The novel influenza virus, pandemic (H1N1) 2009 influenza (or “swine flu”) has spread rapidly since it was first reported in California on 15 April 2009, and in a returned traveller in Australia on 9 May 2009.1 During the early phase of the pandemic, a particularly large number of infections were confirmed in Victoria, making it the location with the greatest density of infections worldwide.2
Seasonal influenza is associated with considerable morbidity and mortality, accounting for about 2100 hospitalisations in Australia annually.3 Early reports pertaining to pandemic (H1N1) 2009 influenza suggested high mortality in younger, previously well patients;4,5 however, subsequent reports have noted a lower mortality. Among the first 553 confirmed cases in California, 30 affected patients had been admitted to hospital, and none had died.6 Other reports found that around half of patients admitted to hospital had traditional risk factors for severe seasonal influenza infection, and that of 22 hospitalised patients with confirmed pandemic (H1N1) 2009 influenza infections, two had died.7,8
All eight public (non-paediatric) hospitals in Melbourne that had infectious diseases physicians were contacted and invited to participate. Patients admitted to participating hospitals between 1 May and 17 July 2009 were eligible for inclusion if they had laboratory-confirmed infection with pandemic (H1N1) 2009 influenza. Admission data were collected using a standardised case report form. The case report form included demographic information, information about comorbid conditions and potential sources of infection, and clinical information about symptoms, treatment and patients’ progress in hospital. Length of stay data were collected for total time in hospital as well as time spent in intensive care units (ICUs). Data for six patients in this series have been reported in an earlier brief communication.9
Testing for pandemic (H1N1) 2009 influenza was performed at the Victorian Infectious Diseases Reference Laboratory, a state-wide service. Testing for influenza A viruses involved RNA extraction from clinical material, followed by reverse transcription. Complementary DNA was amplified using a 7500 Fast Real-Time PCR [polymerase chain reaction] System (Applied Biosystems, Melbourne, Vic), incorporating primers and probe that targeted the matrix gene of influenza type A viruses, including the novel (swine lineage) H1N1 virus. Samples that tested positive in this assay were confirmed as positive or negative for the swine influenza virus in a second real-time PCR assay incorporating primers and probe specific for the haemagglutinin gene of that virus.10
Demographic characteristics and the comorbid conditions of the first 112 patients hospitalised with pandemic (H1N1) 2009 influenza are shown in Box 1.
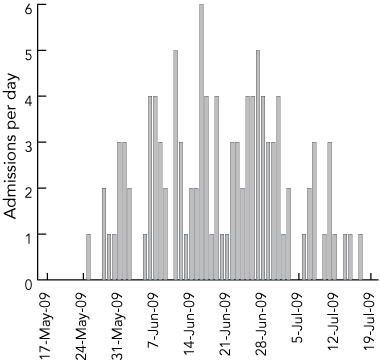
Box 2 shows the daily admissions of patients with confirmed pandemic (H1N1) 2009 influenza infection at the participating hospitals. An apparent peak of 4–6 admissions per day was noted between 12 and 28 June (peak, 16 June), with a decline in admissions thereafter.
Clinical and radiological features at the time of initial presentation are shown in Box 3. Box 4 shows the spectrum of illness observed in patients admitted to participating hospitals with pandemic (H1N1) 2009 influenza; these included some unusual complications, such as febrile seizures (three patients), rhabdomyolysis (one), myocarditis (one) and Guillain–Barré syndrome (one). Other non-respiratory presentations included preterm labour, syncope, lethargy, abdominal pain and diarrhoea.
There were 30 patients with multifocal changes on initial chest x-ray (Box 3), and these patients appeared to have worse in-hospital outcomes. When compared with patients who had normal findings on initial chest x-ray, those with multifocal changes were more likely to require ICU admission (odds ratio, 5.2; 95% CI, 1.9–13.9). Mean hospital length of stay in this group was 10.7 days versus 5.1 days in patients with unifocal or no changes on initial chest x-ray (P < 0.001). Finally, each of the three patients who died had multifocal changes on initial chest x-ray.
Although it is difficult to compare these findings directly with those expected in the case of seasonal influenza,11 of the 112 patients in our series, 30 required admission to an intensive care or high dependency unit, and three died during their acute illness; this severity of illness considerably exceeds that which would be expected during a regular influenza season. Our findings highlight the significant illness and burden of disease that may be associated with pandemic (H1N1) 2009 influenza infections. When taken together with the additional resources required for infection control and isolation practices, our findings indicate that pandemic (H1N1) 2009 influenza has the potential to provide a significant additional burden on hospitals in affected areas.
1 Demographic characteristics and comorbid conditions of the first 112 patients admitted to participating hospitals with pandemic (H1N1) 2009 influenza
2 Number of daily admissions to participating hospitals of patients with confirmed pandemic (H1N1) 2009 influenza, 1 May to 31 July 2009
3 Symptoms and radiological findings in the first 112 patients admitted to participating hospitals with pandemic (H1N1) 2009 influenza
4 Spectrum of illness observed in the first 112 patients admitted to participating hospitals with pandemic (H1N1) 2009 influenza
Classical “influenza-like illness” with fever, cough and dyspnoea
Secondary bacterial pneumonia following influenza-like illness
Afebrile exacerbation of chronic obstructive pulmonary disease
Premature labour due to influenza
Increased frequency of seizures in a patient with known epilepsy
Rhabdomyolysis
Guillain–Barré syndrome
Myocarditis
- Justin T Denholm1
- Claire L Gordon2
- Paul D Johnson2
- Saliya S Hewagama3
- Rhonda L Stuart3
- Craig Aboltins4
- Cameron Jeremiah4
- James Knox5
- Garry P Lane5
- Adrian R Tramontana6
- Monica A Slavin6
- Thomas R Schulz1
- Michael Richards1
- Chris J Birch7
- Allen C Cheng8,9
- 1 Victorian Infectious Diseases Service, Royal Melbourne Hospital, Melbourne, VIC.
- 2 Infectious Diseases Department, Austin Health, Melbourne, VIC.
- 3 Southern Health, Melbourne, VIC.
- 4 Northern Hospital, Melbourne, VIC.
- 5 Western Health, Melbourne, VIC.
- 6 Peter MacCallum Cancer Centre, Melbourne, VIC.
- 7 Victorian Infectious Diseases Reference Laboratory, Melbourne, VIC.
- 8 Alfred Hospital, Melbourne, VIC.
- 9 Monash University, Melbourne, VIC.
None identified.
- 1. Centers for Disease Control and Prevention (US). Swine influenza (H1N1) infection in two children — Southern California, March–April 2009. MMWR Morb Mortal Wkly Rep 2009; 58: 400-402.
- 2. State Government of Victoria, Australia, Department of Health. H1N1 influenza 09 (human swine flu). http://humanswineflu.health.vic.gov.au/ (accessed Jul 2009).
- 3. Brotherton J, Wang H, Schaffer A. Vaccine preventable diseases and vaccination coverage in Australia 2003 to 2005. Commun Dis Intell 2007; 31 (2 Suppl): 1-50.
- 4. Schanzer DL, Langley JM, Tam TW. Co-morbidities associated with influenza-attributed mortality, 1994–2000, Canada. Vaccine 2008; 26: 4697-4703.
- 5. Centers for Disease Control and Prevention (US). Outbreak of swine-origin influenza A (H1N1) virus infection — Mexico, March–April 2009. MMWR Morb Mortal Wkly Rep 2009; 58: 467-470.
- 6. Centers for Disease Control and Prevention (US). Hospitalized patients with novel influenza A (H1N1) virus infection — California, April–May 2009. MMWR Morb Mortal Wkly Rep 2009; 58: 536-546.
- 7. Perez-Padilla R, de la Rosa-Zamboni D, Ponce de Leon S, et al. Pneumonia and respiratory failure from swine-origin influenza A (H1N1) in Mexico. N Engl J Med 2009; 361 : 680-689. Epub 2009 Jun 29.
- 8. Novel Swine-Origin Influenza A (H1N1) Virus Investigation Team. Emergence of a novel swine-origin influenza A (H1N1) virus in humans. N Engl J Med 2009; 360: 2605-2615.
- 9. Kaufman MA, Duke GJ, McGain F, et al. Life-threatening respiratory failure from H1N1 influenza 09 (human swine influenza). Med J Aust, 2009; 191: 154-156. <MJA full text>
- 10. World Health Organization. CDC protocol of realtime RTPCR for swine flu influenza A(H1N1). 28 April 2009, revision 1 (30 April 2009). http://www.who.int/csr/resources/publications/swineflu/CDCreal timeRTPCRprotocol_20090428.pdf (accessed Oct 2009).
- 11. Kelly H, Grant KA, Williams S, et al. Epidemiological characteristics of pandemic influenza H1N1 2009 and seasonal influenza infection. Med J Aust 2009; 191: 146-149. <MJA full text>





Abstract
Objective: To describe the case characteristics and outcomes of patients hospitalised with pandemic (H1N1) 2009 influenza infection during the first 2 months of the epidemic.
Design, participants and setting: Prospective case series of 112 patients admitted to seven hospitals in Melbourne with laboratory-confirmed pandemic (H1N1) 2009 influenza between 1 May and 17 July 2009.
Main outcome measures: Details of case characteristics, risk factors for severe disease, treatment and clinical course.
Results: Of 112 hospitalised patients, most presented with cough (88%) and/or fever (82%), but several (4%) had neither symptom. A quarter of female patients (15) were pregnant or in the post-partum period. Patients presenting with multifocal changes on chest x-ray had significantly longer hospital lengths of stay, and were more likely to require intensive care unit admission. Thirty patients required admission to an intensive care unit, and three died during their acute illness. The median length of intensive care admission was 10.5 days (interquartile range, 5–16 days).
Conclusions: This study highlights risk factors for severe disease, particularly pregnancy. Clinical and public health planning for upcoming influenza seasons should take into account the spectrum and severity of clinical infection demonstrated in this report, and the need to concentrate resources effectively in high-risk patient groups.