In 2001, the University of Western Australia’s Centre for Health Services Research published a report entitled Duty to care: preventable physical illness in people with mental illness, which highlighted some alarming statistics on the physical health of people with mental illness.1 This population-based record linkage study, funded by the National Health and Medical Research Council, had linked records for all users of mental health services in WA with hospital admission records, cancer registrations and death records. The aim of the study was to examine the physical health of people with mental illness. Results showed that this patient group had higher rates of physical illness, higher death rates from major diseases, and physical illnesses that were less likely to be diagnosed. These patients were also less likely to receive appropriate treatment than people in the general population. The report noted the need to provide primary health care to people with mental illness and the need for psychiatrists and mental health professionals to have a wider view of the overall health of patients. It also highlighted the potential for shared care or partnership agreements between general practitioners and mental health clinics.
In September 2002, the WA Office of Mental Health established the HealthRight Advisory Group to respond to the Duty to care report. The Advisory Group’s report, Who is your GP?,2 published in 2004, included eight key recommendations (Box 1). In 2005, the HealthRight Project began to implement these recommendations. One of several tasks identified was to develop clinical guidelines and protocols to help clinicians improve the physical care of people with mental illness. Within this context, designing an evidence-based algorithm to help clinicians screen psychiatric patients at risk of developing the metabolic syndrome (MetS) was identified as a priority. This algorithm, with its accompanying monitoring form, is presented here.
MetS is a well described cluster of interrelated risk factors for developing cardiovascular disease and type 2 diabetes. The condition affects an estimated 19%–29% of Australian adults aged over 25 years.3 The core components of MetS are central obesity, hypertension, hyperglycaemia, and dyslipidaemia. People with MetS are two to three times more likely to have a heart attack or stroke and five times more likely to develop type 2 diabetes than those without.4,5 It is, therefore, a syndrome of considerable public health significance.
A number of international health groups, including the World Health Organization and the American Heart Association, have developed criteria for defining MetS.6,7 However, differences between these definitions have caused some confusion for both clinicians and researchers and have hindered attempts to compare the burden of MetS in different populations. In 2005, the International Diabetes Federation (IDF) published a consensus definition of MetS which it considered to be easy to use in clinical practice, to provide a simple diagnostic and clinical tool, and to ease comparison of data from different studies.3,8 The IDF definition (Box 2), which includes ethnicity-specific cut-off points for central obesity, is particularly relevant to Australia’s multicultural population.
The relationship between psychotic illness and metabolic dysregulation is a complex one. There is evidence that people with schizophrenia and bipolar disorder are at increased risk of hyperglycaemia and diabetes. The reasons for this include the increased likelihood of a family history of diabetes, poor diet, physical inactivity, and obesity.9,10 In addition to these important risk factors, studies have shown that patients receiving antipsychotic medication are at higher risk of developing MetS than people in the general population, with prevalence estimates of MetS in patients with a psychiatric diagnosis ranging from 24% to 53%.11,12 These rates vary depending on the criteria used to define MetS, the patient groups sampled (eg, all patients or only those with selected diagnoses; inpatients or outpatients), and whether all antipsychotic medications or only second-generation antipsychotics are included. These studies have come from the United States and Europe, with no published Australian data currently available.
With regard to the components of MetS, the prevalence of diabetes has been of particular concern to clinicians and researchers. The prevalence of diabetes in patients with psychoses increased with the introduction of first-generation antipsychotics and has increased even further (to 19%) with the introduction of second-generation agents.13 Agents that cause the most weight gain may pose more of a risk in this respect. Current evidence suggests that clozapine and olanzapine are associated with greater weight gain than other antipsychotic agents, as well as increased risk of diabetes and of lipid dysregulation. However, individual patient responses to these drugs are variable in terms of antipsychotic effect and metabolic change.
The relative risks associated with individual antipsychotic drugs in relation to components of MetS remain controversial and need to be clarified by further prospective studies that specifically address this issue.9,10,12,13 The current consensus from leading psychiatrists and endocrinologists is that patients with psychosis are at increased risk of metabolic dysregulation resulting from a number of factors, including the medication used to treat their psychosis.
Australia, the US, the United Kingdom, Belgium, and Canada have all published guidelines for metabolic monitoring of patients taking antipsychotic medication. However, most focus on diabetes rather than MetS.13,14 Guidelines also exist for specific antipsychotic drugs such as clozapine and aripiprazole.15,16 In a review of the major published consensus guidelines for metabolic monitoring, Cohn and Sernyak14 concluded that these guidelines all have areas of similarity but lack consensus regarding precisely who, what and when to monitor and in identifying the cut-off points between “normal” and “abnormal”.
In the wake of these guidelines, surveys in the US and the UK have evaluated the extent of metabolic monitoring and clinicians’ awareness of and attitudes to MetS.17,18 The surveys indicate that clinicians are generally aware of the implications of MetS and the need for careful consideration of the risk of MetS when choosing an antipsychotic to prescribe. No such survey has yet been published from the Australian perspective.
In a busy psychiatric clinic, time pressures, equipment availability and patient cooperation are all possible constraints to monitoring. Added to this, the uncertainty of deciding who to monitor (all patients with serious mental illness or only those taking second-generation antipsychotics) increases the chance that monitoring will not occur. Within the clinical setting, an increase in weight, specifically central obesity, is the most noticeable sign of possible MetS. Thus the quickest and easiest way to identify patients at risk is to measure waist circumference. However, studies have found that waist circumference is rarely measured and that overall monitoring for adverse metabolic effects of antipsychotic medication (eg, hypertension and hyperglycaemia) is poor.17-19
Guidelines have the potential to “improve the care received by patients by promoting interventions of proven benefit” and to “inform clinical practice, promoting effective and cost effective health care”,20 yet current guidelines for metabolic screening in this patient population seem to have had little impact on clinical practice or patient outcomes. Although clinicians may be aware of published guidelines and may receive personal reminders by mail to undertake monitoring, it still does not occur consistently in either inpatient or outpatient settings.21,22 It is worth noting, however, that the mandating of haematological and cardiac monitoring of patients prescribed clozapine has resulted in improved rates of metabolic monitoring of these patients.21,23
Opinion is divided on whose responsibility it is to undertake monitoring. Some authors suggest that, as psychiatrists prescribe the medication and patients with severe mental illness are seen more frequently in mental health settings than in primary care (and are less likely to have a GP), it is important for mental health clinicians to ensure regular screening is carried out.13,14,24 Others take the view that there needs to be a coordinated approach between primary and secondary care.23 However, there is general agreement that patients and carers should be encouraged to self-monitor.14,24
The Who is your GP? report2 included the following specific recommendation within the general recommendation on routine clinical care (Box 1, item 2):
All mental health services should develop and implement clinical protocols relating to the physical health of mental health consumers . . . [P]hysical assessment will be incorporated into all mental health service patient records and documentation including inpatient notes and discharge management plans as well as relevant outpatient documents.
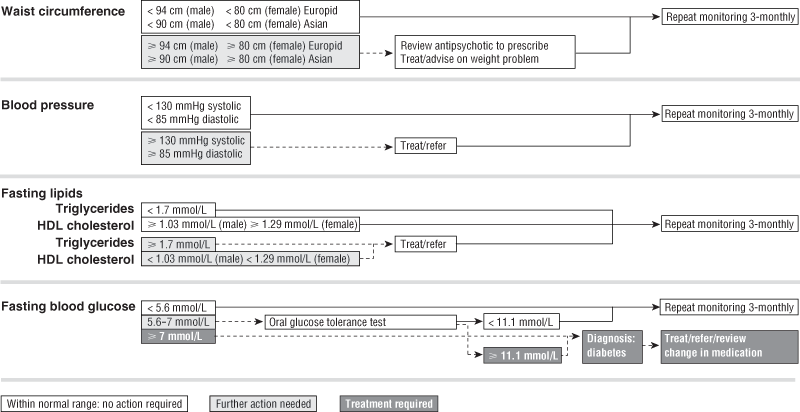
As part of our response to this recommendation we have designed a simple clinical algorithm for metabolic screening (Box 3) and a metabolic monitoring form, adapted from the clinical monitoring system for clozapine.15 Our aim was to produce a simple, all-encompassing algorithm that can be used by all clinicians, whether in a primary care setting or a psychiatric clinic, and a single monitoring form that facilitates tracking changes over time. We reviewed existing guidelines and their recommendations on what and when to monitor as well as which test and cut-off points to use. The algorithm and form we developed are consistent with current evidence in this field and are based on the IDF definition of MetS3 outlined in Box 2. We believe that screening all patients taking antipsychotics using our algorithm and monitoring form is a simpler option than using the more complex and detailed guidelines previously published.13-16
We advocate screening of patients before they commence antipsychotic treatment and, at the minimum, 3-monthly monitoring of all patients on any antipsychotic medication (regardless of the diagnosis or specific antipsychotic drug). This would help reduce confusion as to who should be monitored and would heighten awareness of the risk of MetS in patients taking conventional as well as atypical antipsychotics. Monitoring from baseline is of particular importance to identify high-risk individuals and to ensure early detection of changes in MetS parameters. The use of the monitoring form, as with the clozapine monitoring form on which it was based, enables a comprehensive sequential record of all testing and ensures the ability to track results over time.15
1 Summary of recommendations from the Who is your GP? report*
1. Raise awareness of the physical health needs of mental health patients
5. Enhance the voice of patients, their families and carers
7. Develop targeted health promotion and illness prevention strategies
8. Implement further research and ongoing monitoring and evaluation of new services and programs to improve the physical health of people with mental illness in response to the Duty to care report1
* Adapted from Who is your GP? Final report of the HealthRight Advisory Group.2
2 International Diabetes Federation (IDF) consensus definition of the metabolic syndrome*
3 Clinical algorithm for monitoring the metabolic syndrome in people treated with antipsychotic medication
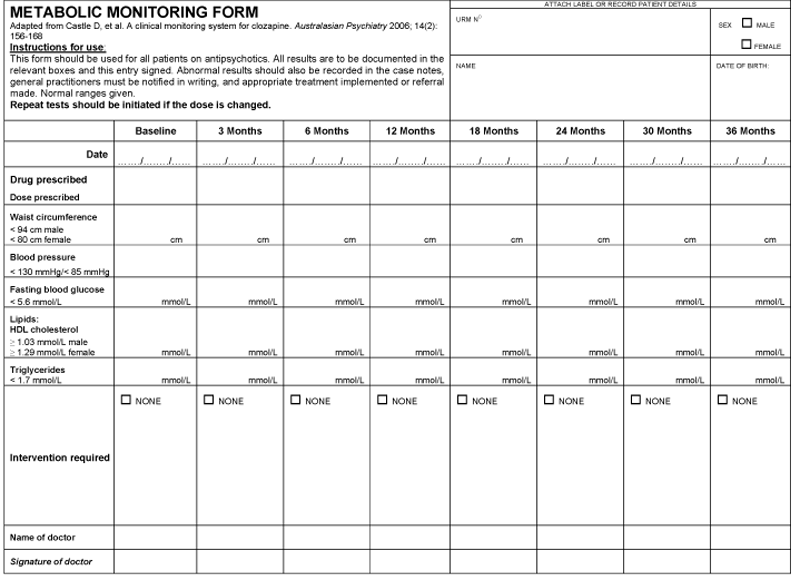 | |||||||||||||||
| For a clearer version of this form see the pdf version of this article. | |||||||||||||||
- Anna J Waterreus1
- Jonathan D E Laugharne2
- 1 School of Psychiatry and Clinical Neurosciences, University of Western Australia, Perth, WA.
- 2 School of Psychiatry and Clinical Neurosciences, University of Western Australia, Fremantle, WA.
None identified.
- 1. Lawrence D, Holman CD, Jablensky AV. Duty to care: preventable physical illness in people with mental illness. Perth: University of Western Australia, 2001. http://www.sph.uwa.edu.au/go/research-programs/centre-for-health-services-research/consumer-information/schools-and-centres/schools/school-of-population-health/duty-to-care-report (accessed Jan 2009).
- 2. Who is your GP? Final report of the HealthRight Advisory Group. Perth: Western Australian Department of Health, 2004. http://www.mental.health.wa.gov.au/one/index.asp (accessed Jan 2009).
- 3. Zimmet PZ, Alberti KG, Shaw JE. Mainstreaming the metabolic syndrome: a definitive definition. Med J Aust 2005; 183: 175-176. <MJA full text>
- 4. Stern MP, Williams K, Gonzalez-Villalpando C, et al. Does the metabolic syndrome improve identification of individuals at risk of type 2 diabetes and/or cardiovascular disease? Diabetes Care 2004; 27: 2676-2681.
- 5. Dekker JM, Girman C, Rhodes T, et al. Metabolic syndrome and 10-year cardiovascular disease risk in the Hoorn study. Circulation 2005; 112: 666-673.
- 6. Alberti KG, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabet Med 1998; 15: 539-553.
- 7. Grundy SM, Cleeman JI, Daniels SR, et al. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung and Blood Institute scientific statement. Circulation 2005; 112: 2735-2752.
- 8. International Diabetes Federation. The IDF consensus worldwide definition of the metabolic syndrome. Brussels: IDF, 2006. http://www.idf.org/webdata/docs/IDF_Meta_def_final.pdf (accessed Jan 2009).
- 9. Holt RI, Peveler CP. Association between antipsychotic drugs and diabetes. Diabetes Obes Metab 2006; 8: 125-135.
- 10. McIntyre RS, Leiter L, Yale J-F, et al. Schizophrenia, glycemia and antipsychotic medications: an expert consensus review. Can J Diabetes 2005; 29: 113-121.
- 11. Bobes J, Arango C, Aranda P, et al; CLAMORS Study Collaborative Group. Cardiovascular and metabolic risk in outpatients with schizophrenia treated with antipsychotics: results of the CLAMORS Study. Schizophr Res 2007; 90: 162-173.
- 12. De Hert MA, van Winkel R, Van Eyck D, et al. Prevalence of the metabolic syndrome in patients with schizophrenia treated with antipsychotic medication. Schizophr Res 2006; 83: 87-93.
- 13. Lambert TJ, Chapman LH; Consensus Working Group. Diabetes, psychotic disorders and antipsychotic therapy: a consensus statement. Med J Aust 2004; 181: 544-548. <MJA full text>
- 14. Cohn TA, Sernyak MJ. Metabolic monitoring for patients treated with antipsychotic medications. Can J Psychiatry 2006; 51: 492-501.
- 15. Castle D, Lambert T, Melbourne S, et al. A clinical monitoring system for clozapine. Australas Psychiatry 2006; 14: 156-168.
- 16. Travis MJ, Burns T, Dursun S, et al. Aripiprazole in schizophrenia: consensus guidelines. Int J Clin Pract 2005; 59: 485-495.
- 17. Newcomer JW, Nasrallah HA, Loebel AD. The atypical antipsychotic therapy and metabolic issues national survey: practice patterns and knowledge of psychiatrists. J Clin Psychopharmacol 2004; 24 (5 Suppl 1): S1-S6.
- 18. Barnes TR, Paton C, Cavanagh MR, et al; UK Prescribing Observatory for Mental Health. A UK audit of screening for the metabolic side effects of antipsychotics in community patients. Schizophr Bull 2007; 33: 1397-1403.
- 19. Mackin P, Bishop DR, Watkinson HM. A prospective study of monitoring practices for metabolic disease in antipsychotic-treated community psychiatric patients. BMC Psychiatry 2007; 7: 28.
- 20. Grimshaw JM, Thomas RE, MacLennan G, et al. Effectiveness and efficiency of guideline dissemination and implementation strategies. Health Technol Assess 2004; 8(6): iii-iv.
- 21. Taylor D, Young C, Esop R, et al. Testing for diabetes in hospitalized patients prescribed antipsychotic drugs. Br J Psychiatry 2004; 185: 152-156.
- 22. Runcie O, Boilson M, Hamilton R. Monitoring weight and blood glucose in in-patients: how helpful is a protocol? Psychiatr Bull R Coll Psychiatr 2007; 31: 88-91.
- 23. Tarrant C. Blood glucose testing for adults prescribed atypical antipsychotics in primary and secondary care. Psychiatr Bull R Coll Psychiatr 2006; 30: 286-288.
- 24. Marder SR, Essock AM, Miller AL, et al. Physical health monitoring of patients with schizophrenia. Am J Psychiatry 2004; 161: 1334-1349.





Abstract
The metabolic syndrome (MetS) is a well described cluster of interrelated risk factors for developing cardiovascular disease and type 2 diabetes. The key components of MetS are central obesity, hypertension, hyperglycaemia and dyslipidaemia.
The 2005 International Diabetes Federation (IDF) consensus definition of MetS aimed to reduce confusion over criteria for MetS and to provide a simple diagnostic and clinical tool.
There is considerable evidence to show that patients prescribed antipsychotic drugs are at increased risk of developing MetS.
Existing clinical guidelines for metabolic screening of patients taking antipsychotics focus on diabetes rather than on the broader syndrome of MetS and are not consistent with the IDF definition of MetS.
Monitoring for MetS in patients taking antipsychotics (both inpatients and outpatients) is generally poor.
We present a user-friendly clinical algorithm and monitoring form, based on current evidence and using the IDF definition of MetS, to help clinicians in primary care or specialist settings to effectively monitor for MetS in these patients.