< 5% |
Rare |
Tiger snake |
||||||||
Hoplocephalus spp§ |
VICC‡ |
— |
— |
< 50% |
< 5% |
Rare |
Tiger or brown snake |
||||||||
Black snake group |
|
|
|
|
|
|
|
||||||||
Mulga snake |
Anticoagulant |
— |
Common |
Common |
— |
— |
Black snake¶ |
||||||||
Red-bellied black snake |
Anticoagulant |
— |
Common |
Common |
— |
— |
Tiger snake |
||||||||
Death adder |
— |
Common |
— |
Common |
— |
— |
Death adder¶ |
||||||||
Taipan |
VICC‡ |
Common |
Rare |
Common |
5% |
Uncommon |
Taipan¶ |
||||||||
Sea snakes |
— |
Uncommon |
Common |
Common |
— |
— |
Sea snake** |
||||||||
VICC = venom-induced consumption coagulopathy. — = does not occur. * Neurotoxicity was more common in the era before antivenom and has been further reduced by the early use of antivenom. † Percentages indicate proportion of cases in which the effect occurs. ‡ Partial VICC occurs in 20%–30% of cases for all snakes that cause VICC, except taipan envenoming, where it occurs in almost 50% of cases. § The Hoplocephalus genus includes Stephens’ banded snake (Hoplocephalus stephensii), the broad-headed snake (Hoplocephalus bungaroides) and the pale-headed snake (Hoplocephalus bitorquatus). ¶ Polyvalent antivenom can be substituted for these large-volume monovalent antivenoms with no increase in risk or cost. ** Polyvalent or tiger snake antivenom cannot be used for sea snake envenoming. |
|||||||||||||||
3 Flowchart for the management of snakebite*
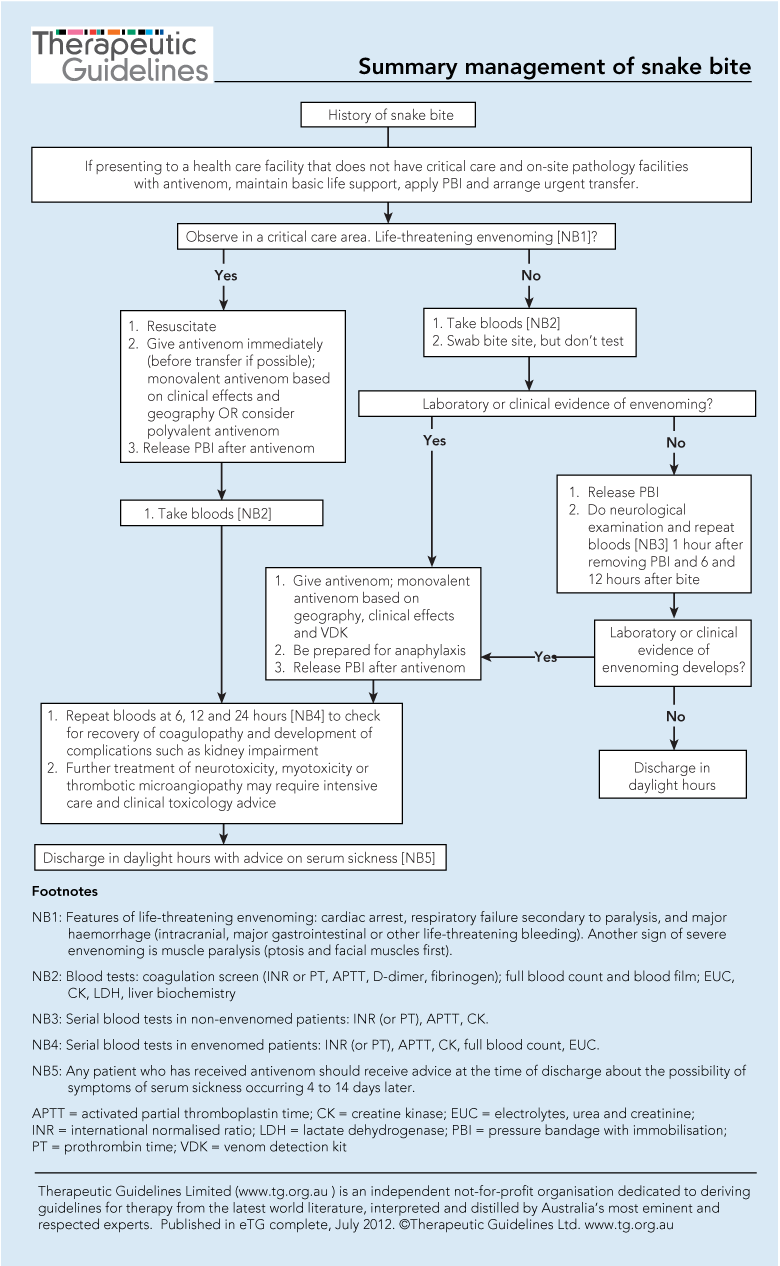
* Reproduced with permission from Toxicology and Wilderness Expert Group. Summary management of snake bite (Figure 17.3) [revised 2012 June]. In: eTG complete [internet]. Melbourne: Therapeutic Guidelines Limited, Jul 2013.
4 Absolute and relative indications for antivenom
Absolute indications |
Relative indications |
||||||||||||||
|
|
||||||||||||||
5 Management of immediate reactions to antivenom*
1 |
Stop antivenom infusion. Many reactions will resolve with this step, and the infusion can then be restarted at a slower rate. |
||||||||||||||
2 |
Lie patient flat, commence high-flow oxygen, support airway and ventilation if required. |
||||||||||||||
3 |
For hypotension, give rapid infusion of 1 L normal saline (20 mL/kg in children). Severe antivenom reactions with hypotension will have reduced venous return; supine posture and fluid resuscitation are essential. |
||||||||||||||
4 |
For hypotension, hypoxaemia, wheeze or upper airway obstruction, give intramuscular adrenaline (0.01 mg/kg to a maximum of 0.5 mg). Alternatively, those experienced with intravenous infusions of adrenaline may go straight to Step 5. |
||||||||||||||
5 |
Consider a cautious intravenous infusion of adrenaline — avoid blood pressure surges. Patients with envenoming may be severely coagulopathic, and high blood pressure may cause or worsen intracerebral haemorrhage. Some patients can have exaggerated, hypertensive responses to intramuscular bolus adrenaline, especially to second doses. If there is no response to Steps 1–4, consider starting a cautious and closely monitored intravenous infusion of adrenaline, which can be reduced as soon as blood pressure starts to recover, preventing blood pressure surges. Use 1 mg in 100 mL by infusion pump: start at 0.5 mL/kg/h and titrate according to response; monitor blood pressure every 3–5 min (using the arm opposite to the infusion). Be aware that as the reaction resolves, adrenaline requirements will fall, the blood pressure will rise and the infusion rate will need to be rapidly reduced.30 |
||||||||||||||
6 |
For persistent hypotension, repeat normal saline bolus. |
||||||||||||||
7 |
For bronchospasm, consider nebulised salbutamol. |
||||||||||||||
8 |
For upper airway obstruction, consider nebulised adrenaline. |
||||||||||||||
9 |
Seek further advice from a National Poisons Information Centre consultant. |
||||||||||||||
* A modification of standard anaphylaxis guidelines31 is recommended because of the risk of coagulopathy. |
|||||||||||||||
Provenance: Not commissioned; externally peer reviewed.
- Geoffrey K Isbister1,2
- Simon G A Brown3
- Colin B Page4,2
- David L McCoubrie5
- Shaun L Greene6
- Nicholas A Buckley7,8
- 1 Discipline of Clinical Pharmacology, University of Newcastle, Newcastle, NSW.
- 2 Department of Clinical Toxicology and Pharmacology, Calvary Mater Newcastle, Newcastle, NSW.
- 3 Centre for Clinical Research in Emergency Medicine, Western Australian Institute for Medical Research, Royal Perth Hospital and University of Western Australia, Perth, WA.
- 4 Emergency Department, Princess Alexandra Hospital, Brisbane, QLD.
- 5 Emergency Department, Royal Perth Hospital, Perth, WA.
- 6 Emergency Department and Victorian Poisons Information Centre, The Austin Hospital, Melbourne, VIC.
- 7 NSW Poisons Information Centre, Sydney Children’s Hospital Network, Sydney, NSW.
- 8 Professorial Medicine Unit, University of New South Wales, Sydney, NSW.
We thank Bart Currie for reviewing the manuscript.
No relevant disclosures.
- 1. Ireland G, Brown SG, Buckley NA, et al. Changes in serial laboratory test results in snakebite patients: when can we safely exclude envenoming? Med J Aust 2010; 193: 285-290. <MJA full text>
- 2. White J. Clinical toxicology of snakebite in Australia and New Guinea. In: Meier J, White J, editors. Handbook of clinical toxicology of animal venoms and poisons. New York: CRC Press, 1995: 595-618.
- 3. Isbister GK, Buckley NA, Page CB, et al. A randomized controlled trial of fresh frozen plasma for treating venom-induced consumption coagulopathy in cases of Australian snakebite (ASP-18). J Thromb Haemost 2013; 11: 1310-1318.
- 4. Currie BJ. Snakebite in tropical Australia: a prospective study in the “Top End” of the Northern Territory. Med J Aust 2004; 181: 693-697. <MJA full text>
- 5. Churchman A, O’Leary MA, Buckley NA, et al. Clinical effects of red-bellied black snake (Pseudechis porphyriacus) envenoming and correlation with venom concentrations: Australian Snakebite Project (ASP-11). Med J Aust 2010; 193: 696-700. <MJA full text>
- 6. Johnston CI, O’Leary MA, Brown SG, et al. Death adder envenoming causes neurotoxicity not reversed by antivenom – Australian Snakebite Project (ASP-16). PLOS Negl Trop Dis 2012; 6: e1841.
- 7. Isbister GK. Snakebite doesn’t cause disseminated intravascular coagulation: coagulopathy and thrombotic microangiopathy in snake envenoming. Semin Thromb Hemost 2010; 36: 444-451.
- 8. Isbister GK, White J, Currie BJ, et al. Clinical effects and treatment of envenoming by Hoplocephalus spp. snakes in Australia: Australian Snakebite Project (ASP-12). Toxicon 2011; 58: 634-640.
- 9. Isbister GK, Scorgie FE, O’Leary MA, et al. Factor deficiencies in venom-induced consumption coagulopathy resulting from Australian elapid envenomation: Australian Snakebite Project (ASP-10). J Thromb Haemost 2010; 8: 2504-2513.
- 10. Currie BJ. Snakebite in tropical Australia, Papua New Guinea and Irian Jaya. Emerg Med 2000; 12: 285-294. doi: 10.1046/j.1442-2026.2000.00150.x.
- 11. Allen GE, Brown SG, Buckley NA, et al. Clinical effects and antivenom dosing in brown snake (Pseudonaja spp.) envenoming – Australian Snakebite Project (ASP-14). PLOS One 2012; 7: e53188.
- 12. Isbister GK, O’Leary MA, Elliott M, Brown SG. Tiger snake (Notechis spp) envenoming: Australian Snakebite Project (ASP-13). Med J Aust 2012; 197: 173-177. <MJA full text>
- 13. Johnston CI, Brown SG, O’Leary MA, et al. Mulga snake (Pseudechis australis) envenoming: a spectrum of myotoxicity, anticoagulant coagulopathy, haemolysis and the role of early antivenom therapy - Australian Snakebite Project (ASP-19). Clin Toxicol (Phila) 2013; 51: 417-424.
- 14. Isbister GK, Brown SG, MacDonald E, et al. Current use of Australian snake antivenoms and frequency of immediate-type hypersensitivity reactions and anaphylaxis. Med J Aust 2008; 188: 473-476. <MJA full text>
- 15. Gan M, O’Leary MA, Brown SG, et al. Envenoming by the rough-scaled snake (Tropidechis carinatus): a series of confirmed cases. Med J Aust 2009; 191: 183-186. <MJA full text>
- 16. Kulawickrama S, O’Leary MA, Hodgson WC, et al. Development of a sensitive enzyme immunoassay for measuring taipan venom in serum. Toxicon 2010; 55: 1510-1518.
- 17. Toxicology and Wilderness Expert Group. Therapeutic guidelines: toxicology and wilderness. Version 2. Melbourne: Therapeutic Guidelines Limited, 2012.
- 18. Cubitt M, Armstrong J, McCoubrie D, et al. Point-of-care testing in snakebite: an envenomed case with false negative coagulation studies. Emerg Med Australas 2013; 25: 372-373.
- 19. Isbister GK, Maduwage K, Shahmy S, et al. Diagnostic 20-min whole blood clotting test in Russell’s viper envenoming delays antivenom administration. QJM 2013; 106: 925-932.
- 20. White J, Williams V, Duncan B. Lymphopenia after snakebite. Lancet 1989; 2: 1448-1449.
- 21. Isbister GK, Brown SG. Bites in Australian snake handlers — Australian snakebite project (ASP-15). QJM 2012; 105: 1089-1095.
- 22. White J. CSL antivenom handbook. 2nd ed. Melbourne: CSL Ltd, 2001.
- 23. Sutherland SK, Coulter AR, Harris RD. Rationalisation of first-aid measures for elapid snake bite. Lancet 1979; 1: 183-185.
- 24. Canale E, Isbister GK, Currie BJ. Investigating pressure bandaging for snakebite in a simulated setting: bandage type, training and the effect of transport. Emerg Med Australas 2009; 21: 184-190.
- 25. Howarth DM, Southee AE, Whyte IM. Lymphatic flow rates and first-aid in simulated peripheral snake or spider envenomation. Med J Aust 1994; 161: 695-700.
- 26. Pearn J, Morrison J, Charles N, Muir V. First-aid for snake-bite: efficacy of a constrictive bandage with limb immobilization in the management of human envenomation. Med J Aust 1981; 2: 293-295.
- 27. Lalloo DG, Trevett AJ, Korinhona A, et al. Snake bites by the Papuan taipan (Oxyuranus scutellatus canni): paralysis, hemostatic and electrocardiographic abnormalities, and effects of antivenom. Am J Trop Med Hyg 1995; 52: 525-531.
- 28. Isbister GK, Duffull SB, Brown SG. Failure of antivenom to improve recovery in Australian snakebite coagulopathy. QJM 2009; 102: 563-568.
- 29. Yeung JM, Little M, Murray LM, et al. Antivenom dosing in 35 patients with severe brown snake (Pseudonaja) envenoming in Western Australia over 10 years. Med J Aust 2004; 181: 703-705. <MJA full text>
- 30. Brown SG, Blackman KE, Stenlake V, Heddle RJ. Insect sting anaphylaxis; prospective evaluation of treatment with intravenous adrenaline and volume resuscitation. Emerg Med J 2004; 21: 149-154.
- 31. Brown SG, Mullins RJ, Gold MS. Anaphylaxis: diagnosis and management. Med J Aust 2006; 185: 283-289. <MJA full text>
- 32. Isbister GK, Shahmy S, Mohamed F, et al. A randomised controlled trial of two infusion rates to decrease reactions to antivenom. PLOS One 2012; 7: e38739.





Summary
Snakebite is a potential medical emergency and must receive high-priority assessment and treatment, even in patients who initially appear well.
Patients should be treated in hospitals with onsite laboratory facilities, appropriate antivenom stocks and a clinician capable of treating complications such as anaphylaxis.
All patients with suspected snakebite should be admitted to a suitable clinical unit, such as an emergency short-stay unit, for at least 12 hours after the bite. Serial blood testing (activated partial thromboplastin time, international normalised ratio and creatine kinase level) and neurological examinations should be done for all patients.
Most snakebites will not result in significant envenoming and do not require antivenom.
Antivenom should be administered as soon as there is evidence of envenoming. Evidence of systemic envenoming includes venom-induced consumption coagulopathy, sudden collapse, myotoxicity, neurotoxicity, thrombotic microangiopathy and renal impairment.
Venomous snake groups each cause a characteristic clinical syndrome, which can be used in combination with local geographical distribution information to determine the probable snake involved and appropriate antivenom to use. The Snake Venom Detection Kit may assist in regions where the range of possible snakes is too broad to allow the use of monovalent antivenoms.
When the snake identification remains unclear, two monovalent antivenoms (eg, brown snake and tiger snake antivenom) that cover possible snakes, or a polyvalent antivenom, can be used.
One vial of the relevant antivenom is sufficient to bind all circulating venom. However, recovery may be delayed as many clinical and laboratory effects of venom are not immediately reversible.
For expert advice on envenoming, contact the National Poisons Information Centre on 13 11 26.