In 2005, the World Health Organization launched a Global Patient Safety Challenge with the aim of focusing every health care system simultaneously on the same key patient safety issues.1 The first challenge, Clean care is safer care, was chosen for the launch because hand hygiene is pivotal to patient safety.2 Low transglobal levels of compliance with hand hygiene practices by health care workers (HCWs) were the motivator for the WHO challenge. By 2007, the Clean care is safer care campaign had 45% of all countries pledging their commitment.
A decade before the launch, a study conducted in a Victorian paediatric intensive care unit (observed on weekdays between 7 am and 6 pm) showed that, on average, HCWs followed hand hygiene procedures 12.4% of the time before contact with a patient and 10.6% of the time after contact with a patient.3 After receiving feedback, the HCWs increased their hand hygiene compliance for both before- and after-patient contacts to half the level of recommended practice.3 In a Queensland study carried out in a tertiary referral hospital between 2000 and 2004, hand hygiene compliance by HCWs (observed for 15-minute periods between 9 am and 5 pm) ranged from 25% to 67% of the time for before-patient contact and from 27% to 70% of the time for after-patient contact.4 After rebuilding and relocation of the hospital (allowing greater accessibility to sinks for hand-washing), compliance by the same staff ranged from 15% to 47% for before-patient contact and from 23% to 72% for after-patient contact, with slightly higher rates among HCWs in the infectious diseases unit.4 In 2001, a 3-year program to improve hand hygiene practice in several Victorian hospitals began with an overall compliance rate of 21% (identified from 20-minute observation periods).5 In a New South Wales teaching hospital in 2001, hand hygiene compliance among HCWs (based on 2-hour continuous observation periods chosen at random between 9 am and 5 pm) ranged from 10% to 75% before patient contact and from 30% to 76% after patient contact.6
In March 2006, the Clinical Excellence Commission (CEC) and New South Wales Department of Health (NSW Health) launched the Clean hands save lives campaign, a statewide campaign with the primary aim of assisting HCWs with hand hygiene compliance before and after contact with patients.7 Secondary goals (described in detail elsewhere8) included improving the supply of alcohol-based hand rub (AHR) at the point of patient care, increasing HCWs’ awareness of hand hygiene, surveying patients’ opinions on the importance of hand hygiene and their perceptions of HCWs’ hand hygiene practices, and studying the acquisition of a select number of multiresistant organisms (MROs). This article reports on the impact on hand hygiene compliance of introducing AHR to all wards in all public hospitals in NSW.
The Clean hands save lives campaign was rolled out in all 208 NSW public health care facilities located in 11 health authorities — eight geographical area health services (AHSs), the Children’s Hospital at Westmead, the Ambulance Service of NSW and Justice Health. Details of the content of the campaign and timelines are provided elsewhere.8 The campaign aimed to increase awareness of AHR and improve hand hygiene compliance, with the eventual goal of reducing rates of acquisition of MROs in wards providing clinical care. The campaign did not address surgical scrub techniques; nor was it specifically aimed at improving hand hygiene in patients or hand hygiene associated with food handling.
Staff attended training workshops on observational survey methods in February 2006 (see Appendix for instructions). A standardised data collection tool was developed from survey tools used in the cleanyourhands campaign in the United Kingdom10 and the Geneva program11 (see Appendix for survey tool). Using this tool, hand hygiene surveys were undertaken by ward staff appointed by the nurse unit manager, facility director of nursing or nurse manager and trained by the hand hygiene project officer. Ward staff responsible for collecting the surveys and submitting data were trained and tested by the CEC. When required, they could receive support from one of the CEC-funded project officers in each AHS. Data were de-identified and submitted to the local AHS before further aggregation and submission to the CEC for analysis.
A pre-implementation overt survey of hand hygiene practices was undertaken by all hospitals in February and March 2006, and post-implementation surveys followed in August 2006, November 2006, February 2007 and July 2008. Observation methods were in line with the Geneva program described by Pittet et al11 and a program used by Johnson et al in an Australian hospital,5 except with regard to sample sizes. Representative sample sizes were determined by the NSW Hand Hygiene Steering Committee based on hospital category:
Group 1 (major teaching or referral hospitals): four 20-minute observation periods were conducted in each of three wards;
Group 2 (district hospitals): three 20-minute observation periods were conducted in each of three wards; and
Group 3 (community-based hospitals): two 20-minute observation periods were conducted in each of two wards.
Each occasion for hand-washing or using AHR before or after patient contact was regarded as a “hand hygiene opportunity”.11,12 An HCW who decontaminated his or her hands immediately after contact with a patient and then directly attended another patient — without touching any object (eg, medical notes, telephone, computer keyboard, monitor, curtain) or any other patient — was considered to have complied with before-patient hand hygiene practice in relation to the second patient. Hand hygiene compliance was expressed as a percentage, according to the following formula:
Compliance rate (%) = |
Hand hygiene events observed Hand hygiene opportunities |
× 100 (%) |
Observations were categorised by:
Fulkerson’s contact risk category (Box 1),9 a scale developed by the United States Centers for Disease Control and Prevention (CDC) to rank patient and environmental contacts for the potential to contaminate hands; and
Professional group:
Nurses: registered nurses, enrolled nurses, assistants in nursing, nurse managers, clinical nurse educators, clinical nurse consultants, clinical nurse specialists;
Doctors: visiting medical officers, consultants, registrars, junior medical officers, residents, interns, medical students;
Allied health staff: physiotherapists, occupational therapists, dietitians, social workers; or
Other staff: staff in environmental services, hotel services, security, pastoral care.
Small sample sizes for allied health staff and “other staff” groups meant that only the data for medical staff and nurses could be analysed by risk activity category. Data for hand hygiene opportunities that were associated with low- or medium-risk activities in accordance with Fulkerson’s scale9 were combined to increase sample sizes for analysis of compliance by nursing and medical staff, stratified by AHS and before- and after-patient contact. A median hand hygiene compliance rate was calculated across the eight AHSs. The pre-campaign rate in each AHS was used as the referent against which post-implementation rates (November 2006 to July 2008) were compared.
A minimum of 212 overt surveys, each lasting 20 minutes, was conducted in each of the five data collection periods. The mean number of hand hygiene opportunities observed for the five observation periods was 7747 (range, 6972–8251) (Box 2). Over the five observation periods, the hand hygiene burden was three times greater for nursing staff (24 197 opportunities) than for medical staff (7645 opportunities).
Hand hygiene compliance increased significantly from the pre-campaign rate of 47% (3795/8057) to 58% (4041/6972) by July 2008 (χ2, 405.0; P < 0.001) (Box 3). After the initial run-in period, the average compliance rate from November 2006 to July 2008 was 61% (14 335/23 448).
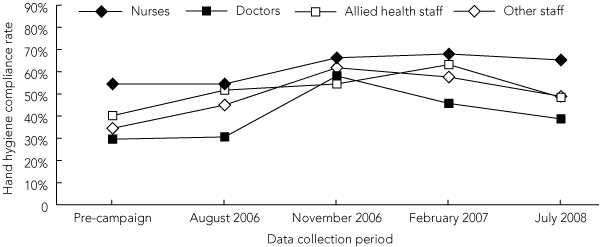
Over the campaign period, all professional groups, with the exception of medical staff, increased their hand hygiene compliance significantly (Box 4). Prior to the campaign, hand hygiene compliance by medical staff was 30% (417/1408). Although they had increased their compliance rate to 58% (1055/1816) by November 2006, the rate at 18 months after the intervention (July 2008) had dropped by 19 percentage points (PP). By contrast, nursing staff maintained significant improvement in hand hygiene compliance, with an average of 67% compliance (9718/14 601) across the three post-intervention data collection periods.
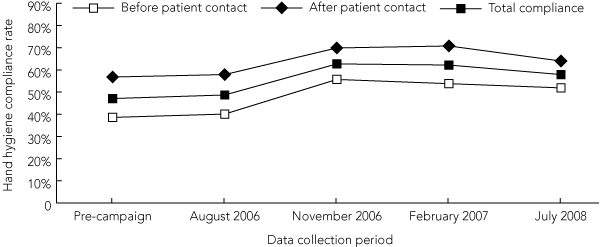
The rate of hand hygiene compliance before patient contact improved significantly, from 39% (1653/4287) to 52% (1822/3510) (P < 0.001), as did compliance after patient contact, from 57% (2142/3770) to 64% (2219/3462) (P < 0.001) (Box 5). After the initial run-in period, the average compliance rates before and after patient contact were 54% (6363/11 809) and 68% (7972/11 639), respectively. The higher compliance rate associated with after-patient contact compared with before-patient contact was consistent across all observation periods.
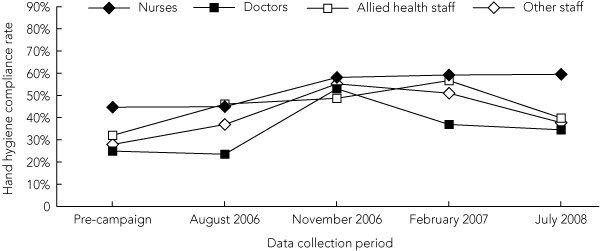
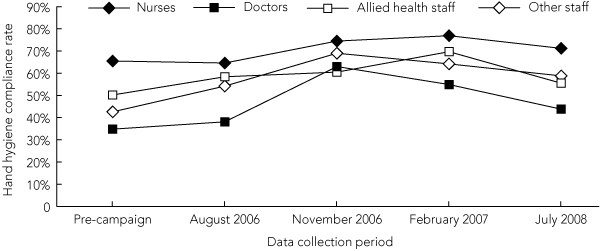
All professional groups except allied health staff increased their rates of hand hygiene compliance before patient contact over the observation periods (Box 6). Nursing staff maintained their compliance rates for before-patient contact at an average of 59% (4339/7361) from November 2006 to July 2008. All staff significantly increased their after-patient hand hygiene compliance (Box 7). Over the period November 2006 to July 2008, average after-patient compliance rates were 74% (5379/7240) for nursing staff and 55% (1311/2381) for medical staff.
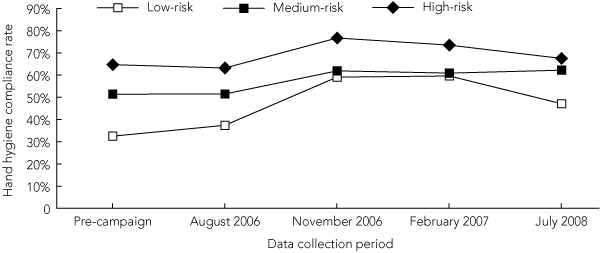
Staff responded to a key campaign message — hand hygiene associated with low- and medium-risk contacts — with significant increases in rates of hand hygiene compliance in relation to both low- and medium-risk contacts (Box 8). Compliance associated with medium-risk contacts increased from an average of 51% (4126/8017) in the earlier observation periods to an average of 62% (7070/11 461) between November 2006 and July 2008. The corresponding compliance rates associated with low-risk contacts were 35% (1742/5007) and 56% (4859/8678), respectively, and while this improvement was significant, it was not sustained at the same level to July 2008. Compliance rates associated with high-risk contacts also increased significantly, from an average of 64% (1447/2262) to an average of 73% (2406/3309).
Between November 2006 and July 2008, the average compliance rate associated with before-patient contacts of low and medium risk was 57% (3550/6240) for nursing staff and 41% (855/2090) for medical staff (Box 9). Over the same period, the average compliance rate associated with after-patient contacts of low and medium risk was 73% (4424/6082) for nursing staff and 54% (1086/2025) for medical staff. Thus, in relation to low- and medium-risk patient contacts, nursing staff were 1.4 times more likely than medical staff to comply with both before-patient hand hygiene practices (RR, 1.4 [95% CI, 1.3–1.5]; P < 0.001) and after-patient hand hygiene practices (RR, 1.4 [95% CI, 1.3–1.4]; P < 0.001). The rates of compliance by nurses and doctors both before and after low- or medium-risk contacts differed significantly across the nine AHSs.
We chose overt observation because it was a repeatable and easy method that could be sustained past the project timeline. This method, whose cost-effectiveness is supported by a WHO expert advisory panel,12 was based on the Geneva program,11 which has been successfully replicated in Australia.5 Patient mix, a wide range of individual hand hygiene practices and the Hawthorne effect13 will always have an impact on the measurement of compliance and cannot be overcome by indirect observation.14
The level of activity in a ward during 20-minute observation periods may underestimate the number of hand hygiene opportunities. However, the same methodology was used in the seminal Geneva study,11 which made a mean of 2869 observations over a total of 7 months between 1994 and 1997, and also in a Victorian study5 involving a mean of 711 observations made at baseline, 4 months and 12 months. Our rates were based on a mean of 7747 hand hygiene opportunities for five observation periods, making our study three times larger than the seminal study11 and 11 times larger than the Victorian study.5 As long as the measurement of hand hygiene compliance is fairly simple, it can be made to be strictly repeatable and result in the Hawthorne effect having a similar impact across survey periods. Although the potential for the Hawthorne effect was high in our study, we believe the impact was limited, as the compliance rate recorded in the first audit (August 2006) remained similar to the pre-campaign rate. Moreover, the Hawthorne effect from peer group pressure does not need to be considered a negative outcome, as it may provide positive assistance towards culture change.15
All 208 NSW public hospitals engaged in a 12-month rigorous campaign,8 collectively focusing on improving the cornerstone of infection control — hand hygiene. After an initial run-in period, the average compliance rate of 61% reflected an improvement of 14 PP overall (15 PP in before-patient compliance and 11 PP in after-patient compliance). By international standards, this can be considered a success, with similar projects on a smaller scale (using slightly different but repeatable observational methods) achieving compliance rates of 48% at 12 months16 and 63%–66% after 3 years.17,18 The Victorian project, conducted in 75 hospitals, achieved compliance rates of 48% at 12 months and 47% at 24 months, an improvement of 26 PP.16 Although the PP increase in our study was only half that of the Victorian study, our baseline compliance level began at the Victorian study’s final compliance level. While we commend our clinicians for their endeavour to change entrenched behaviour, there were individual HCWs whose hand hygiene compliance remained below a level that could be considered contributory to the overall success of the initial statewide promotion.
The baseline compliance rate for our nursing staff (55%) was higher than for any other clinical group, as was nurses’ average post-campaign compliance over the last three observational periods (67%). This 12 PP improvement was lower than the PP improvements for allied health staff (16 PP), doctors (18 PP) and other staff (22 PP). However, it must be remembered that the burden of contact for nursing staff is always higher than for other clinicians, with our nursing staff averaging three times the number of patient contacts made by medical staff. Doctors, allied health staff and other staff are more mobile throughout the hospital than nursing staff, and conceivably could move pathogenic organisms easily around the hospital. The relatively low compliance levels observed among our medical staff reflect a worldwide pattern.11,19-21 With nursing staff having the greatest number of patient contacts and medical staff being more mobile, how do we help these groups of clinicians achieve even higher levels of compliance?
At the time our campaign was being conducted, three different hand hygiene campaigns were being tested in a single Queensland teaching hospital.22 Each achieved vastly different rates of improvement. A passive intervention of simply providing AHR without any visual, verbal or administrative support resulted in rates remaining unaffected. When staff engaged in the design of their own campaign with overt administrative support, rates improved by an impressive 48%. The Geneva program in wards without a clinical champion produced little change. Yet wards using the Geneva program led by a clinical champion experienced an outstanding 56% improvement.22 As hand hygiene behaviour is strongly influenced by perceptions of peer group behaviour and the expectations of medical clinicians,15,19 future campaigns need to focus on our medical staff. Because nursing staff witness and respond to the hand hygiene behaviour of doctors,15,20 a focus on doctors will not only increase nurses’ hand hygiene compliance but also reinforce doctors’ own positive attitudes towards the behaviour.20
The behaviour affecting hand hygiene compliance in a hospital environment is complex. It has been hypothesised that hand hygiene behaviour among HCWs mirrors behaviour learnt in childhood, and that it is largely motivated by self-protection rather than protection of others.15 The principal drivers of hand hygiene are therefore dependent on the type of patient contact,17 with higher compliance associated with higher-risk contacts.4,11 The patterns of compliance observed in our study support this hypothesis. Where gloves were used for high-risk contacts, staff may have used either AHR or hand-washing, whereas AHR will most likely always be used with low-risk contacts.23 The most inspiring outcome of our campaign was the increase in hand hygiene compliance associated with low-risk contacts — from 33% before the campaign to an average of 56% in the post-intervention period. When low- and medium-risk contacts were aggregated, nurses across nine AHSs complied with just over half of before-patient contact opportunities and 73% of after-patient contact opportunities.
The next step for hand hygiene in Australian hospitals is to continue a roll-out of AHR. It is clear that further activity beyond the initial 1-year campaign is required to continue to improve hand hygiene compliance in NSW. Our qualitative interviews24 and the results of other studies support our belief that deliberate lack of cooperation by HCWs is not the issue. Rather, compliance associated with low-risk contacts mirrors beliefs among hospital staff and in the community about hand hygiene practice for self-protection.15 Campaigns that have not been behaviourally based have achieved important inroads,11,16,19 but none have achieved perfect compliance. The next steps towards improvement may be the hardest, but perhaps the most rewarding — behavioural interventions.
4 Hand hygiene compliance rates, by professional group
Hand hygiene compliance rate % (95% CI) [n/N] |
|||||||||||||||
5 Hand hygiene compliance before and after patient contact (all staff)
Hand hygiene compliance rate % (95% CI) [n/N] |
|||||||||||||||
6 Hand hygiene compliance before patient contact, by professional group
Hand hygiene compliance rate % (95% CI) [n/N] |
|||||||||||||||
7 Hand hygiene compliance after patient contact, by professional group
Hand hygiene compliance rate % (95% CI) [n/N] |
|||||||||||||||
8 Hand hygiene compliance, by patient contact risk category (all staff)
Hand hygiene compliance rate % (95% CI) [n/N] |
|||||||||||||||
9 Hand hygiene compliance associated with with low- and medium-risk patient contacts, by area health service (AHS) and before- or after-patient contact (nursing and medical staff), November 2006 to July 2008*
APPENDIX: hand hygiene observation tool and instructions
Hand hygiene observation tool — feedback form
Compliance rate (%) = |
Hand hygiene events observed Hand hygiene opportunities |
× 100 (%) |
Score by staff group (if requested):
The model used here is a modified version of the one used by Pittet et al9 and is used extensively internationally.
Compliance rate (%) = |
Hand hygiene events observed Hand hygiene opportunities |
× 100 (%) |
The methods used to observe HCWs’ hand hygiene compliance will be overt observations and will undoubtedly result in a “Hawthorne effect” and an additional hand hygiene learning opportunity. All observational periods will use this method, comparing compliance rates with similar biases.
- The staff member undertaking observation should undertake a number of practice observations to get familiar with the tool to reduce reporting bias.
- The overt observation technique will be used by just one person or with a partner.
- Identify an area within your ward/department where you can comfortably observe staff. Stay in this place for 20 minutes and observe your ‘window’ of activity. If staff walk away without you seeing whether they perform hand hygiene, you are to assume hand hygiene has not been undertaken.
- Observe for 20-minute periods.
- Using the observation sheet, tally hand hygiene opportunities and hand hygiene events observed for before- and after-patient care. If hand hygiene does not take place, leave it blank.
- The observation sheet offers you the chance to identify opportunities as low, medium or high and whether hand hygiene activity has taken place before and after patient contact.
- When you have completed 20 minutes’ observation, give feedback to the staff — a feedback form is included in this pack. When you give verbal feedback, try to stress positive findings first, and if you give negative feedback, give examples and suggestions for improvement. Also, ask for feedback from the observed staff about why they did not comply — use this as a learning feedback opportunity.
- Keep hold of the completed observations and hand them to the nurse unit manager. Provide a copy of the completed observations and feedback form to your AHS Hand Hygiene Project Officer, which will be forwarded to the Clinical Excellence Commission. These data will be de-identified.
- While you are observing, you may identify issues which are barriers to hand hygiene, eg, no soap, obstructed sinks, no alcohol hand rub by the bed, alcohol hand rub dispensers not working, alcohol hand rub dispensers empty – include this in your feedback.
- If you find activities which are not identified on the chart, add them and let the infection control team know.
Fulkerson risk scale for hand hygiene opportunities
Sterile or autoclaved materials
Thoroughly cleaned or washed materials
Materials that are not necessarily cleaned but are free from patient contact (ie, notes, papers, telephone and nurses’ desk area)
Materials in contact with patients but with little contamination risk (ie, furniture in patient area)
Objects or materials that have been in close contact with patients but are not contaminated with patient secretions or other sources of pathogenic bacteria (ie, relatively clean patient gowns, linen, used cutlery or plates, bed rails and tops of patient tables)
A patient: minimal contact without touching excretions or secretions and for a limited period of time, such as shaking hands, taking a pulse or giving a back rub
Materials and inanimate objects that have been in contact with, or bear, patient secretions such as saliva, not known to be contaminated
Setting up and removing intravenous infusion and giving injections (eg, subcutaneous, intramuscular or intravenous injections)
A patient: directly touching areas of secretions, such as mouth, nose and so forth
Materials contaminated with patient urine
Patient urine (direct contact)
Materials bearing faecal soilage
Faecal soilage (direct contact)
Materials that have been in direct contact with known infected secretions or excretions
Secretions or excretions known to be contaminated (direct contact)
Handling urine, faeces, blood (eg, bed pans, commodes, catheter bags)
Insertion and removal of intravenous cannulas
Infected patient sites, such as infected wounds (direct contact)
- Mary-Louise McLaws1
- Annette C Pantle2
- Kimberley R Fitzpatrick2
- Clifford F Hughes2
- 1 Hospital Infection Epidemiology and Surveillance Unit, School of Public Health and Community Medicine, University of New South Wales, Sydney, NSW.
- 2 Clinical Excellence Commission, Sydney, NSW.
Our project was a joint initiative of the CEC and NSW Health.
Mary-Louise McLaws has joined the WHO First Global Patient Safety Challenge pilot country project to provide epidemiological advice. Clifford Hughes operates the Australia and New Zealand Heart Valve Registry, which tracks patients with a Björk-Shiley convexo/concave heart valve. He distributes guidelines to these patients through their doctors as developed by the medical supervisory panel of the Bowling–Pfizer Heart Valve Settlement. He is the Principal Investigator of the On-X Heart Valve Study in Australia, for which On-X Life Technologies pays the expenses. He has received funding from Roche Diagnostics for travel expenses to attend meetings.
- 1. World Health Organization. The launch of the World Alliance for Patient Safety: “please do me no harm”. http://www.who.int/patientsafety/launch/en/print.html (accessed Sep 2008).
- 2. Pittet D, Donaldson L. Clean care is safer care: the first global challenge of the WHO World Alliance for Patient Safety. Am J Infect Control 2005; 33: 476-479.
- 3. Tibballs J. Teaching hospital medical staff to handwash. Med J Aust 1996; 164: 395-398. <MJA full text>
- 4. Whitby M, McLaws ML. Handwashing in healthcare workers: accessibility of sink location does not improve compliance. J Hosp Infect 2004; 54: 247-253.
- 5. Johnson PDR, Martin R, Burrell LJ, et al. Efficacy of an alcohol/chlorhexidine hand hygiene program in a hospital with high rates of nosocomial methicillin-resistant Staphylococcus aureus (MRSA) infection. Med J Aust 2005; 183: 509-514. <MJA full text>
- 6. Bahal A, Karamchandani D, Fraise AP, McLaws ML. Hand hygiene compliance: universally better post-contact than pre-contact in healthcare workers in the UK and Australia. Br J Infect Control 2007; 8: 24-28.
- 7. Clinical Excellence Commission, NSW Health. Clean hands save lives: final report of the NSW Hand Hygiene Campaign. Sydney: NSW Health, 2007. http://www.cec.health.nsw.gov.au/moreinfo/cleanhands_report.html (accessed Aug 2009).
- 8. Pantle AC, Fitzpatrick KR, McLaws ML, Hughes CF. A statewide approach to systematising hand hygiene behaviour in hospitals: Clean hands save lives, Part I. Med J Aust 2009; 191 (8 Suppl): S8-S12. <MJA full text>
- 9. Rotter ML. Hand washing and hand disinfection. In: Mayhall CG, editor. Hospital epidemiology and infection control. Baltimore: Williams and Wilkins, 1996: 1052-1068.
- 10. Gould DJ, Hewitt-Taylor J, Drey NS, et al. The CleanYourHands Campaign: critiquing policy and evidence base. J Hosp Infect 2007; 65: 95-101.
- 11. Pittet D, Hugonnet S, Harbarth S, et al. Effectiveness of a hospital-wide programme to improve compliance with hand hygiene. Infection Control Programme. Lancet 2000; 356: 1307-1312.
- 12. World Health Organization. WHO guidelines on hand hygiene in health care (advanced draft): a summary. Clean hands are safer hands. Geneva: WHO, 2005. http://www.who.int/patientsafety/events/05/HH_en.pdf (accessed Nov 2008).
- 13. Whitby M, McLaws ML. Methodological difficulties in hand hygiene research. J Hosp Infect 2007; 67: 194-195.
- 14. Haas JP, Larson EL. Measurement of compliance with hand hygiene. J Hosp Infect 2007; 66: 6-14.
- 15. Whitby M, McLaws ML, Ross MW. Why healthcare workers don’t wash their hands: a behavioral explanation. Infect Control Hosp Epidemiol 2006; 27: 484-492.
- 16. Grayson ML, Jarvie LJ, Martin R, et al. Significant reductions in methicillin-resistant Staphylococcus aureus bacteraemia and clinical isolates associated with a multisite, hand hygiene culture-change program and subsequent successful statewide roll-out. Med J Aust 2008; 188: 633-640. <MJA full text>
- 17. Boyce JK, Ligi C, Kohan C, et al. Lack of association between the increased incidence of Clostridium difficile-associated disease and the increasing use of alcohol-based hand rubs. Infect Control Hosp Epidemiol 2006; 27: 479-483.
- 18. Albert RK, Condie F. Hand-washing patterns in medical intensive care units. N Engl J Med 1981; 304: 1465-1466.
- 19. Larson E, Killien M. Factors influencing handwashing behavior of patient care personnel. Am J Infect Control 1982; 10: 93-99.
- 20. Sax H, Uçkay I, Richet H, et al. Determinants of good adherence to hand hygiene among healthcare workers who have extensive exposure to hand hygiene campaigns. Infect Control Hosp Epidemiol 2007; 28: 1267-1274.
- 21. Pittet D, Mourouga P, Perneger TV. Compliance with handwashing in a teaching hospital. Infection Control Program. Ann Intern Med 1999; 130: 126-130.
- 22. Whitby M, McLaws ML, Slater K, et al. Three successful interventions in healthcare workers that improve compliance with hand hygiene: is sustained replication possible? Am J Infect Control 2008; 36: 349-355.
- 23. Whitby M, Pessoa-Silva CL, McLaws ML, et al. Behavioural considerations for hand hygiene practices: the basic building blocks. J Hosp Infect 2006; 65: 1-8.
- 24. Fitzpatrick KR, Pantle AC, McLaws ML, Hughes CF. Culture change for hand hygiene: Clean hands save lives, Part II. Med J Aust 2009; 191 (8 Suppl): S13-S18. <MJA full text>





Abstract
Objective: To describe improvements in hand hygiene compliance after a statewide hand hygiene campaign conducted in New South Wales public hospitals.
Design and setting: The campaign was conducted in all area health services in NSW (covering all 208 public hospitals). Alcohol-based hand rub (AHR) was introduced into all hospitals between March and June 2006. In each hospital, five overt observation surveys of hand hygiene compliance by health care workers (HCWs) were conducted: one pre-implementation survey and four post-implementation surveys (in August 2006, November 2006, February 2007 and July 2008).
Main outcome measures: Overtly observed hand hygiene compliance rates by HCWs, stratified by before- and after-patient contact, Fulkerson’s contact risk categories, and four health care professional groupings.
Results: The overall hand hygiene compliance rate improved from 47% before the intervention to an average of 61% over the last three observation periods (P < 0.001). All professional groups sustained improved compliance rates except medical staff, whose practices reverted to pre-intervention rates. Nursing staff maintained significantly improved compliance, with an average rate of 67% after the intervention. Overall hand hygiene compliance before patient contact improved from 39% (pre-campaign) to 52% (July 2008) (P < 0.001). Overall compliance after patient contact improved from 57% to 64% (P < 0.001) over the same period. Compliance associated with medium-risk contacts increased from an average of 51% in the first two observation periods to an average of 62% over the last three observation periods (P < 0.001). The corresponding compliance rates associated with low-risk contacts were 35% and 56%, respectively (P < 0.001).
Conclusion: An overall improvement in hand hygiene rates was achieved with the introduction of AHR. Increased adherence to before-patient contact compliance, especially by nursing staff, contributed to the progress made, but an acceptable overall level of hand hygiene practice is yet to be achieved. It is now time to focus on a long-term behavioural change program directed specifically at medical staff.