The known: In early overseas reports, mortality among patients with COVID‐19 admitted to intensive care was very high.
The new: During the first four months of the pandemic in Australia, mortality among invasively ventilated COVID‐19 patients was lower than in some overseas reports, and the median duration of intensive care longer. Median peak intensive care unit bed occupancy by patients with COVID‐19 was 14%.
The implications: The prognosis for patients with severe COVID‐19 disease may not be as poor in Australia as overseas, but the intensive care resource burden may be greater. Our findings could inform critical care planning, crucial as the COVID‐19 pandemic continues.
The coronavirus disease 2019 (COVID‐19) pandemic has massively increased the burden on intensive care units (ICUs) around the world. Despite advanced intensive care support, including invasive mechanical ventilation and renal replacement therapy, ICU mortality rates of 40–80% were reported for early case series.1,2,3 In the control arm of a large randomised trial of dexamethasone treatment in the United Kingdom, 28‐day mortality for invasively ventilated patients was 40%.4 ICU mortality rates for patients with COVID‐19 have been substantially higher than during epidemics of viral pneumonitis, including during the 2009 H1N1 influenza pandemic (14–23%).5,6
Many reports on outcomes for patients with COVID‐19, however, were from countries in which the number of cases exceeded the capacity of the health care systems. In parts of China, Italy, and New York, the rapid increase in numbers permitted little time for preparation, resulting in shortages of beds,7 equipment (including personal protective equipment and ventilators), and appropriately trained staff.8,9 But mortality may have been underestimated in some reports, as many patients were still undergoing treatment at the time of reporting.10,11
In Australia, the first recorded case of COVID‐19 was identified on 25 January 2020,12 and 8566 infections had been confirmed by 5 July 2020.13 During this period, the Short PeRiod IncideNce sTudy of Severe Acute Respiratory Infection (SPRINT‐SARI) Australia collected comprehensive observational data for patients with COVID‐19 admitted to ICUs, with the aims of improving our understanding of the natural history of the disease and providing contemporary local data on ICU outcomes and resource use.
Methods
Study design and setting
SPRINT‐SARI Australia is a multicentre, prospective, observational study of patients with COVID‐19 admitted to 77 participating public and private ICUs in all Australian states and territories; these units had been invited by the Australian and New Zealand Intensive Care Society (ANZICS) Clinical Trials Group or were already affiliated with SPRINT‐SARI Australia. The study design, case report form, and protocol were developed in conjunction with the International Severe Acute Respiratory and emerging Infection Consortium (ISARIC; https://isaric.tghn.org/sprint-sari). The standardised case report form, which enabled a rapidly scalable data collection platform for acquiring clinical information and sharing, was developed in response to multiple outbreaks of severe acute respiratory infection over the past 10 years.14 SPRINT‐SARI Australia is supported by the ANZICS Clinical Trials Group and is coordinated by the Australian and New Zealand Intensive Care Research Centre (ANZIC‐RC; https://www.monash.edu/medicine/sphpm/anzicrc/home) at Monash University.
Australian ICUs are predominantly staffed by fulltime specialists. The decision to admit a patient to an ICU is largely determined by their likely response to treatment, their prognosis, and the expected long term outcome.15 The standard nurse to patient ratio in Australian ICUs is 1:1, or 1:2 for high dependency patients.16
Participants
The study population included patients of any age who had severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) infections confirmed by polymerase chain reaction (PCR) and index COVID‐19‐related admissions to participating ICUs. We excluded PCR‐negative patients and those whose test results were not available by the end of the study period. Samples for PCR testing were collected from the nasopharynx, trachea, or lower airways by bronchoscopy according to local policy.
Data collection
Data for ICU admissions reported to SPRINT‐SARI Australia by 77 ICUs across Australia during 27 February 2020 – 30 June 2020 were extracted on 28 July 2020. The participating ICUs, including most of the major metropolitan ICUs, included a total of 1187 ICU beds. Dedicated research staff at each ICU screened all admissions for patients with COVID‐19. Data from electronic medical records or paper notes were entered into a database (REDCap) without personal identifiers. The ANZIC‐RC maintained the database and performed all analyses. Start‐up meetings, a detailed data dictionary, and quality checks were completed to ensure data quality and protocol standardisation, and to minimise bias.
We collected data on baseline demographic and clinical characteristics. The Acute Physiology and Chronic Health Evaluation II (APACHE‐II) score17 and the Sequential Organ Failure Assessment (SOFA) score18 for the first 24 hours in intensive care were calculated when possible. Data on investigations, ICU treatments, and interventions were collected daily until day 28 of the ICU admission. Outcomes were recorded as death or ICU length of stay.
To assess the comprehensiveness of our data collection, we cross‐referenced SPRINT‐SARI Australia admissions data with data collected by the Australian Department of Health, according to which 225 patients with COVID‐19 had been admitted to ICUs to 5 July 2020.13 To determine maximum site occupancy, we divided the peak number of COVID‐19 cases by the total number of ICU beds at each study site, as reported by the Australian and New Zealand Intensive Care Society Centre for Outcomes and Resource Evaluation.19
Statistical analysis
Median ICU length of stay was derived from Kaplan–Meier survival curves, censored at the date of the final daily record for patients without ICU discharge dates. The ICU mortality rate, with exact binomial 95% confidence intervals (CIs), is reported. We used Weibull survival regression analysis to assess risk factors for ICU death and length of stay in patients alive at discharge.20 Time to ICU death was defined as time from ICU admission to date of death, censored at either ICU discharge or the date of the final daily record for patients alive and still in intensive care. ICU length of stay for patients was modelled as time from ICU admission to ICU discharge, censored for both death and those still in intensive care. Age, sex, APACHE‐II score, and invasive ventilation were included as variables in multivariate models because of their clinical relevance. Further variables were selected by a forward stepwise approach and likelihood ratio test; the criterion for variable removal was P ≥ 0.05; for inclusion, P < 0.01. A parametric survival model with a Weibull distribution was fitted that incorporated ICU site as a random effect.20 Hazard ratios (HRs) with 95% CIs are reported. We included all available data in our analyses, and did not impute missing data. All analyses were performed in Stata 16.
Ethics approval
Approval for data collection, with a waiver of informed consent by patients, was granted under the National Mutual Acceptance scheme, and by the Human Research Ethics Committees of Alfred Health (reference, HREC/16/Alfred/59) and the individual sites. Research governance approval was granted by the Chief Health Officers of South Australia and Victoria, and supported by the Chief Health Officer of Queensland. Individual site research governance approval was granted at all sites where it was required.
Results
Of the 77 participating ICUs, 44 had admitted at least one patient with confirmed COVID‐19 during the 18‐week study period, 32 sites had admitted none, and one had not completed data entry. ICU bed occupancy peaked on 5 April 2020 (total, 90 patients), falling to fewer than 25 patients by early May (Box 1). Median peak ICU bed occupancy at hospitals was 14% (interquartile range [IQR], 9–16%; range 4–40%) (Supporting Information, figure 1). The most frequently reported ICU nurse to patient ratios were 1:1 on 1766 of 2270 patient‐days (77.8%) and 1:2 on 171 patient‐days (7.5%).
Clinical characteristics and intensive care treatment
The participating ICUs admitted 204 of the 225 patients with COVID‐19 admitted to Australian ICUs during the study period13 (91%; Supporting Information, figure 2). Of the 204 patients, 140 were men (69%) and 64 women (31%); their median age was 63.5 years (IQR, 53–72 years). The most frequently reported comorbid conditions were obesity (80 patients, 39%), diabetes (57, 28%), hypertension treated with angiotensin‐converting enzyme inhibitors or angiotensin II receptor blockers (49, 24%), and chronic cardiac disease (40, 20%); 73 patients (36%) reported no comorbidity. Median duration from symptom onset to hospital admission was 6 days (IQR, 3–9 days), and from hospital admission to ICU admission, one day (IQR, 0–3 days). The median APACHE‐II score on ICU day 1 (available for 195 patients) was 14 (IQR, 10–18); the median SOFA score (available for 98 patients) was 6 (IQR, 4–10). Infection was acquired during international travel by 114 patients (56%), including 55 cruise ship passengers (27%). Close contact with a person with confirmed or probable COVID‐19 infection was reported by 92 patients (45%); 16 patients (8%) were health care workers (Box 2).
Invasive ventilation was provided for 119 patients (58%). Their median age (68 years; IQR, 57–73 years) was greater than for the 85 patients not requiring invasive ventilation (61 years; IQR, 46–69 years), and larger proportions of ventilated patients were obese (44% v 33%), or had diabetes (37% v 15%) or chronic cardiac disease (23% v 15%); a smaller proportion had chronic pulmonary disease (6% v 11%) (Box 2).
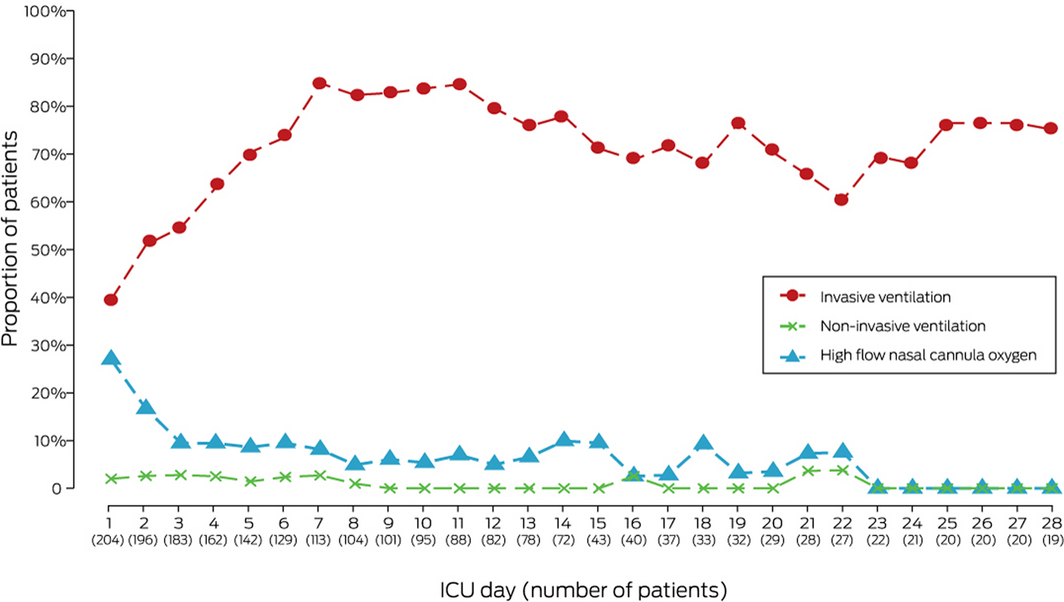
Seventy‐nine of 204 patients (39%) commenced invasive mechanical ventilation on ICU day 1, and 54 (26%) were supported with high flow oxygen therapy (Box 3). The number of invasively ventilated patients increased to 94 of 113 (83%) by the end of the first week. Four patients (2%) underwent non‐invasive ventilation on day 1 and three of 113 (3%) on day 7. The most frequent additional interventions were inotropic agents (111 of 204 patients, 54%), neuromuscular blockade (87, 43%), prone positioning (56, 27%), and corticosteroids (58, 28%); renal replacement therapy (23 of 204 patients, 11%) and veno‐venous extracorporeal membrane oxygenation (ECMO) (two, 1%) were used less frequently. Thirty‐two patients (16%) received hydroxychloroquine.
Clinical outcomes
Hospital outcomes follow‐up data were complete for 194 of 204 patients (95%); four patients were still receiving intensive care at the end of the study period, and six were receiving care in other hospital wards. The median ICU length of stay for the 204 patients was 16 days (IQR, 9–28 days) for invasively ventilated patients, and 3 days (IQR, 2–5 days) for patients who had not required invasive mechanical ventilation (log‐rank test, P < 0.001) (Box 4). Of the 119 mechanically ventilated patients, 27 (23%) were in intensive care for 30 days or longer. Mixed effects survival regression analysis of length of stay in ICU indicated that invasive ventilation (adjusted HR, 0.07; 95% CI, 0.04–0.11) and renal replacement therapy (adjusted HR, 0.44; 95% CI, 0.24–0.80) were each associated with a lower probability of being discharged alive from ICU at any given time point (Supporting Information, tables 1 and 2).
Thirty of the 200 ICU patients not in intensive care at the end of the study period had died (15%; 95% CI, 10–21%), including 26 of 117 patients requiring invasive ventilation (22%; 95% CI, 15–31%) and four of 83 who did not (5%; 95% CI, 1–12%). All but two of the patients who died were aged 60 years or more (Box 5). In univariate analyses, ICU death was more frequent for patients over 64 years of age, those with higher APACHE‐II scores on day 1, and patients with chronic cardiac, pulmonary, or kidney disease (Box 6). After adjusting for age, sex and invasive ventilation, APACHE‐II score on day 1 (adjusted HR, 1.15; 95% CI, 1.09–1.21) and chronic cardiac disease (adjusted HR, 3.38; 95% CI, 1.46–7.83) significantly influenced the risk of ICU death (Box 7).
Discussion
In our study of patients admitted to ICUs during the first four months of the COVID‐19 pandemic in Australia, we found that mortality among invasively ventilated patients (22%) was markedly lower than reported overseas, despite the relatively high proportion of patients for whom final outcomes were known. Further, the median ICU length of stay (16 days) was longer than overseas (7–14 days),1,2,3 and 23% of invasively ventilated patients were in intensive care for 30 days or more.
Several factors may underlie these differences. The numbers of COVID‐19 patients in some European countries, the United States, and parts of China increased so rapidly that they quickly overwhelmed health care system capacity; further, the proportions of patients invasively ventilated on day 1 of intensive care in these countries were also very high (70–90%).2,10,11,21 In our study, the number of COVID‐19 patients was low (peak ICU bed occupancy, 90 patients), and only 79 of the 204 patients (39%) commenced invasive ventilation during their first 24 hours in intensive care (Box 3). Moreover, patients were distributed across many hospitals, and the proportion of ICU beds occupied by people with COVID‐19 at any single site remained low (median, 14%).
As the capacity of the health care system in Australia was never challenged, patients may have had access to intensive care earlier in the course of their illness, thereby benefiting from interventions associated with lower mortality.22,23 The rapid rise in infection numbers in Wuhan was associated with higher mortality rates than in parts of China where the rise in infections was slower.24
Further, the patients in our study differed from those in overseas reports in several key features. First, the most frequent source of infection was international travel (56% of patients), whereas locally acquired infections dominated in many other countries.1,10 These travellers may have been healthier than people who were infected locally, particularly those infected in long term care facilities.1 The median age of patients in our study (63 years) was similar to that reported in the United States (64 years)11 and Italy (63 years),2 but lower than in the United Kingdom (73 years).10 In addition, comorbidity was less common in our study. A greater number of comorbid conditions increases the risks of ICU admission, the need for mechanical ventilation, and death for patients with COVID‐19.25
We also found that ICU length of stay for invasively ventilated patients was longer (median, 16 days) than reported in the United Kingdom (9.7 days)10 and the United States (12 days).11 While the reported frequency of other ICU support strategies, including proning (28–40%), neuromuscular blockade (39%), renal replacement therapy (20%), and ECMO (0–3%)10,11 were similar to the rates in our study, the longer duration of support in Australian ICUs may have been possible only because the system had the capacity to provide it.
During the early phase of the pandemic, COVID‐19 in Australian ICUs was associated with resource‐intense, protracted admissions, and a low mortality rate. It remains to be seen whether this situation, which has important implications for service provision and planning, would apply during a further rise in case numbers, particularly in the context of more widespread community transmission. Our findings indicate that the resource burden of caring for these patients is substantial, and, if not met, could also affect outcomes.25
Strengths and limitations
Our study included most major ICUs in Australia, providing nationally representative data for a health care system operating within its capacity. The data were collected by experienced data collectors on a standardised case report form that included daily data fields for the first 28 days of each ICU admission. The long follow‐up allowed near complete outcomes data. Limitations included the observational nature of the study and potential confounding by therapeutic and other factors that influence mortality.
We did not collect data on patients with COVID‐19 not admitted to ICUs, so our study did not explore decision making about admission to intensive care. This may limit the generalisability of our findings, but we chose a pragmatic approach that allowed us to rapidly collect, analyse, and report data on the interventions and outcomes for patients with confirmed COVID‐19 admitted to Australian ICUs. Further, as we focused on intensive care interventions, treatments provided prior to ICU admission that could have affected the primary outcomes were not assessed. Finally, it is uncertain whether our findings will apply to patients with COVID‐19 treated in Australian ICUs during the future course of the pandemic.
Conclusion
During the early phase of the pandemic in Australia, mortality among patients with COVID‐19 admitted to ICUs was lower and their stay in intensive care longer than reported overseas. Our findings reinforce the importance of ensuring adequate local ICU capacity, particularly as the end of the pandemic is not yet in sight.
Box 1 – Cumulative number of intensive care unit (ICU) admissions of patients with confirmed COVID‐19, and number of ICU beds occupied by patients with COVID‐19, by day,* 27 February 2020 – 30 June 2020
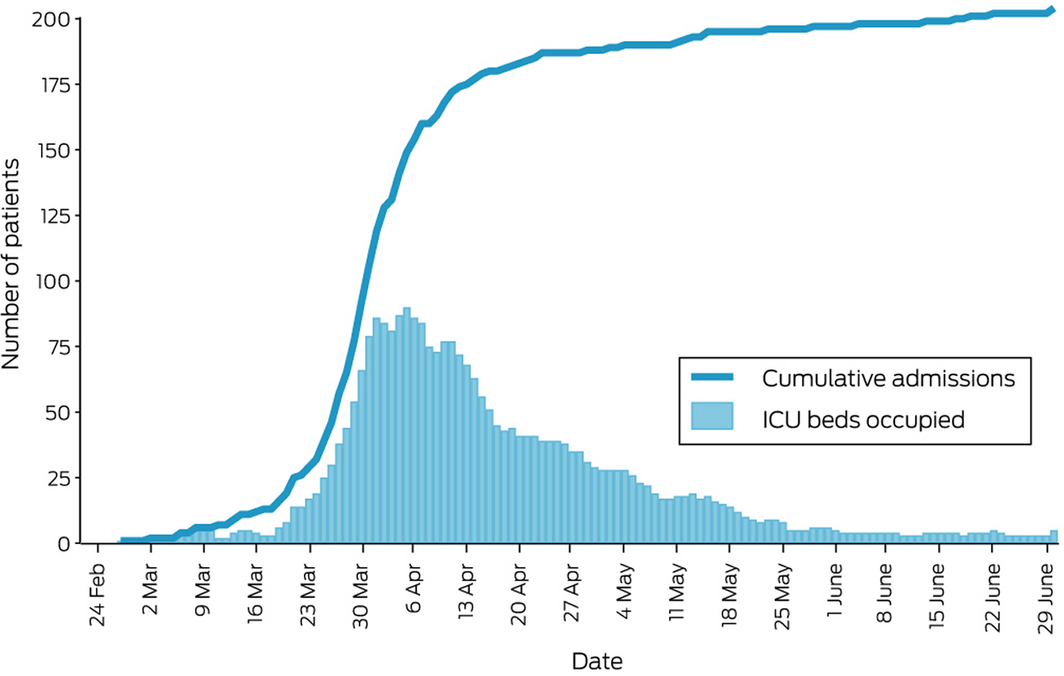
* According to ICU admission and discharge dates; if no ICU discharge date was recorded, it was assumed that the patient still occupied an ICU bed.
Box 2 – Characteristics of 204 patients with confirmed COVID‐19 admitted to participating intensive care units, 27 February 2020 – 30 June 2020
|
Characteristic |
With invasive ventilation |
Without invasive ventilation |
Total |
||||||||||||
|
|
|||||||||||||||
|
Number of patients |
119 |
85 |
204 |
||||||||||||
|
Age (years), median (IQR) |
68 (57–73) |
61 (46–69) |
63.5 (53–72) |
||||||||||||
|
≤ 54 |
22 (18%) |
33 (39%) |
55 (27%) |
||||||||||||
|
55–64 |
27 (23%) |
23 (27%) |
50 (24%) |
||||||||||||
|
65–85 |
70 (59%) |
29 (34%) |
99 (48%) |
||||||||||||
|
Sex |
|
|
|
||||||||||||
|
Women |
37 (31%) |
27 (32%) |
64 (31%) |
||||||||||||
|
Men |
82 (69%) |
58 (68%) |
140 (69%) |
||||||||||||
|
Body mass index* (kg/m2), median (IQR) |
29.7 (24.8–33.3) |
28.5 (23.6–32.1) |
28.8 (24.6–32.3) |
||||||||||||
|
Underweight (< 18.5) |
1 (0.8%) |
1 (1%) |
2 (1%) |
||||||||||||
|
Normal weight (18.5–24.9) |
28 (24%) |
24 (29%) |
52 (26%) |
||||||||||||
|
Overweight (25–29.9) |
30 (25%) |
21 (25%) |
51 (25%) |
||||||||||||
|
Obese (≥ 30) |
52 (44%) |
28 (34%) |
80 (40%) |
||||||||||||
|
Not stated |
7 (6%) |
9 (11%) |
16 (8%) |
||||||||||||
|
Smoking status |
|
|
|
||||||||||||
|
Never smoker |
94 (79%) |
75 (88%) |
169 (83%) |
||||||||||||
|
Smoking history |
20 (17%) |
7 (8%) |
27 (13%) |
||||||||||||
|
Not stated |
5 (4%) |
3 (4%) |
8 (4%) |
||||||||||||
|
APACHE‐II score on ICU day 1,† median (IQR) |
17 (14–9) |
10 (6–13) |
14 (10–18) |
||||||||||||
|
SOFA score on ICU day 1 |
|
|
|
||||||||||||
|
Patients (all components for computation recorded) |
65 |
33 |
98 |
||||||||||||
|
Median score (IQR) |
8 (6–11) |
4 (3–6) |
6 (4–10) |
||||||||||||
|
Comorbid conditions |
|
|
|
||||||||||||
|
Diabetes |
44 (37%) |
13 (15%) |
57 (28%) |
||||||||||||
|
Hypertension‡ |
29 (24%) |
20 (24%) |
49 (24%) |
||||||||||||
|
Chronic cardiac disease |
27 (23%) |
13 (15%) |
40 (20%) |
||||||||||||
|
Asthma |
12 (10%) |
10 (12%) |
22 (11%) |
||||||||||||
|
Chronic pulmonary disease |
7 (6%) |
9 (11%) |
16 (8%) |
||||||||||||
|
Chronic immunosuppression |
7 (6%) |
6 (7%) |
13 (6%) |
||||||||||||
|
Chronic kidney disease |
8 (7%) |
3 (4%) |
11 (5%) |
||||||||||||
|
Chronic haematologic disease |
7 (6%) |
2 (2%) |
9 (4%) |
||||||||||||
|
Liver disease |
2 (2%) |
3 (4%) |
5 (2%) |
||||||||||||
|
None reported |
37 (31%) |
36 (42%) |
73 (36%) |
||||||||||||
|
Returning international traveller |
60 (50%) |
54 (64%) |
114 (56%) |
||||||||||||
|
Cruise ship traveller |
28 (24%) |
27 (32%) |
55 (27%) |
||||||||||||
|
Close contact with person with confirmed or probable COVID‐19 infection |
56 (47%) |
36 (42%) |
92 (45%) |
||||||||||||
|
Health care worker |
7 (6%) |
9 (11%) |
16 (8%) |
||||||||||||
|
Symptom onset to hospital admission (days) |
|
|
|
||||||||||||
|
Patients (both dates reported) |
103 |
82 |
185 |
||||||||||||
|
Median (IQR) |
6 (3–9) |
6 (3–9) |
6 (3–9) |
||||||||||||
|
Symptom onset to ICU admission (days) |
|
|
|
||||||||||||
|
Patients (both dates reported) |
105 |
82 |
187 |
||||||||||||
|
Median (IQR) |
8 (5–11) |
8 (5–11) |
8 (5–11) |
||||||||||||
|
Hospital admission to ICU admission (days) |
|
|
|
||||||||||||
|
Patients (both dates reported) |
119 |
85 |
204 |
||||||||||||
|
Median (IQR) |
1 (0–3) |
0 (0–2) |
1 (0–3) |
||||||||||||
|
|
|||||||||||||||
|
APACHE‐II = Acute Physiology And Chronic Health Evaluation II; IQR = interquartile range; SOFA = Sequential Organ Failure Assessment. * For patients aged 18 years or more for whom weight and height were available (with invasive ventilation: 111; without invasive ventilation: 74; total: 185). † For patients for whom APACHE‐II data were available (with invasive ventilation: 115; without invasive ventilation: 80; total: 195). ‡ As indicated by use of angiotensin‐converting enzyme inhibitors or angiotensin receptor II blockers. |
|||||||||||||||
Box 3 – Respiratory support provided to 204 patients with COVID‐19 admitted to intensive care units (ICUs), 27 February 2020 – 30 June 2020
Box 4 – Intensive care unit (ICU) length of stay* (with 95% confidence intervals) for 204 patients with COVID‐19, 27 February 2020 – 30 June 2020, by ventilation status
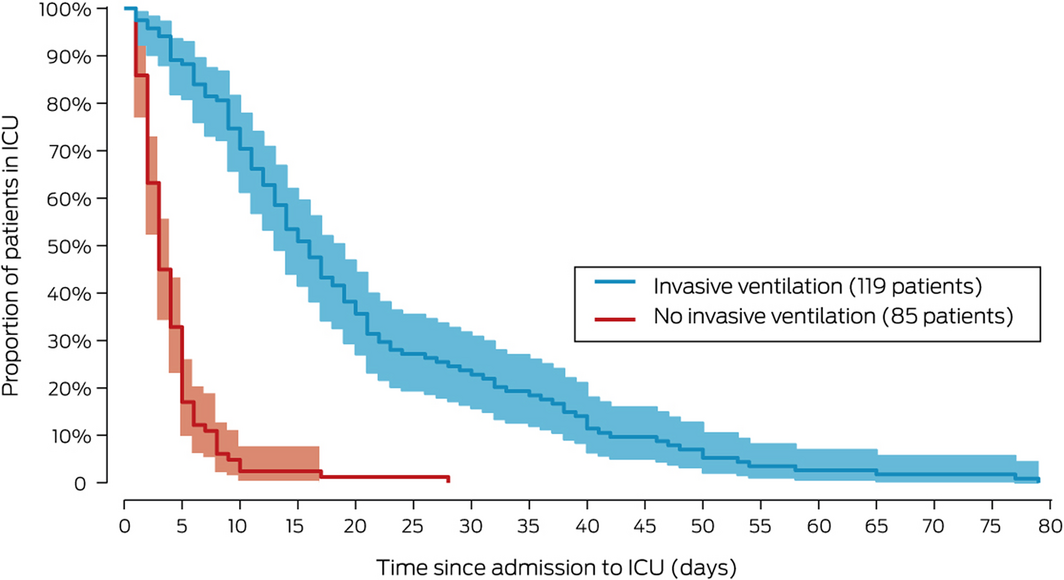
* Defined as time between ICU admission and discharge dates for all discharged patients (alive and dead); patients were censored at the date of final daily record if no ICU discharge date was recorded.
Box 5 – Outcomes (death or discharged from intensive care alive) for 200 patients (137 men, 63 women) with COVID‐19 admitted to intensive care units (ICUs), by age group and sex*
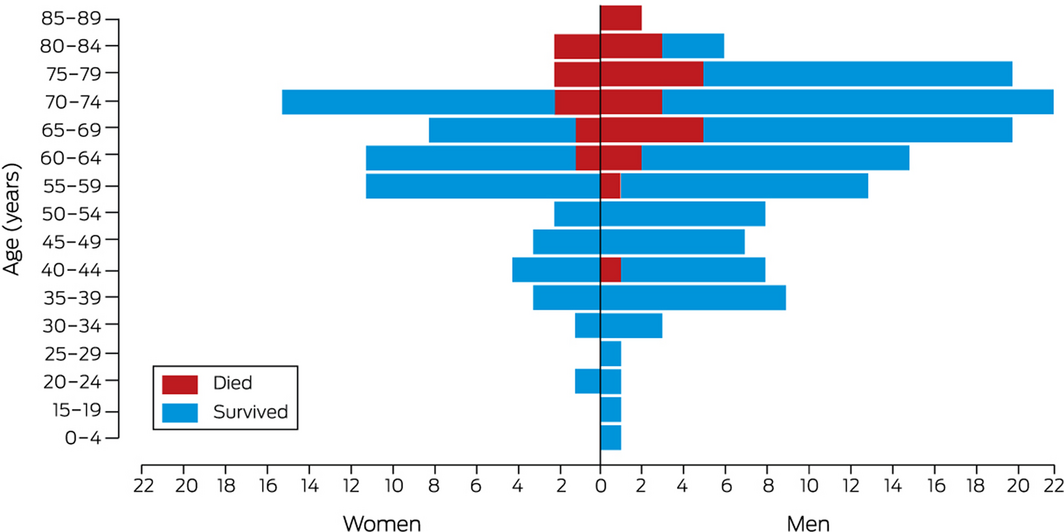
* Excludes four patients not discharged from intensive care unit by end of study period.
Box 6 – Mortality among 204 patients with COVID‐19* admitted to intensive care units (ICUs): univariate survival regression analyses
|
Covariate |
Deaths |
Hazard ratio (95% CI) |
|||||||||||||
|
|
|||||||||||||||
|
Age (years) |
|
|
|||||||||||||
|
≤ 54 |
1 (3%) |
1 |
|||||||||||||
|
55–64 |
4 (13%) |
3.0 (0.33–27) |
|||||||||||||
|
65–85 |
25 (83%) |
9.0 (1.2–67) |
|||||||||||||
|
Sex |
|
|
|||||||||||||
|
Women |
8 (27%) |
1 |
|||||||||||||
|
Men |
22 (73%) |
1.02 (0.46–2.3) |
|||||||||||||
|
Smoking status |
|
|
|||||||||||||
|
Never smoker |
22 (73%) |
1 |
|||||||||||||
|
Smoking history (any) |
6 (20%) |
1.42 (0.58–3.5) |
|||||||||||||
|
Not stated |
2 (7%) |
3.05 (0.71–13.1) |
|||||||||||||
|
Body mass index (kg/m2) |
|
|
|||||||||||||
|
Underweight (< 18.5) |
0 |
— |
|||||||||||||
|
Normal weight (18.5–24.9) |
11 (37%) |
1 |
|||||||||||||
|
Overweight (25–29.9) |
6 (20%) |
0.40 (0.15–1.07) |
|||||||||||||
|
Obese (≥ 30) |
8 (27%) |
0.32 (0.13–0.79) |
|||||||||||||
|
Not stated |
5 (17%) |
2.40 (0.82–7.03) |
|||||||||||||
|
Returned traveller |
14 (47%) |
0.69 (0.34–1.42) |
|||||||||||||
|
Cruise ship traveller |
7 (23%) |
0.76 (0.33–1.78) |
|||||||||||||
|
Health care worker |
1 (3%) |
0.44 (0.06–3.25) |
|||||||||||||
|
SOFA score on ICU day 1: respiratory, per point |
— |
1.46 (0.87–2.45) |
|||||||||||||
|
SOFA score on ICU day 1: cardiovascular, per point |
— |
1.10 (0.86–1.39) |
|||||||||||||
|
APACHE‐II score on ICU day 1, per point |
— |
1.16 (1.11–1.22) |
|||||||||||||
|
Comorbid conditions |
|
|
|||||||||||||
|
Diabetes |
12 (40%) |
1.30 (0.62–2.70) |
|||||||||||||
|
Hypertension† |
8 (27%) |
1.28 (0.57–2.88) |
|||||||||||||
|
Chronic cardiac disease |
13 (43%) |
4.11 (1.99–8.51) |
|||||||||||||
|
Asthma |
3 (10%) |
0.79 (0.24–2.60) |
|||||||||||||
|
Chronic pulmonary disease |
6 (20%) |
6.57 (2.60–16.6) |
|||||||||||||
|
Chronic immunosuppression |
4 (13%) |
2.77 (0.97–7.94) |
|||||||||||||
|
Chronic kidney disease |
5 (17%) |
3.60 (1.38–9.42) |
|||||||||||||
|
Chronic haematologic disease |
3 (10%) |
2.44 (0.74–8.04) |
|||||||||||||
|
Liver disease |
1 (3%) |
2.30 (0.31–17.1) |
|||||||||||||
|
Treatments |
|
|
|||||||||||||
|
Invasive ventilation |
26 (87%) |
1.06 (0.34–3.32) |
|||||||||||||
|
Inotropic/vasopressor support |
24 (80%) |
1.18 (0.47–2.97) |
|||||||||||||
|
Prone positioning |
16 (53%) |
1.47 (0.71–3.06) |
|||||||||||||
|
Renal replacement therapy |
8 (27%) |
1.38 (0.61–3.14) |
|||||||||||||
|
Neuromuscular blocking agents |
20 (67%) |
0.99 (0.45–2.17) |
|||||||||||||
|
Corticosteroids |
14 (47%) |
1.36 (0.66–2.81) |
|||||||||||||
|
|
|||||||||||||||
|
APACHE‐II = Acute Physiology And Chronic Health Evaluation II; CI = confidence interval; SOFA = Sequential Organ Failure Assessment. * Exceptions: SOFA: respiratory system, 147 patients; SOFA: cardiovascular system, 159 patients; APACHE‐II, 195 patients. † Indicated by use of angiotensin‐converting enzyme inhibitors or angiotensin receptor II blockers. |
|||||||||||||||
Box 7 – Mortality among 195 patients with COVID‐19* admitted to intensive care units: multivariate mixed effects survival regression analysis
|
Covariate |
Adjusted hazard ratio†* (95% CI) |
||||||||||||||
|
|
|||||||||||||||
|
Age (years) |
|
||||||||||||||
|
≤ 54 |
1 |
||||||||||||||
|
55–64 |
0.88 (0.09–8.4) |
||||||||||||||
|
65–85 |
2.8 (0.36–22) |
||||||||||||||
|
Sex |
|
||||||||||||||
|
Women |
1 |
||||||||||||||
|
Men |
0.85 (0.35–2.04) |
||||||||||||||
|
APACHE‐II score on ICU day 1, per point |
1.15 (1.09–1.21) |
||||||||||||||
|
Comorbid conditions |
|
||||||||||||||
|
Chronic cardiac disease |
3.38 (1.46–7.83) |
||||||||||||||
|
Treatments |
|
||||||||||||||
|
Invasive ventilation |
0.42 (0.11–1.58) |
||||||||||||||
|
|
|||||||||||||||
|
APACHE‐II = Acute Physiology And Chronic Health Evaluation II; CI = confidence interval; SOFA = Sequential Organ Failure Assessment. * APACHE‐II data were not available for nine patients. † Adjusted for all other variables in model. |
|||||||||||||||
Received 27 June 2020, accepted 15 September 2020
- Aidan JC Burrell1,2
- Breanna Pellegrini3
- Farhad Salimi3
- Husna Begum1,2
- Tessa Broadley1,2
- Lewis T Campbell4,5
- Allen C Cheng2,3
- Winston Cheung6
- D James Cooper1,2
- Arul Earnest3
- Simon J Erickson7
- Craig J French1,8
- John M Kaldor9
- Edward Litton10,11
- Srinivas Murthy12
- Richard E McAllister13
- Alistair D Nichol1,2
- Annamaria Palermo10,11
- Mark P Plummer14
- Mahesh Ramanan15
- Benjamin AJ Reddi16,17
- Claire Reynolds18
- Tony Trapani1,2
- Steve A Webb1,11
- Andrew A Udy1,2
- 1 Australian and New Zealand Intensive Care Research Centre, Monash University, Melbourne, VIC
- 2 The Alfred Hospital, Melbourne, VIC
- 3 School of Public Health and Preventive Medicine, Monash University, Melbourne, VIC
- 4 Royal Darwin Hospital, Darwin, NT
- 5 Menzies School of Health Research, Darwin, NT
- 6 Concord Repatriation General Hospital, Sydney, NSW
- 7 Perth Children’s Hospital, Perth, WA
- 8 Western Health, Melbourne, VIC
- 9 The Kirby Institute, University of New South Wales, Sydney, NSW
- 10 Fiona Stanley Hospital, Perth, WA
- 11 St John of God Health Care, Perth, WA
- 12 The University of British Columbia, Vancouver, BC, Canada
- 13 Royal Hobart Hospital, Hobart, TAS
- 14 Royal Melbourne Hospital, Melbourne, VIC
- 15 Caboolture Hospital, Caboolture, QLD
- 16 Royal Adelaide Hospital, Adelaide, SA
- 17 The University of Adelaide, Adelaide, SA
- 18 St Vincent’s Hospital Sydney, Sydney, NSW
The Australian Department of Health and the Australian Partnership for Preparedness Research on Infectious Disease Emergencies (APPRISE) provided support for data collection and data analysis, and research staff.
No relevant disclosures.
- 1. Bhatraju PK, Ghassemieh BJ, Nichols M, et al. Covid‐19 in critically ill patients in the Seattle region: case series. N Engl J Med 2020; 382: 2012–2022.
- 2. Grasselli G, Zangrillo A, Zanella A, et al. Baseline characteristics and outcomes of 1591 patients infected with SARS‐CoV-2 admitted to ICUs of the Lombardy region, Italy. JAMA 2020; 323: 1574–1581.
- 3. Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS‐CoV-2 pneumonia in Wuhan, China: a single‐centered, retrospective, observational study. Lancet Respir Med 2020; 8: 475–481.
- 4. RECOVERY Collaborative Group. Dexamethasone in hospitalized patients with Covid‐19: preliminary report. N Engl J Med 2020; https://doi.org/10.1056/NEJMoa2021436 [online ahead of print].
- 5. ANZIC Influenza Investigators; Webb SAR, Pettilä V, Seppelt I, et al. Critical care services and 2009 H1N1 influenza in Australia and New Zealand. N Engl J Med 2009; 361: 1925‐1934.
- 6. Rice TW, Rubinson L, Uyeki TM, et al; NHLBI ARDS Network. Critical illness from 2009 pandemic influenza A virus and bacterial coinfection in the United States. Crit Care Med 2012; 40: 1487–1498.
- 7. Grasselli G, Pesenti A, Cecconi M. Critical care utilization for the COVID‐19 outbreak in Lombardy, Italy: early experience and forecast during an emergency response. JAMA 2020; 323: 1545–1546.
- 8. Murthy S, Gomersall CD, Fowler RA. Care for critically ill patients with COVID‐19. JAMA 2020; 323: 1499–1500.
- 9. Xie J, Tong Z, Guan X, et al. Critical care crisis and some recommendations during the COVID‐19 epidemic in China. Intensive Care Med 2020; 46: 837–840.
- 10. Docherty AB, Harrison EM, Green CA, et al; ISARIC4C investigators. Features of 20 133 UK patients in hospital with covid‐19 using the ISARIC WHO Clinical Characterisation Protocol: prospective observational cohort study. BMJ 2020; 369: m1985.
- 11. Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID‐19 in the New York City area. JAMA 2020; 323: 2052–2059.
- 12. Caly L, Druce J, Roberts J, et al. Isolation and rapid sharing of the 2019 novel coronavirus (SARS‐CoV-2) from the first patient diagnosed with COVID‐19 in Australia. Med J Aust 2020; 212: 459–462. https://www.mja.com.au/journal/2020/212/10/isolation-and-rapid-sharing-2019-novel-coronavirus-sars-cov-2-first-patient
- 13. COVID‐19 National Incident Room Surveillance Team. COVID‐19, Australia: epidemiology report 20 (fortnightly reporting period ending 5 July 2020). https://www1.health.gov.au/internet/main/publishing.nsf/Content/1D03BCB527F40C8BCA258503000302EB/$File/covid_19_australia_epidemiology_report_20_fortnightly_reporting_period_ending_5_july_2020.pdf (viewed Sept 2020).
- 14. International Severe Acute Respiratory and Emerging Infections Consortium. COVID‐19 report: 19 May 2020. https://media.tghn.org/medialibrary/2020/05/ISARIC_Data_Platform_COVID-19_Report_19MAY20.pdf (viewed Aug 2020).
- 15. Warrillow S, Austin D, Cheung W, et al. ANZICS guiding principles for complex decision making during the COVID‐19 pandemic. Crit Care Resusc 2020; 22: 98–102.
- 16. Australian and New Zealand Intensive Care Society (ANZICS) COVID‐19 guidelines, version 2. Apr 2020. https://www.anzics.com.au/wp-content/uploads/2020/04/ANZI_3367_Guidelines_V2.pdf (viewed Aug 2020).
- 17. Knaus WA, Draper EA, Wagner DP, Zimmerman JE. APACHE II: a severity of disease classification system. Crit Care Med 1985; 13: 818–829.
- 18. Vincent JL, Moreno R, Takala J, et al. The SOFA (Sepsis‐related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis‐Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 1996; 22: 707–710.
- 19. Australian and New Zealand Intensive Care Society. ANZICS Centre for Outcome and Resource Evaluation. 2018 report. https://www.anzics.com.au/wp-content/uploads/2019/10/2018-ANZICS-CORE-Report.pdf (viewed Aug 2020).
- 20. Therneau TM, Grambsch PM. Modeling survival data: extending the Cox model. New York (NY): Springer, 2000.
- 21. Guan WJ, Ni ZY, Hu Y, et al; China Medical Treatment Expert Group for Covid‐19. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020; 382: 1708–1720.
- 22. Peng Y, Xu B, Sun B, et al. Importance of timely management of patients in reducing fatality rate of coronavirus disease 2019. J Infect Public Health 2020; 13: 890–892.
- 23. Litton E, Bucci T, Chavan S, et al. Surge capacity of intensive care units in case of acute increase in demand caused by COVID‐19 in Australia. Med J Aust 2020; 212: 463–467. https://www.mja.com.au/journal/2020/212/10/surge-capacity-intensive-care-units-case-acute-increase-demand-caused-covid-19
- 24. Ji Y, Ma Z, Peppelenbosch MP, Pan Q. Potential association between COVID‐19 mortality and health‐care resource availability. Lancet Glob Health 2020; 8: e480.
- 25. Mitra AR, Fergusson NA, Lloyd‐Smith E, et al. Baseline characteristics and outcomes of patients with COVID‐19 admitted to intensive care units in Vancouver, Canada: a case series. CMAJ 2020; 192: E694–E701.





Abstract
Objectives: To describe the characteristics and outcomes of patients with COVID‐19 admitted to intensive care units (ICUs) during the initial months of the pandemic in Australia.
Design, setting: Prospective, observational cohort study in 77 ICUs across Australia.
Participants: Patients admitted to participating ICUs with laboratory‐confirmed COVID‐19 during 27 February – 30 June 2020.
Main outcome measures: ICU mortality and resource use (ICU length of stay, peak bed occupancy).
Results: The median age of the 204 patients with COVID‐19 admitted to intensive care was 63.5 years (IQR, 53–72 years); 140 were men (69%). The most frequent comorbid conditions were obesity (40% of patients), diabetes (28%), hypertension treated with angiotensin‐converting enzyme inhibitors or angiotensin II receptor blockers (24%), and chronic cardiac disease (20%); 73 patients (36%) reported no comorbidity. The most frequent source of infection was overseas travel (114 patients, 56%). Median peak ICU bed occupancy was 14% (IQR, 9–16%). Invasive ventilation was provided for 119 patients (58%). Median length of ICU stay was greater for invasively ventilated patients than for non‐ventilated patients (16 days; IQR, 9–28 days v 3 days; IQR, 2–5 days), as was ICU mortality (26 deaths, 22%; 95% CI, 15–31% v four deaths, 5%; 95% CI, 1–12%). Higher Acute Physiology and Chronic Health Evaluation II (APACHE‐II) scores on ICU day 1 (adjusted hazard ratio [aHR], 1.15; 95% CI, 1.09–1.21) and chronic cardiac disease (aHR, 3.38; 95% CI, 1.46–7.83) were each associated with higher ICU mortality.
Conclusion: Until the end of June 2020, mortality among patients with COVID‐19 who required invasive ventilation in Australian ICUs was lower and their ICU stay longer than reported overseas. Our findings highlight the importance of ensuring adequate local ICU capacity, particularly as the pandemic has not yet ended.