The Australian National Hand Hygiene Initiative (NHHI) and Hand Hygiene Australia (HHA) were established by the Australian Commission on Safety and Quality in Health Care after studies demonstrated that multimodal culture-change programs and increased use of alcohol-based hand rub reduced rates of health care-associated infections, especially those caused by methicillin-resistant Staphylococcus aureus (MRSA).1-4
The aim of the NHHI was to implement a standardised hand hygiene (HH) culture-change program throughout all Australian hospitals to improve HH compliance among Australian health care workers (HCWs); establish a validated system of HH compliance auditing to allow local, national and international benchmarking; and establish a reliable system of health care-associated disease reporting, initially focusing on S. aureus bacteraemia (SAB) as a practical outcome measure of HH efficacy.5
Here, we describe the outcomes from the first 2 years of the NHHI. Further details of the program’s structure, methods and outcomes can be found in the supplementary material available at http://www.hha.org.au/mjasupplement.aspx.6
Although several Australian states (New South Wales, Queensland, South Australia, Victoria, Western Australia) had implemented infection control initiatives to improve HH among HCWs,1,2,7-11 benefits could be gained from a standardised national approach. The NHHI program was based on the World Health Organization “5 Moments for Hand Hygiene” program,5 which defines the five key “Moments” when hand-cleaning is required during patient care (1: before touching a patient; 2: before a procedure; 3: after a procedure or body fluid exposure risk; 4: after touching a patient; 5: after touching a patient’s surroundings). The program included standardised educational materials and a regular audit system of HH compliance. The NHHI was undertaken as a quality improvement initiative in all jurisdictions and did not require ethics approval. Implementation began in January 2009.
To ensure standardisation and reliability of HH compliance data according to the HHA-adapted WHO 5 Moments tool,5,12 HHA conducted up to 200 training workshops in all Australian states and territories to establish a network of “gold standard” auditors, who helped train other HCWs to become HHA auditors. Only accredited HHA auditors could submit data to HHA.6
A 4-monthly schedule of data submission was established at all sites (Periods 1–3 in 2009 and 2010).6 The size and nature of each participating hospital generally determined the type of wards and the total number of “HH moments” that were to be audited for each submission, such that the audit intensity was potentially consistent with the hospital’s likely infection control risk.6 Although the initial focus was on public hospitals, there was subsequently a rapid roll-out and uptake of the program among a majority of the large private hospital groups.
HH compliance data were analysed by overall compliance rates (ward, hospital, state and national [public hospitals, private hospitals, total]), HH moments and HCW category.6
For the purpose of this study and as a means of establishing a national benchmark for SAB rates, we collated available SAB data from all states and territories for the 2 years before (2007–2008) and the 2 years after (2009–2010) implementation of the NHHI, noting where differences in definitions and denominator data applied. We calculated the national SAB rate per month using previously agreed definitions of patients with SAB2,12-14 as the numerator, and either patient days or occupied bed-days as the measure of hospital activity for each state/territory as the denominator.6 Patient-days were defined as the total number of days for patients who were admitted for an episode of care and separated during a specified reference period. A patient admitted and separated on the same day was allocated 1 patient-day.15 Occupied bed-days were defined as the sum of the number of daily occupied beds for the surveillance period. A patient episode of SAB was considered to be hospital-onset (HO) if the blood culture was collected > 48 hours after hospital admission.
Statistical analyses included χ2 tests, confidence intervals for proportions, and linear regression where appropriate; 95% confidence intervals were calculated for all HH compliance rates. Trends for SAB (total, MRSA and methicillin-susceptible S. aureus [MSSA]) rates over time were assessed as previously described1,2 using interrupted time-series segmented regression analysis.16 Statistical analyses were performed using Prism 5.01 for Windows (GraphPad Software, La Jolla, Calif, USA).
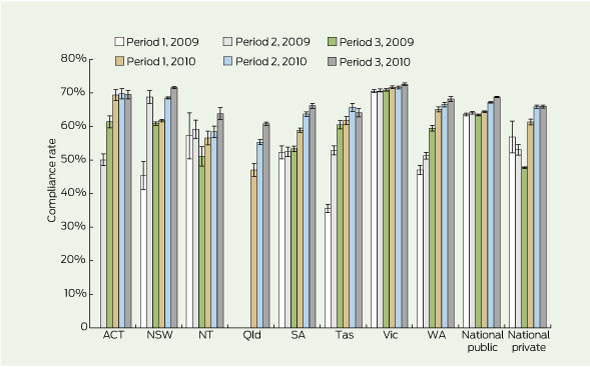
During 2009–2010, a total of 917 622 HH moments were assessed nationally; HH compliance rates are shown in Box 1. NHHI commencement varied by location, with the Northern Territory, SA, Tasmania, Victoria and WA starting in early 2009, NSW and the Australian Capital Territory in mid 2009, and Queensland in early 2010. In late 2010, the overall national rate of HH compliance was 68.3% (95% CI, 68.1%–68.5%; 168 641/246 931 moments) in 521 hospitals, representing about 90% of acute Australian public non-psychiatric hospital beds and about 50% of acute private hospital beds. These data compare with a national HH compliance rate at the start of the NHHI of 63.6% (95% CI, 63.2%–64.0%; 36 213/56 978; P < 0.001) (Box 1) — however, these baseline data were heavily influenced by Victoria, where a program using a similar audit tool had been in place for some years.1,2 For non-Victorian sites, overall HH compliance improved from mean baseline rates of 43.6% (95% CI, 42.8%–44.4%; 6431/14 740) and 53.5% (95% CI, 52.9%–54.0%; 16 547/30 934) in 2009 audit Periods 1 and 2, respectively, to 67.8% (95% CI, 67.5%–68.0%; 106 851/157 708) at the end of 2010 (P < 0.001 for both).
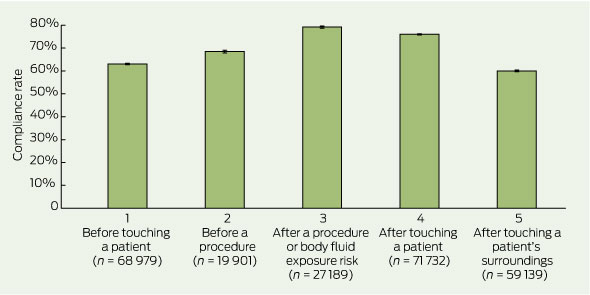
National compliance rates for each of the five HH moments 2 years after commencing NHHI implementation are shown in Box 2. HH compliance before touching a patient (Moments 1–2) was 12.6% lower than after patient contact (Moments 3–4) (64.3% [57 119/88 880] v 76.9% [76 051/98 921]) (P < 0.001). HH compliance before a procedure was 68.4% (13 620/19 901), compared with 79.1% (21 520/27 189) after a procedure (P < 0.001).
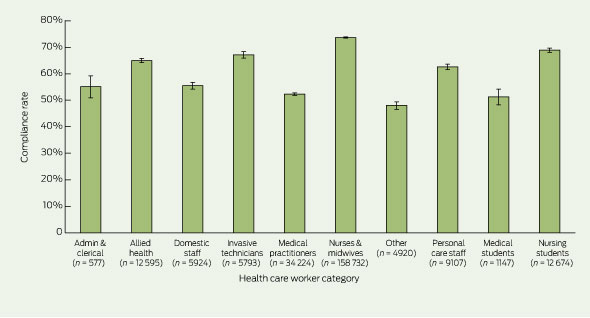
HH compliance by HCW category at the end of 2010 is shown in Box 3. The best overall compliance rates were noted for nursing staff (73.6%; 116 851/158 732). Compliance among medical staff improved only slightly from a baseline of 50.5% to 52.3% (4378/8669 v 17 897/34 224 moments; P = 0.003) — a rate that was significantly lower than that for all non-medical HCWs (70.9%, 149 919/211 469; P < 0.001). Among medical staff in states that participated for the entire audit period (SA, Tas, Vic and WA), HH compliance improved from 51.0% (4329/8496) to 54.6% (6640/12 158) (P < 0.001).
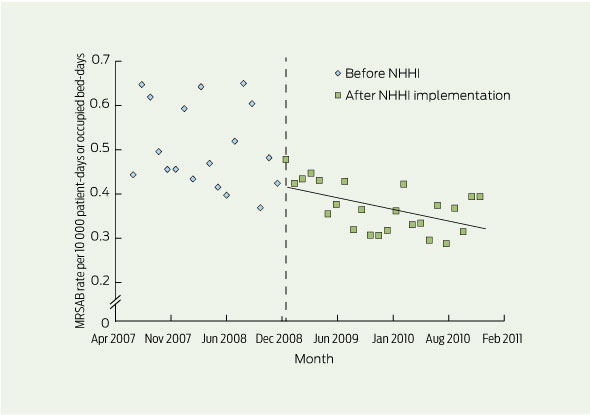
Suitably robust data were available6 to describe national total (HO and non-HO) incidence rates of MRSA bacteraemia (MRSAB) for 18 of the 24 months before NHHI implementation (July 2007–2008) and the 2 years after implementation (2009–2010) (Box 4). MRSAB rates were statistically stable before the NHHI (P = 0.366) but declined during 2009–2010 (P = 0.008). Mean annual national rates of total MRSAB in 2008, 2009 and 2010 were 0.4998, 0.3902 and 0.3497 per 10 000 patient-days, respectively.
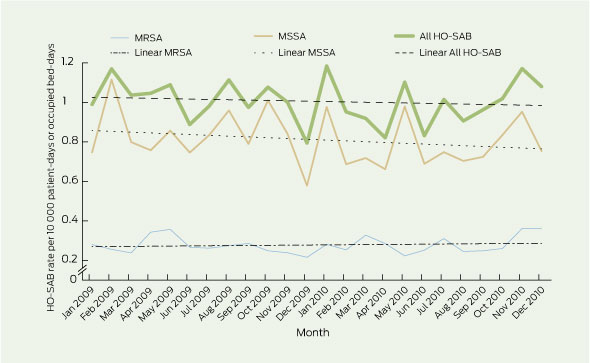
National rates of HO-SAB for 2009–2010 are shown in Box 5. Rates for total HO-SAB, HO-MRSAB and HO-MSSA bacteraemia (HO-MSSAB) were statistically stable during this period (P = 0.59, P = 0.58 and P = 0.30, respectively). For 2009, annual rates of HO-SAB, HO-MRSAB and HO-MSSAB were 1.004, 0.268 and 0.826 per 10 000 patient-days, respectively, and in 2010 these rates were 0.995, 0.284 and 0.784. For each of these years, MSSA accounted for about 75% of HO-SAB, although some SAB episodes included both MSSA and MRSA.
Use of the HHA–WHO 5 Moments auditing tool allows HH performance to be benchmarked nationally and internationally and provides important information for targeting educational efforts. The finding that HH compliance before patient contact or performing a procedure was 10%–15% lower than compliance after patient contact is a major concern — albeit consistent with previous Australian and international studies1,2,5 — and highlights the need for focused educational efforts in this area.
Nursing staff appear to have readily adopted HH culture change, while medical staff lag significantly behind both the overall national average rate of compliance (52.3% v 68.3%, P < 0.001) and that observed for non-medical HCWs (70.9%, P < 0.001). Although such differences have been noted previously,2,5 the reasons for this lower compliance are likely to be complex and require further investigation. The HHA program is standardised and homogeneous, whereas consumer profiling and marketing studies suggest multiple educational approaches are likely to have the greatest impact on various HCW groups.17 Not unexpectedly, medical staff had far fewer moments when HH was required than nursing staff (Box 3). This may be beneficial, given their lower compliance rates, although clearly the type of patient contact (eg, invasive v non-invasive) is important. Furthermore, it is only recently that education on appropriate HH compliance has become a regular feature of Australian medical school curricula (Geoff McColl, Director, Medical Education Unit, University of Melbourne, personal communication). Nevertheless, our data raise important concerns about the assumed leadership role of medical staff in terms of HH compliance.
Although population-based SAB rates have been previously estimated,18,19 these do not necessarily reflect hospital activity, but can now potentially be reconciled with our data. Aside from establishing an Australian benchmark rate for MRSAB and HO-SAB, our data highlight some interesting features. First, the finding that total MRSAB rates declined significantly during 2009–2010, yet HO-MRSAB did not, suggests that SAB episodes occurring < 48 hours after admission are likely to be important. Clarification of whether these are non-inpatient health care-associated or community-acquired is likely to influence future disease prevention strategies and highlights the importance of establishing a standard national system of SAB data reporting. Second, our data suggest that about three-quarters of HO-SAB is due to MSSA rather than MRSA. Given that in 2009–2010 there were 25 738 000 acute non-psychiatric patient-days in Australian public and private hospitals,15 we could estimate from our data that there were about 1000 patient-episodes of MRSAB and about 2600 of HO-SAB nationally.
Although changes in the total MRSAB rate during 2009–2010 cannot be definitively linked to the NHHI and may be related to other factors,20,21 the general decline is consistent with reports from previous Australian and international HH culture-change programs.1-3 Based on these data, we expect to see a steady decline in the national SAB rates over the next 1–2 years as the impact of the NHHI roll-out takes effect.
Our study had some limitations. First, our data are likely to be an underestimate as implementation occurred in a stepwise fashion, so that national HH compliance rates represent data from some sites with a longer history of the program than others. As the HH program becomes established throughout all Australian hospitals, the compliance rate should improve and become more homogeneous. Second, the HH compliance rate does not directly correlate with changes in the risk of disease transmission. Instead, a “power band” of disease reduction appears to occur when HH compliance rates improve to 55%–70% using the 5 Moments tool (Didier Pittet, Director, WHO Collaborating Centre on Patient Safety, Geneva, Switzerland, personal communication). Subsequent improvements in HH compliance are likely to be associated with continuing reductions in health care-associated disease rates, but other factors (eg, intravenous catheter insertion and maintenance protocols)11,20 may increasingly play a role, depending on the disease outcome being measured. Further research into quantifying the impact of various infection control measures on disease outcomes such as SAB is needed to identify priorities for interventions. Third, although SAB data were available for all states and territories, audit data for NSW were available from only four of the 12 principal referral hospitals,22 representing 22% of NSW acute public non-psychiatric hospital beds. However, we believe the inclusion of data from 41 of the state’s hospitals, including the large Hunter New England Area Health Service, allows a meaningful estimate of the national SAB rate. The adoption by all jurisdictions of a national definition of health care-associated SAB6 should provide more standardised and robust SAB data from 2011.
1 Hand hygiene compliance rates for public and private hospitals during the first 2 years of the National Hand Hygiene Initiative*
 | |||||||||||||||
2 National hand hygiene (HH) compliance rates 2 years after National Hand Hygiene Initiative implementation, by HH moment
 | |||||||||||||||
3 National hand hygiene compliance rates 2 years after National Hand Hygiene Initiative implementation, by HCW category6
 | |||||||||||||||
|
HCW = health care worker. n = moments audited per category in Period 3, 2010. | |||||||||||||||
Received 20 June 2011, accepted 3 October 2011
- M Lindsay Grayson1
- Philip L Russo1
- Marilyn Cruickshank2
- Jacqui L Bear3
- Christine A Gee4
- Clifford F Hughes5
- Paul D R Johnson1
- Rebecca McCann6
- Alison J McMillan7
- Brett G Mitchell8
- Christine E Selvey9
- Robin E Smith10
- Irene J Wilkinson11
- 1 Infectious Diseases Department, Austin Health, Melbourne, VIC.
- 2 Australian Commission on Safety and Quality in Health Care, Sydney, NSW.
- 3 ACT Health, Canberra, ACT.
- 4 Toowong Hospital, Brisbane, QLD.
- 5 Clinical Excellence Commission, Sydney, NSW.
- 6 Western Australian Department of Health, Perth, WA.
- 7 Victorian Department of Health, Melbourne, VIC.
- 8 Tasmanian Department of Health and Human Services, Hobart, TAS.
- 9 Queensland Health, Brisbane, QLD.
- 10 Northern Territory Department of Health, Katherine, NT.
- 11 SA Health, Adelaide, SA.
We are grateful to the many infection control professionals, HCWs and other staff throughout Australia who participated in the NHHI and assisted with data collection and analysis. The NHHI is funded by the Australian Commission on Safety and Quality in Health Care. Members of the HHA team, the National Hand Hygiene Advisory Committee and state/territory contributors are listed at http://www.hha.org.au/ mjasupplement.aspx. These data have been presented in part at the Australasian Society for Infectious Diseases Annual Scientific Meeting, Lorne, Victoria, April 2011.
No relevant disclosures.
- 1. Johnson PDR, Martin R, Burrell LJ, et al. Efficacy of an alcohol/chlorhexidine hand hygiene program in a hospital with high rates of nosocomial methicillin-resistant Staphylococcus aureus (MRSA) infection. Med J Aust 2005; 183: 509-514. <MJA full text>
- 2. Grayson ML, Jarvie LJ, Martin R, et al; Hand Hygiene Study Group and Hand Hygiene Statewide Roll-out Group, Victorian Quality Council. Significant reductions in methicillin-resistant Staphylococcus aureus bacteraemia and clinical isolates associated with a multisite, hand hygiene culture-change program and subsequent successful statewide roll-out. Med J Aust 2008; 188: 633-640. <MJA full text>
- 3. Pittet D, Hugonnet S, Harbarth S, et al; Infection Control Programme. Effectiveness of a hospital-wide programme to improve compliance with hand hygiene. Lancet 2000; 356: 1307-1312.
- 4. Grayson ML, Russo PL. The National Hand Hygiene Initiative [editorial]. Med J Aust 2009; 191: 420-421. <MJA full text>
- 5. World Health Organization. WHO guidelines on hand hygiene in health care. First Global Patient Safety Challenge: Clean Care is Safer Care. Geneva: WHO, 2009. http://whqlibdoc.who.int/publications/2009/9789241597906_eng.pdf (accessed Oct 2011).
- 6. Grayson ML, Russo PL, Cruickshank M, et al. Outcomes from the first 2 years of the Australian National Hand Hygiene Initiative: supplementary data. http://www.hha.org.au/mjasupplement.aspx (accessed Oct 2011).
- 7. Pantle AC, Fitzpatrick KR, McLaws ML, Hughes CF. A statewide approach to systematising hand hygiene behaviour in hospitals: Clean hands save lives, Part I. Med J Aust 2009; 191 (8 Suppl): S8-S12. <MJA full text>
- 8. Fitzpatrick KR, Pantle AC, McLaws ML, Hughes CF. Culture change for hand hygiene: Clean hands save lives, Part II. Med J Aust 2009; 191 (8 Suppl): S13-S17. <MJA full text>
- 9. McLaws ML, Pantle AC, Fitzpatrick KR, Hughes CF. Improvements in hand hygiene across New South Wales public hospitals: Clean hands save lives, Part III. Med J Aust 2009; 191 (8 Suppl): S18-S25. <MJA full text>
- 10. McLaws ML, Pantle AC, Fitzpatrick KR, Hughes CF. More than hand hygiene is needed to affect methicillin-resistant Staphylococcus aureus clinical indicator rates: Clean hands save lives, Part IV. Med J Aust 2009; 191 (8 Suppl): S26-S31. <MJA full text>
- 11. Collignon P, Dreimanis D, Ferguson J, et al. Bloodstream infection. In: Cruickshank M, Ferguson J, editors. Reducing harm to patients from healthcare associated infection: the role of surveillance. Sydney: Australian Commission on Safety and Quality in Health Care, 2008: 53-89. http://www.safetyandquality.gov.au/ internet/safety/publishing.nsf/Content/con-pubs-hai-report (accessed Sep 2011).
- 12. Grayson ML, Russo P, Ryan K, et al; editors. HHA manual: 5 Moments for hand hygiene. Melbourne: Hand Hygiene Australia, 2010. http://www.hha.org.au/ForHealthcareWorkers/manual.aspx (accessed Sep 2011).
- 13. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing; fifteenth informational supplement. M100-S15. Wayne, Pa: CLSI, 2005.
- 14. Australian Commission on Safety and Quality in Health Care. Draft data set specification. Surveillance of healthcare associated infections: Staphylococcus aureus bacteraemia & Clostridium difficile infection. Version 3.0. Sydney: ACSQHC, Oct 2010.
- 15. Australian Institute of Health and Welfare. Australian hospital statistics 2009–10. Canberra: AIHW, 2011. (AIHW Cat. No. HSE 107; Health Services Series No. 40.) http://www.aihw.gov.au/publication-detail/?id=10737418863&tab=2 (accessed Sep 2011).
- 16. Wagner AK, Soumerai SB, Zhang F, Ross-Degnan D. Segmented regression analysis of interrupted time series studies in medication use research. J Clin Pharm Ther 2002; 27: 299-309.
- 17. Xuereb C. How to best sell your message. Australasia – South East Asia Hand Hygiene Collaborative Inaugural Workshop; 2010 Jun 18–19; Palm Cove, Qld.
- 18. Collignon P, Nimmo GR, Gottlieb T, Gosbell IB; Australian Group on Antimicrobial Resistance. Staphylococcus aureus bacteremia, Australia. Emerg Infect Dis 2005; 11: 554-561.
- 19. Ferguson JK, Van Gessel H. Methicillin-resistant Staphylococcus aureus in hospitals: time for a culture change [letter]. Med J Aust 2008; 188: 62. <MJA full text>
- 20. Wilson J, Guy R, Elgohari S, et al. Trends in sources of meticillin-resistant Staphylococcus aureus (MRSA) bacteraemia: data from the national mandatory surveillance of MRSA bacteraemia in England, 2006–2009. J Hosp Infect 2011; 79: 211-217. [Epub Jul 20 ahead of print.]
- 21. Pearson A, Chronias A, Murray M. Voluntary and mandatory surveillance for methicillin-resistant Staphylococcus aureus (MRSA) and methicillin-susceptible S. aureus (MSSA) bacteraemia in England. J Antimicrob Chemother 2009; 64 Suppl 1: i11-i17.
- 22. Bureau of Health Information (NSW). NSW public hospital emergency departments by peer group. http://www.bhi.nsw.gov.au/publications/hospital_quarterly_report/PR_grp_report2 (accessed Sep 2011).





Abstract
Objective: To report outcomes from the first 2 years of the National Hand Hygiene Initiative (NHHI), a hand hygiene (HH) culture-change program implemented in all Australian hospitals to improve health care workers’ HH compliance, increase use of alcohol-based hand rub and reduce the risk of health care-associated infections.
Design and setting: The HH program was based on the World Health Organization 5 Moments for Hand Hygiene program, and included standardised educational materials and a regular audit system of HH compliance. The NHHI was implemented in January 2009.
Main outcome measures: HH compliance and Staphylococcus aureus bacteraemia (SAB) incidence rates 2 years after NHHI implementation.
Results: In late 2010, the overall national HH compliance rate in 521 hospitals was 68.3% (168 641/246 931 moments), but HH compliance before patient contact was 10%–15% lower than after patient contact. Among sites new to the 5 Moments audit tool, HH compliance improved from 43.6% (6431/14 740) at baseline to 67.8% (106 851/157 708) (P < 0.001). HH compliance was highest among nursing staff (73.6%; 116 851/158 732) and worst among medical staff (52.3%; 17 897/34 224) after 2 years. National incidence rates of methicillin-resistant SAB were stable for the 18 months before the NHHI (July 2007–2008; P = 0.366), but declined after implementation (2009–2010; P = 0.008). Annual national rates of hospital-onset SAB per 10 000 patient-days were 1.004 and 0.995 in 2009 and 2010, respectively, of which about 75% were due to methicillin-susceptible S. aureus.
Conclusions: The NHHI was associated with widespread sustained improvements in HH compliance among Australian health care workers. Although specific linking of SAB rate changes to the NHHI was not possible, further declines in national SAB rates are expected.