Endemic methicillin-resistant Staphylococcus aureus (MRSA) adds to the net burden of nosocomial sepsis, costs more per patient than methicillin-sensitive S. aureus infection, and independently predicts poorer patient outcome.1-6
The most important mode of transmission of MRSA within institutions appears to be poor hand hygiene.7 Once introduced into a hospital, MRSA can be spread until a large silent reservoir of colonised patients develops. By the time serious infections such as bacteraemia draw attention to the problem, asymptomatic colonisation will typically be widespread8 and contamination of the environment may be extensive.9,10 These observations suggest that improved hand hygiene by health care workers and better cleaning of shared hospital equipment could reduce the likelihood of patients becoming colonised and lead to subsequent reductions in MRSA infection.
The introduction of alcohol/chlorhexidine hand hygiene solution (ACHRS) combined with education and motivation programs can improve hand hygiene compliance and reduce total nosocomial infections.11 In view of this, alcohol-based hand hygiene has been recommended as a clinical standard for hospitals in the United States.12 However, there is uncertainty about the efficacy of these programs in institutions where nosocomial MRSA is endemic but isolation or cohorting of MRSA-colonised patients is not practical.
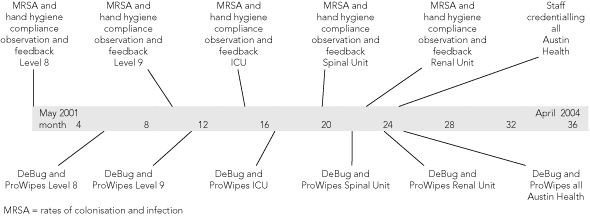
MRSA is the major nosocomial pathogen affecting most large hospitals along the eastern seaboard of Australia.13,14 Early in 2001, we planned to measure and then reduce the burden of MRSA in our hospital in preparation for relocation to a new facility (opened in May 2005). Before the move, we recognised that we did not have the resources to isolate all MRSA-colonised patients, even though this may be the ideal means of control. Instead, modelled on the approach taken by Pittet et al,11 we designed and implemented a 3-year program (Operation Clean Start) to try to improve hand hygiene compliance, to introduce better cleaning of shared ward equipment, and to offer MRSA decolonisation to a targeted group of past carriers on readmission to selected wards. This article describes the implementation and outcome of the program.
Austin Health is an 840-bed University of Melbourne teaching hospital, with three separate campuses. Despite a longstanding policy to isolate patients with MRSA-infected sputum or discharging wounds, our infrastructure limitations have prevented full implementation of this policy.
Operation Clean Start (OCS) was an integrated 3-year program consisting of MRSA screening of patients, health care workers, and the environment; feedback of results; and introduction of a series of specific interventions, which were supported by a detailed educational and promotional package. OCS was introduced from May 2001 in a stepwise, timetabled manner to five “sentinel areas” (Box 1), which had been identified as having high rates of MRSA infection, all located at the main acute campus (Austin Hospital). The sentinel areas comprised two clusters of acute-surgical wards (level 8 [3 wards] and level 9 [3 wards]), the intensive care unit (ICU), the renal unit and the spinal injuries unit. From April 2003, aspects of OCS were extended across the remaining 23 wards and all three campuses. The program was funded by a Quality Improvement Fund grant from the Department of Human Services, Victoria (AUD$584 000 over 3 years).
We developed our own alcohol/chlorhexidine hand hygiene solution (ACHRS), similar to that used by Pittet et al,11 which could be applied repeatedly without causing irritation or leaving a sticky residue (DeBug; 70% isopropyl alcohol, 0.5% chlorhexidine, skin emollient; manufactured under licence by Orion Laboratories, Western Australia). Dispensers for 500 mL bottles of ACHRS were placed in defined locations, including at the foot of every patient bed, on intravenous (IV) catheter trolleys and wound dressing trolleys, at nurses’ stations and outside patient rooms. Changes were made to ward assistants’ workplace agreements to ensure prompt replacement when bottles were empty. Education sessions were conducted regarding the correct use of ACHRS.15 Any health care worker who reported cutaneous adverse reactions was referred to one dermatologist with expertise in occupational dermatology;16 OCS covered any associated medical costs.
The introduction of ACHRS and alcohol-impregnated wipes to each sentinel ward was accompanied by a detailed promotional package designed to foster a new clean culture. We organised promotional events, including coffee break seminars, and provided promotional materials such as quizzes with prizes, a newsletter, T-shirts, wine-labels, “DeBug” pens, and notices attached to pay advice slips. We developed specific promotional slogans: DeBug before and after every patient contact (for ACHRS use) and Clean between every re-use (alcohol-impregnated wipes for shared equipment); and hired an advertising consultant to optimise the delivery of our messages. These slogans were repeated and displayed widely, initially in sentinel areas and later throughout the institution. Feedback sessions were held with senior nurses on sentinel wards to provide information regarding recent outcome data. Medical, surgical and nursing grand-rounds on MRSA were held; these included discussions of the OCS program. Where appropriate, ward staff presented their ward’s data to engender a sense of ownership.
We introduced our own version of “Talking Walls”.11 Health care workers in each sentinel area generated ideas for a poster that promoted aspects of OCS using art and humour. An artist (also an Austin Health nurse) captured and refined these concepts and created a large coloured wall poster that was printed and displayed throughout the institution.
To ensure that newly appointed staff and students were fully aware of OCS and the new hand hygiene culture, we developed a computer-based educational package that could be accessed online (www.DeBug.net.au). With support from senior hospital management, we changed employment commencement procedures at Austin Health so that successful completion of the learning package became a requirement before staff or students could receive a security identification badge or be registered in the hospital’s pay system.
We measured hand hygiene compliance using methods based on those of Pittet et al11 and recently described by Brown et al.17 Nurse observers (R M, J O’K, L J B, D E, W B, C B) were standardised against each other to reduce inter-observer error. Compliance was assessed in each sentinel area immediately before the introduction of OCS and 4 and 12 months later. Appropriate compliance was when the observed health care worker used ACHRS, disposable gloves or another acceptable hand hygiene option in a manner consistent with previously published definitions, for general wards and for the ICU.11,15 In routine (non-ICU) wards and the ICU, targets of > 600 and > 800, respectively, hand hygiene opportunities were observed (about 40–60 and 60, respectively, observation sessions) to ensure statistical power to compare compliance rates between observation periods.
Observation of health care workers in patient care areas was undertaken during morning and afternoon shifts using a repeated block randomisation schedule to ensure that similar numbers of observations were conducted in each patient care bay. Each assigned patient care bay was observed for 20 minutes unless “no activity” occurred during a 2 minute period, following which observers proceeded to the next assigned bay. “No activity” was defined as either no health care worker entering the room, all health care worker activities performed behind closed curtains (except in the ICU), or no patient present. Although some health care workers could be observed more than once, the observation strategy meant that most health care workers were observed at some time.
In sentinel areas, surveillance of MRSA colonisation of patients (nose and groin cultures) was conducted twice weekly for at least 4 weeks before intervention and for a similar period 4 and 12 months after intervention. Health care workers were assessed anonymously for MRSA colonisation by culturing swabs from nares and hands weekly for 4 weeks before intervention and in a similar manner 4 and 12 months after intervention.
Swabs from both nares of patients and health care workers and both groins from patients were cultured on mannitol salt agar (Oxoid, Basingstoke, UK) containing 4 μg/mL oxacillin (MSO). Health care worker hands were tested by taking impressions of finger tips and nails directly onto MSO. Environmental contamination was assessed using MSO contact plates and sterile saline moistened swabs inoculated into 10 mL tryptone soya broth (Oxoid, Basingstoke, UK), which were later sub-cultured on MSO. The presence of MRSA was verified by standard methods.18
The MRSA colonisation rate for patients was defined as the number of patients with MRSA in nose and/or groin as a proportion of the total number of patients assessed during each observation period. The health care worker colonisation rate was defined as the proportion of anonymous hand and nose samples from which MRSA was cultured. The environmental MRSA contamination rate was the proportion of positive results obtained during each period.
Although not part of OCS, we retrospectively assessed the rate of laboratory detection of extended-spectrum β-lactamase (ESBL)-producing Escherichia coli and Klebsiella spp. (ESBLs) using the same method and periods as for MRSA. ESBL-producing E. coli or Klebsiella isolates were those obtained from clinical specimens with a minimum inhibitory concentration (MIC) to either ceftazidime or ceftriaxone of > 1 μg/mL and a confirmatory test demonstrating at least three twofold reductions in MIC to that antibiotic in the presence of clavulanic acid.19
Differences in proportions were assessed using Fisher’s exact test (Stata 8.2 for Windows; Stata Corporation, College Station, Tex, USA), and confidence intervals were derived from the standard error of the proportion and T-scores for the sample using Excel 2000 for Windows (Microsoft Corporation, Redmond, Calif, USA).
Trends for MRSA (or ESBL) rates over time were assessed using interrupted time-series segmented regression analysis.20 Statistical analyses of the slopes of the linear regression curves were performed using GraphPad Prism version 4.0 for Windows (GraphPad Software, San Diego, Calif, USA).
Percentage reduction was calculated from the differences in rates measured in May 2001 (commencement of OCS) and April 2004 (completion of OCS), as determined by the linear regression curves. Isolates or patient-episodes of bacteraemia “prevented” are notional (not measured events) and were computed from the difference in area under the fitted curve and the hypothetical projected curve. Confidence intervals (95%) for these estimates were derived from the confidence intervals produced by GraphPad for the linear regression curve.
A mean (SD) of 711 (329) (median, 668; range, 329–1554) hand hygiene opportunities was observed in each sentinel area at each time point. Overall hand hygiene compliance improved from 21% (95% CI, 20.3%–22.9%) at baseline to 41% (95% CI, 39.5%–43.0%) at 4 months, and was maintained at 42% (40.2%–43.8%) at 12 months (Box 2). In individual sentinel areas, compliance rates improved significantly between pre-intervention and 4 months post-intervention in all areas (P < 0.001). On level 9 and in the spinal unit there were further improvements between 4 and 12 months. Notably, the 12-month observations on level 8 showed a return to baseline. When this was investigated, we found 80% of the health care workers assessed on this occasion had been new student nurses who had received no education regarding ACHRS or OCS. When hand hygiene compliance was reassessed 2 weeks later on level 8 without these students, rates were similar to the 4-month result and more similar to other areas (Box 2).
Use of ACHRS products increased in all sentinel areas, from a mean of 5.7 L/1000 patient bed-days pre-intervention to 28.6 L/1000 patient bed-days during the last 3 months of OCS (range, 16.5 L/1000 patient bed-days [level 8] to 91.8 L/1000 patient bed-days [ICU]) (Box 3).
MRSA colonisation was assessed in more than 90% of eligible patients: 1507 patients pre-intervention and 627 and 697 patients, 4 and 12 months post-intervention, respectively. Rates of patient MRSA colonisation varied between wards, with long-stay units such as the spinal unit having colonisation rates of 40%–50%, but did not change in any of the sentinel areas during OCS (Box 4A). Of the 1283 patients screened for MRSA colonisation post-intervention, 178 (13.9%) were positive (41 patients were tested at both 4 and 12 months post-intervention).
About 1% of patients admitted to Austin Health were identified as “ever” colonised with MRSA in the past and were offered decolonisation during the 36-month intervention period. Nursing staff compliance with this intervention appeared poor, with less than 20% of patients receiving all doses of prescribed medication in one audit (data not shown).
Health care workers were assessed for colonisation on 1166 separate occasions (> 80% of those eligible): 52 (4.4%) were colonised (28 [2.4%] hands, 19 [1.6%] nose, 5 [0.4%] both). Rates of health care worker MRSA colonisation (nose, hands or both) did not decrease in sentinel areas except in the ICU (Box 4B) (P = 0.02, Fisher’s exact test).
A total of 2882 hospital environmental cultures were collected, of which 14% were positive before intervention and 15.5% were positive 12 months after intervention. Among the various sites sampled, the following were most notable (number of cultures; percentage positive cultures): blood pressure cuffs (483; 12%), bed curtains (391; 7.7%), stethoscopes (287; 7%), tap handles (447; 3.6%) and drug/IV trolleys (144; 15.3%).
Environmental contamination did not change significantly during OCS, with the exception of level 9, where there was a transient improvement, and the renal unit, in which contamination was worse at 12 months (Box 4C).
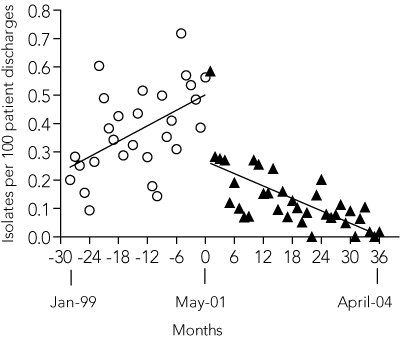
The rate of total clinical MRSA isolates per 100 patient-discharges per month was static during the 28-month pre-intervention period (slope = 0; P = 0.85), but declined significantly in the 36-month post-intervention period (slope < 0; P < 0.001). By the 36th month, the rate had decreased by 40% (95% CI, 23%–58%) compared with baseline (Box 5A). We calculated that 1008 fewer MRSA clinical isolates than expected were obtained in this period (95% CI, 575–1440 isolates).
Similarly, for patient-episodes of MRSA bacteraemia, the monthly rate during the 28-month pre-intervention period was static (slope = 0; P = 0.93), but fell significantly in the post-intervention period (slope < 0; P = 0.003; by 36 months post-intervention the rate was 0.39/1000 discharges) (Box 5B). By the 36th month, the monthly rate of MRSA bacteraemia had decreased by 57% compared with baseline (95% CI, 38%–74%). We calculated that 53 fewer patient-episodes of MRSA bacteraemia than expected occurred during this period (95% CI, 36–68 episodes). Raw data for rates of infection and bacteraemia are available from the authors.
The mean number of bacteriology specimens per month submitted to the microbiology laboratory in the two periods increased slightly in the post-intervention period (4477 specimens [755 blood cultures] pre-intervention; 4661 [861 blood cultures] post-intervention).
We have confirmed the findings by Pittet et al,11 in which the introduction and promotion of ACHRS to a large teaching hospital was followed by sustained improvements in both hand hygiene compliance and reductions in MRSA infections. Since their report, other smaller studies have reported benefits from ACHRS programs.21-23 Only one group assessed an institution-wide approach such as ours, but unlike us, they systematically screened patients and isolated those with MRSA.21 Although isolation and cohorting of MRSA colonised patients is recommended,24-26 hospital design may prevent the use of this option, and a recent study suggests it may not be effective, at least in the ICU.27 Our study shows that, even in a highly MRSA-endemic setting, rates of clinical MRSA infection, including bacteraemia, can be cut substantially by promoting ACHRS, improving cleaning of shared ward equipment, and using targeted patient decontamination. Although isolation and cohorting of MRSA-infected patients would have been optimal, this was not practical given our hospital infrastructure.
At the commencement of OCS, we expected to uncover a silent reservoir of MRSA-colonised patients, but were surprised at the extent of the problem. Thirteen per cent of all screened patients were colonised, and about 15% of all environmental sites tested were contaminated. The failure of OCS to reduce rates of patient MRSA colonisation and environmental MRSA contamination initially disappointed us. However, our hospital’s infrastructure was old and difficult to clean (hence the reason for building our new facility), which may partly explain the continuing high levels of environmental contamination.
Although rates of health care worker colonisation with MRSA did not change in four of five sentinel areas, there was significant improvement in the ICU. Potentially, ready access to ACHRS may mean that health care workers’ hands are less likely to be contaminated in the ICU and other critical areas where the likelihood of performing high-risk tasks such as attending central or peripheral cannula dressings or surgical wounds is highest.28 Partially interrupting the link between colonisation and infection may explain the effect of the OCS program on clinical MRSA infection rates, but not on MRSA colonisation per se.29
The practical realities of maintaining high levels of hand hygiene education were highlighted by the effect of a large group of new nursing students 12 months after OCS had been introduced on level 8. This episode led directly to the development of our web-based educational package. Because this was short and accessible through the Internet, we could reasonably insist that all new students and staff complete the package before commencing work. To introduce this requirement, we sought and received unqualified support from the Austin Health Executive.
Estimates of the rates of hospital-acquired MRSA bacteraemia in 17 Australian hospitals from 1999 to 2002 were recently published.30 In that report, same-day admissions with MRSA bacteraemia were counted as community-acquired. With the caveat that we included all cases of MRSA bacteraemia, our post-intervention rate of 0.39/1000 discharges appears broadly similar to the Australian median of 0.22/1000 admissions (range, 0–0.89).
We recognise that our findings are subject to a number of caveats. Firstly, OCS was a quality improvement project, not primarily research, and consisted of several simultaneous interventions. We cannot therefore determine the relative merits of each intervention.
Secondly, the use of computer databases to measure total clinical isolates of MRSA (or ESBLs) overestimates the true burden of nosocomial infections. However, this method is robust and easier to perform than chart reviews, provides institution-wide data, and avoids artificial assumptions about when or where an individual became infected within our institution.
Because the number of microbiology specimens received by our laboratory did not alter significantly between the two periods, it is unlikely that a systematic change in the way clinicians ordered pathology tests has confounded our results. Furthermore, the close correlation between patient-episodes of MRSA bacteraemia and total clinical isolates confirmed that our interventions resulted in real reductions in clinical infections.
Other nosocomial pathogens, such as vancomycin-resistant enterococci (VRE), Clostridium difficile, or multiresistant Acinetobacter spp, could also have been assessed. However, despite two outbreaks of VRE colonisation detected by targeted screening and managed with strict patient isolation and improved cleaning, we observed only three cases of VRE bacteraemia (one pre-OCS, two in the OCS intervention period). Multiresistant gram-negative infections, apart from ESBLs, were rare and, although we do not actively monitor C. difficile infection, the rate of new isolation of this pathogen fell 30% by the third year of OCS (data not shown).
Thirdly, the reliance on historical controls could mean that changes apart from our interventions are responsible for the improvements we observed. The “Hawthorn effect” (the tendency to improve just because you are being observed) may have contributed. Also, a computerised antibiotic control program has been increasingly used at our institution in the past 3 years, but this has resulted in stable rates of antibiotic use, rather than reductions in use. It is also possible that changes in antibiotic use or infection control policies outside our hospital may have altered the prevalence of ESBLs and MRSA infections among our patients — but this seems unlikely.
We did not perform a detailed cost analysis of OCS. However, we estimated that to maintain the OCS program at the same level of intensity (labour, promotions, ACHRS, mupirocin and alcohol-impregnated wipes) would require about $180 000 per year. This is equivalent to about $2.50 per discharged patient — roughly two-thirds the cost of a Big Mac. Although detailed Australian financial data are lacking, US estimates suggest that a case of MRSA bacteraemia has a median attributable cost of about US$27 000.31 Thus, for our program to be cost-neutral, we would need to prevent 5–6 patients from developing MRSA bacteraemia annually in our hospital. We estimate that we have prevented about 17 cases per year during the 3 years of OCS. Even allowing for higher hospital costs in the US, our program is likely to have been highly cost-effective.
Active surveillance, screening and cohorting of colonised patients for MRSA control is probably the ideal in countries that can afford it, and we are hoping to move towards this now that we have new hospital facilities and many more single rooms. However, since Semmelweis in 1847,32 we have known that compliance by health care workers with effective hand hygiene remains the single most important way to prevent nosocomial infections. ACHRS can improve hand hygiene compliance because of its efficacy, convenience and acceptability.16,33,34
Our program has shown that improvements in MRSA control can be made under less than ideal circumstances in Australian hospitals. However, ACHRS and other interventions need to be introduced as part of a concerted hand hygiene culture-change program which includes a readily accessible education initiative that instils and maintains the new hand hygiene culture that must prevail if MRSA and other nosocomial infections are to be better controlled.
2 Health care worker hand hygiene compliance
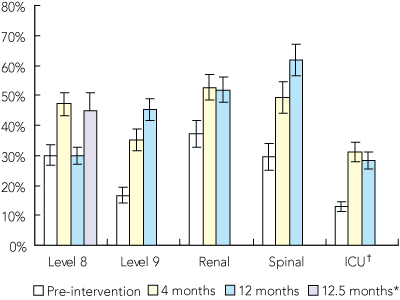
The bars show proportion of observed hand-hygiene opportunities in which recommended protocols were followed, with 95% confidence intervals.
* The screening was repeated 2 weeks after the 12-month assessment on level 8 because results were confounded by a group of new student nurses who had just arrived on the ward. †The method for measuring compliance is not directly comparable between the intensive care unit (ICU) and other areas.
3 Use of alcohol/chlorhexidine solution
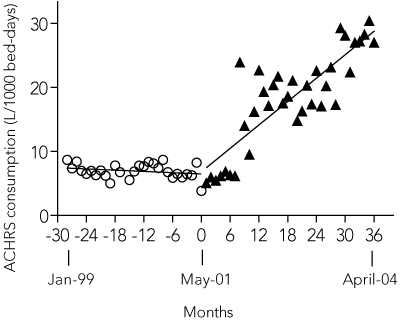
Total use of alcohol/chlorhexidine hand hygiene solution (ACHRS) in the five sentinel wards before and after intervention. Fitted linear regression curves are shown for each period.
4 MRSA colonisation rates and hospital contamination
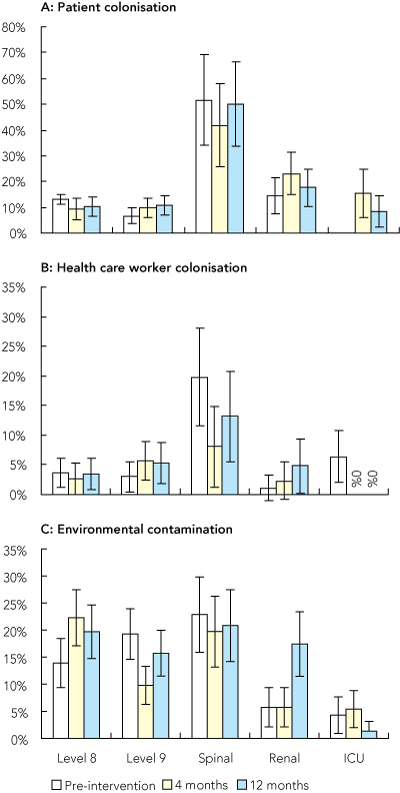
A: Proportion of patients with MRSA detected in nose or groin. No pre-intervention data available for the intensive care unit (ICU). B: Proportion of anonymous samples from health care workers in which MRSA was detected. C: Proportion of positive results from environmental sampling.
In the ICU only, health care worker colonisation rates 4 and 12 months post-intervention were significantly lower than baseline.
5 MRSA isolates and patient-episodes of bacteraemia
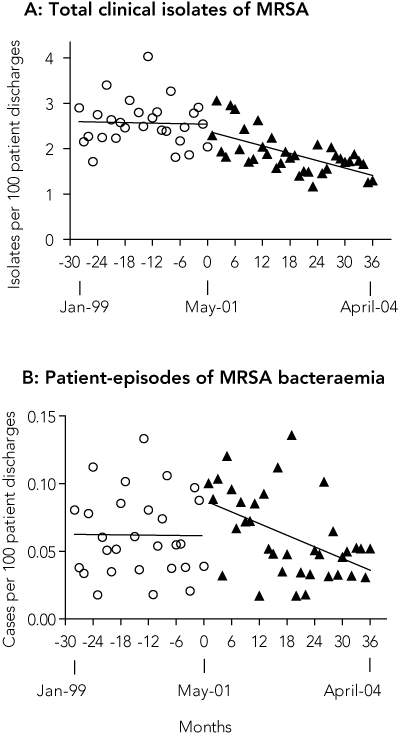
A: Total clinical isolates of MRSA and patient-episodes of MRSA bacteraemia before and after intervention for all Austin Health. Fitted linear regression curves are shown for each period. All clinical isolates, including multiple isolates from the same patient, were included, but screening swabs were specifically excluded.
B: Patient episodes of MRSA bacteraemia before and after intervention for all Austin Health. Fitted linear regression curves are shown for each period. Only the first isolate was recorded for a given patient, unless 14 days with negative cultures had elapsed, in which case a subsequent episode(s) was recorded.
6 Clinical isolates of ESBL-producing E. coli and Klebsiella spp.
Total clinical isolates of ESBL-producing E. coli and Klebsiella spp. before and after intervention for all Austin Health. Fitted linear regression curves are shown for each period. All clinical isolates, including multiple isolates from the same patient, were included, but screening swabs were specifically excluded.
Received 1 July 2005, accepted 19 September 2005
- Paul D R Johnson1
- Rhea Martin2
- Laurelle J Burrell3
- Elizabeth A Grabsch4
- Susan W Kirsa5
- Jason O’Keeffe6
- Barrie C Mayall7
- Deidre Edmonds8
- Wendy Barr9
- Christopher Bolger10
- Humsha Naidoo11
- M Lindsay Grayson12
- Austin Health, Heidelberg, VIC.
We thank Sara Elkerton from pharmacy for assistance with product useage data and Courtney Thornely, Natalie Plumbley and Shirley Xie from Microbiology for assistance with MRSA screening.
DeBug™ (a trademark for the hand hygiene product referred to in this article) was developed by the authors (employees of Austin Health) with funding in part from the Victorian Department of Human Services. The intellectual property for this development is held by Austin Health, which handles all patent, trademark and licensing issues. Austin Health, but no individual author, receives a small income stream from the sale of DeBug™.
- 1. Kopp BJ, Nix DE, Armstrong EP. Clinical and economic analysis of methicillin-susceptible and -resistant Staphlococcus aureus infections. Ann Pharmacother 2004; 38: 1377-1382.
- 2. McHugh CG, Riley LW. Risk factors and costs associated with methicillin-resistant Staphylococcus aureus bloodstream infections. Infect Control Hosp Epidemiol 2004; 25: 425-430.
- 3. Engemann JJ, Carmeli Y, Cosgrove SE, et al. Adverse clinical and economic outcomes attributable to methicillin resistance among patients with Staphylococcus aureus surgical site infection. Clin Infect Dis 2003; 36: 592-598.
- 4. Selvey LA, Whitby M, Johnson B. Nosocomial methicillin-resistant Staphylococcus aureus bacteremia: is it any worse than nosocomial methicillin-sensitive Staphylococcus aureus bacteremia? Infect Control Hosp Epidemiol 2000; 21: 645-648.
- 5. Abramson MA, Sexton DJ. Nosocomial methicillin-resistant and methicillin-susceptible Staphylococcus aureus primary bacteremia: at what costs? Infect Control Hosp Epidemiol 1999; 20: 408-411.
- 6. Whitby M, McLaws ML, Berry G. Risk of death from methicillin-resistant Staphylococcus aureus bacteraemia: a meta-analysis. Med J Aust 2001; 175: 264-267.
- 7. Boyce JM. MRSA patients: proven methods to treat colonization and infection. J Hosp Infect 2001; 48: S9-S14.
- 8. Farr BM. Methicillin-resistant Staphylococcus aureus infections. Curr Infect Dis Rep 1999; 1: 328-333.
- 9. Boyce JM, Potter-Bynoe G, Chenevert C, King T. Environmental contamination due to methicillin-resistant Staphylococcus aureus: possible infection control implications. Infect Control Hosp Epidemiol 1997; 18: 622-627.
- 10. Oie S, Hosokawa I, Kamiya A. Contamination of room door handles by methicillin-sensitive/methicillin-resistant Staphylococcus aureus. J Hosp Infect 2002; 51: 140-143.
- 11. Pittet D, Hugonnet S, Harbarth S, et al. Effectiveness of a hospital-wide programme to improve compliance with hand hygiene. Infection Control Programme. Lancet 2000; 356: 1307-1312.
- 12. Boyce JM, Pittet D. Guideline for hand hygiene in health-care settings. Recommendations of the Healthcare Infection Control Practices Advisory Committee and the HICPAC/SHEA/APIC/IDSA Hand Hygiene Task Force. Society for Healthcare Epidemiology of America/Association for Professionals in Infection. Control/Infectious Diseases Society of America. MMWR Morb Mortal Wkly Rep 2002; 51: 1-45.
- 13. Turnidge JD, Nimmo GR, Francis G. Evolution of resistance in Staphylococcus aureus in Australian teaching hospitals. Australian Group on Antimicrobial Resistance (AGAR). Med J Aust 1996; 164: 68-71.
- 14. McLaws ML, Murphy C, Whitby M. Standardising surveillance of nosocomial infections: the HISS program. Hospital Infection Standardised Surveillance. J Qual Clin Pract 2000; 20: 6-11.
- 15. Pittet D, Boyce JM. Revolutionising hand hygiene in health-care settings: guidelines revisited. Lancet Infect Dis 2003; 3: 269-270.
- 16. Graham M, Nixon R, Burrell LJ, et al. Low rates of cutaneous adverse reactions to alcohol-based hand hygiene solution during prolonged use in a large teaching hospital. Antimicrob Agents Chemother 2005; 49: 4404-4405.
- 17. Brown TL, Burrell LJ, Edmonds D, et al. Hand-hygiene: a standardised tool for assessing compliance. Aust Infect Control 2005; 10: 51-58.
- 18. Murray PR, Baron EJ, Jorgensen JH, et al, editors. Manual of clinical microbiology. 8th ed. Herndon, VA: American Society for Microbiology, 2003.
- 19. Performance standards for antimicrobial susceptibility testing: fourteenth informational supplement document. NCCLS document MS100-S14. Wayne, PA: Clinical and Laboratory Standards Institute, 2004.
- 20. Wagner AK, Soumerai SB, Zhang F, Ross-Degnan D. Segmented regression analysis of interrupted time series studies in medication use research. J Clin Pharm Ther 2002; 27: 299-309.
- 21. Tomic V, Svetina Sorli P, Trinkaus D, et al. Comprehensive strategy to prevent nosocomial spread of methicillin-resistant Staphylococcus aureus in a highly endemic setting. Arch Intern Med 2004; 164: 2038-2043.
- 22. Silvestri L, Petros AJ, Sarginson RE, et al. Handwashing in the intensive care unit: a big measure with modest effects. J Hosp Infect 2005; 59: 172-179.
- 23. Tvedt C, Bukholm G. Alcohol-based hand disinfection: a more robust hand-hygiene method in an intensive care unit. J Hosp Infect 2005; 59: 229-234.
- 24. Verhoef J, Beaujean D, Blok H, et al. A Dutch approach to methicillin-resistant Staphylococcus aureus. Eur J Clin Microbiol Infect Dis 1999; 18: 461-466.
- 25. British Society for Antimicrobial Chemotherapy, Hospital Infection Society, Infection Control Nurses Association. Revised guidelines for the control of methicillin-resistant Staphylococcus aureus infection in hospitals. J Hosp Infect 1998; 39: 253-290.
- 26. Hospital Infection Control Practices Advisory Committee. Guideline for isolation precautions in hospitals, part II: recommendations for isolation precautions in hospitals. Am J Infect Control 1996; 24: 32-52.
- 27. Cepeda JA, Whitehouse T, Cooper B, et al. Isolation of patients in single rooms or cohorts to reduce spread of MRSA in intensive-care units: prospective two-centre study. Lancet 2005; 365: 295-304.
- 28. Huskins WC, Goldmann DA. Controlling methicillin-resistant Staphylococcus aureus, aka “Superbug”. Lancet 2005; 365: 273-275.
- 29. Harbarth S, Martin Y, Rohner P, et al. Effect of delayed infection control measures on a hospital outbreak of methicillin-resistant Staphylococcus aureus. J Hosp Infect 2000; 46: 43-49.
- 30. Collignon P, Nimmo GR, Gottlieb T, Gosbell IB, Australian Group on Antimicrobial Resistance. Staphylococcus aureus bacteremia, Australia. Emerg Infect Dis 2005; 11: 554-561.
- 31. Abramson MA, Sexton DJ. Nosocomial methicillin-resistant and methicillin-susceptible Staphylococcus aureus primary bacteremia: at what costs? Infect Control Hosp Epidemiol 1999; 20: 408-411.
- 32. Semmelweis IF, Carter KC. The etiology, concept, and prophylaxis of childbed fever. Madison, WI: University of Wisconsin Press, 1983.
- 33. Wimer AF. Replace hand washing with use of a waterless alcohol hand rub? Clin Infect Dis 2000; 31: 136-143.
- 34. Zaragoza M, Salles M, Gomez J, et al. Handwashing with soap or alcoholic solutions? A randomized clinical trial of its effectiveness. Am J Infect Control 1999; 27: 258-261.





Abstract
Objective: To assess the effect of a multifaceted hand hygiene culture-change program on health care worker behaviour, and to reduce the burden of nosocomial methicillin-resistant Staphylococcus aureus (MRSA) infections.
Design and setting: Timetabled introduction of interventions (alcohol/chlorhexidine hand hygiene solution [ACHRS], improved cleaning of shared ward equipment, targeted patient decolonisation, comprehensive “culture change” package) to five clinical areas of a large university teaching hospital that had high levels of MRSA.
Main outcome measures: Health care worker hand hygiene compliance; volume of ACHRS used; prevalence of patient and health care worker MRSA colonisation; environmental MRSA contamination; rates of clinical MRSA infection; and rates of laboratory detection of ESBL-producing Escherichia coli and Klebsiella spp.
Results: In study wards, health care worker hand hygiene compliance improved from a pre-intervention mean of 21% (95% CI, 20.3%–22.9%) to 42% (95% CI, 40.2%–43.8%) 12 months post-intervention (P < 0.001). ACHRS use increased from 5.7 to 28.6 L/1000 bed-days. No change was observed in patient MRSA colonisation or environmental colonisation/contamination, and, except in the intensive care unit, colonisation of health care workers was unchanged. Thirty-six months post-intervention, there had been significant reductions in hospital-wide rates of total clinical MRSA isolates (40% reduction; P < 0.001), patient-episodes of MRSA bacteraemia (57% reduction; P = 0.01), and clinical isolates of ESBL-producing E. coli and Klebsiella spp (90% reduction; P < 0.001).
Conclusions: Introduction of ACHRS and a detailed culture-change program was effective in improving hand hygiene compliance and reducing nosocomial MRSA infections, despite high-level MRSA endemicity.