The known: Aggressive reduction of lipid levels following acute coronary syndrome (ACS) reduces the risk of recurrence. In Australia, the proportion of people who attain target lipid levels within 12 months of ACS is unknown.
The new: 45% of people admitted to hospital with ACS did not achieve recommended blood lipid levels within 12 months of discharge. Women, people under 65, and those using lipid‐lowering therapy or with elevated cholesterol levels before admission were particularly unlikely to reach targets, as were those not prescribed intensive lipid‐lowering therapy or at least four evidence‐based medications on discharge.
The implications: Optimising lipid control after ACS could include using more intensive therapy and using combinations of medications, including novel agents.
In Australia, more than 60 000 hospitalisations each year and more than 7000 hospital deaths are of people with acute coronary syndrome (ACS).1 The risk of recurrence and adverse events is very high for people who experience ACS,2 and secondary preventive strategies are consequently important. Intensive lipid‐lowering therapy reduces the risk of ACS recurrence and the associated morbidity and mortality.2,3,4
Elevated low‐density lipoprotein cholesterol (LDL‐C) levels are a major risk factor for acute and recurrent ACS: for each 1 mmol/L reduction in LDL‐C level, all‐cause mortality declines by 10% and the number of recurrent myocardial infarction events is reduced by 13%.3 HMG‐Co‐A reductase inhibitors (statins) reduce the incidence of adverse cardiovascular events during the six months after ACS by 25–40%.4,5 High intensity therapy is more effective than standard dose therapy, and the lowest ACS recurrence rates are for people who achieve the lowest LDL‐C levels.6 Further, combination statin and ezetimibe therapy achieves a 24% greater reduction in LDL‐C level than statin alone, and is associated with significantly lower cardiovascular event rates during long term follow‐up.7
Local and overseas guidelines recommend lipid‐lowering therapy for people who have experienced ACS (class 1 [strong] recommendation).8,9,10 Australian guidelines recommend that therapy be goal‐directed, with the aim of reducing LDL‐C to 1.8 mmol/L or lower or total cholesterol to 4.0 mmol/L or lower, and that patients with ACS commence intensive anti‐lipid therapy early in their index admission.10,11 However, the Cooperative National Registry of Acute Coronary Care, Guideline Adherence and Clinical Events (CONCORDANCE) reported in 2019 that only 55% of patients were using intensive lipid‐lowering therapy 12 months after discharge from hospital.12 This probably contributes to suboptimal control of LDL‐C levels in many people, but neither this nor other factors that influence whether people reach target lipid levels have been investigated in Australia.
We therefore analysed CONCORDANCE lipid levels data for people assessed six or 12 months after hospitalisation with ACS in Australia. We also examined factors associated with not achieving target lipid levels by the time of follow‐up.
Methods
CONCORDANCE was a prospective ACS registry that enrolled patients in 43 geographically diverse Australian hospitals during 2009–2018.13 Each month, participating hospitals recruited the first ten adult patients (18 years or older) admitted with ACS who had ACS‐associated electrocardiographic changes, elevated cardiac biomarkers, new or previously documented coronary artery disease, or two high risk comorbid conditions, such as diabetes and chronic kidney disease. Patients with ACS of non‐cardiovascular origin (eg, trauma, anaemia) were not recruited. Informed consent to inclusion in the registry was obtained from patients in an opt‐out process. Data on pre‐hospital management, demographic characteristics and medical history, and in‐hospital investigations and management were collected. Clinical events and reported use of prescribed therapy were assessed six months after discharge from hospital; patients recruited during 2014–2018 were also followed‐up at 12 months.
Medications of interest were evidence‐based lipid‐reducing therapies, particularly statins and ezetimibe. Intensive lipid‐lowering therapy was defined as atorvastatin (40 mg/day or more), rosuvastatin (20 mg/day or more), or simvastatin (80 mg/day or more), with or without ezetimibe; lower intensity lipid‐lowering therapy was defined as standard statin therapy with ezetimibe, or ezetimibe alone.8 Biochemical and clinical data were also collected, including lipid levels measured on admission and at follow‐up.
Study endpoint
The study endpoint was not achieving target lipid levels (LDL‐C ≤ 1.8 mmol/L or total cholesterol ≤ 4.0 mmol/L) by the most recent follow‐up (six or 12 months). As LDL‐C levels are more strongly associated with clinical outcomes than those of total cholesterol, LDL‐C data were used to determine whether target level was achieved if only one of the lipid targets was achieved, regardless of total cholesterol level. We included LDL‐C measurements at 12 months if available, and 6‐month measurements if they were not. If LDL‐C measurements were unavailable for both the 6‐ and 12‐month follow‐ups, total cholesterol measurements at 12 months were included, or 6‐month values if 12‐month measurements were unavailable (Box 1).
Statistical analysis
Categorical historical data missing at baseline were coded as “no” or “normal”, assuming that if it was not documented, it had not been present. Other missing data were not included in analyses. Categorical variables were summarised as counts and proportions, and the Rao–Scott test was used to correct for the clustering effect of the hospitals and to test for difference between the two groups.14 Numeric variables were summarised as means and standard deviations (SDs), and the difference between the two groups was assessed in a generalised estimating equations (GEE) model, adjusted only for the effect of hospital clustering. Factors associated with not achieving lipid targets were assessed by adjusted logistic regression within the GEE framework; covariates were included in the adjusted model according to their clinical or statistical significance in unadjusted analyses: sex, age group (under 55, 55–64, 65–73, 74 or more years), any lipid‐lowering therapy at time of admission, total cholesterol level greater than 4 mmol/L at time of admission, intensive statin therapy prescribed at discharge, and number of evidence‐based medications prescribed at discharge. In subgroup analyses, adjusted logistic regression models were recalculated using only LDL‐C or total cholesterol measurements. P < 0.05 was deemed statistically significant. All analyses were conducted in SAS 9.4.
Ethics approval
Each hospital obtained human research ethics committee approval prior to participating in CONCORDANCE. Approval for the analysis reported in this article was provided by the Sydney Local Health District (Concord Repatriation General Hospital) Human Research Ethics Committee (HREC/08/CRGH/180).
Results
Of 10 578 patients with ACS enrolled in CONCORDANCE who left hospital alive, lipid measurements were recorded at 6‐ or 12‐month follow‐ups for 2671 people (Box 1). The baseline clinical characteristics of this group were similar to those of the entire CONCORDANCE population. Their mean age was 63 years (SD, 12 years), and 1951 were men (73%) (Supporting Information, table 1). A total of 1194 people had not achieved lipid targets at the time of their most recent follow‐up (45%; 360 of 818 at 6 months [44%], 834 of 1853 at 12 months [45%]) (Box 2).
Evidence‐based therapy before and on discharge from the index admission
Intensive lipid‐lowering therapy was used at the time of admission by 334 people who did not achieve lipid targets (28%) and by 436 who did (30%). Intensive lipid‐lowering therapy was prescribed on discharge for 2077 people, including 876 of those who did not achieve lipid targets (73%) and 1201 of those who did (81%; P < 0.001). The proportion of patients prescribed fewer than four evidence‐based therapies on discharge was larger for those who did not achieve their target (331 [28%] v 302 [20%]; P < 0.001) (Box 3).
Lipid‐lowering treatment at most recent follow‐up
At the most recent follow‐up, intensive lipid‐lowering therapy was being used by 480 of 1194 people who had not reached their lipid target (40%) and by 692 of 1477 who had (47%; P = 0.033). Conversely, 692 of 1172 people using intensive therapy (59%), 238 of 433 using non‐intensive therapy (55%), and 75 of 257 using no lipid‐lowering therapy (29%) had achieved their lipid target. The proportion not using any lipid‐lowering therapy was larger for those who had not achieved the lipid target than those who had (182, 15% v 75, 5%; P < 0.001) (Box 4).
Factors associated with not achieving lipid targets
Women were more likely than men to not reach lipid targets (odds ratio [OR], 1.54; 95% confidence interval [CI], 1.20–1.99), and people under 65 years of age were more likely than those aged 75 years or more to not achieve them. Any lipid‐lowering therapy on admission (OR, 1.51; 95% CI, 1.16–1.96), total cholesterol exceeding 4 mmol/L on admission (OR, 3.09; 95% CI, 2.36–4.04), fewer than four evidence‐based medications prescribed at discharge, and intensive lipid‐lowering therapy not being prescribed at discharge (OR, 1.61; 95% CI, 1.17–2.21) were each associated with greater likelihood of not reaching lipid targets (Box 5). Similar results were yielded by subgroup analyses restricted to the 2004 people for whom LDL‐C measurements were available (Supporting Information, table 2) or the 667 people for whom total cholesterol levels but not LDL‐C were available (Supporting Information, table 3).
Discussion
We found that 45% of people hospitalised with ACS in Australia did not achieve recommended lipid levels by their most recent follow‐up (six or 12 months after discharge). People under 65 years of age, those who had used any lipid‐lowering therapy prior to hospitalisation or with higher cholesterol levels on admission, patients prescribed fewer than four evidence‐based therapies or not prescribed intensive lipid‐lowering therapy on discharge, and women were more likely to not reach lipid level targets by follow‐up.
Other cohort studies of lipid target attainment after ACS suggest that outcomes were poorer for women because they were typically older, less likely to be prescribed or to adhere to therapy, and more likely to not tolerate statins.15,16,17 Sex differences in the protective effect of high‐density lipoprotein cholesterol (HDL‐C) may cause practitioners to underestimate cardiovascular risk in women.17,18 Our secondary analysis restricted to people with LDL‐C data indicated that women were also more likely to not achieve this specific target. As LDL‐C levels are independent of those of HDL‐C, we concluded that higher HDL‐C levels did not appreciably influence whether women achieved lipid targets.
Using lipid‐lowering therapy at the time of admission was associated with not achieving target lipid levels by follow‐up, perhaps because prior intolerance or therapeutic inertia made intensifying these therapies less likely. However, several studies have found that patients using statins at the time of presentation with ACS were less likely to experience myocardial infarction or in‐hospital ischaemic complications.19,20
Prescribing discharged patients fewer than four evidence‐based therapies for ACS was associated with greater likelihood of not achieving lipid goals, suggesting a broader association between failure to provide evidence‐based care and inadequate treatment with lipid‐lowering therapies, as also documented in other studies.21,22,23
An earlier analysis of CONCORDANCE data found that 45% of people who had been hospitalised with ACS were not using intensive lipid‐lowering therapy six or 12 months after discharge.12 Our new analysis extends this finding by quantifying the impact of undertreatment on lipid levels: 16% of people who had not achieved target levels were using moderate or low dose statin therapy and 15% were on no lipid‐lowering therapy at all. This suggests that outcomes could be improved by increasing the use of evidence‐based therapy.
Further, 37% of people who did not achieve lipid targets were using high‐intensity statin therapy without ezetimibe,7 although even modest reductions in serum cholesterol levels are associated with lower rates of ACS recurrence and improved survival.3 In the United States, guidelines recommend prescribing the highest dose of lipid‐lowering agent tolerated by the patient.8 This “fire and forget” strategy contrasts with the Australian goal‐targeted approach to lipid control; our practice guidelines recommend a “treat to target” strategy, titrating anti‐lipid agents or using combinations of medications to achieve the target LDL‐C and cholesterol levels in patients at high risk of ACS recurrence.10,11 This strategy not only ensures that peoples achieve the targets, but also facilitates regular monitoring of their adherence and response to therapy and of medication intolerance.
For patients who are resistant to or do not tolerate standard lipid‐lowering medications, newer agents could also be considered. For example, a recent Cochrane meta‐analysis found high grade evidence that proprotein convertase subtilisin/kexin type 9 (PCSK9) monoclonal antibodies (eg, evolocumab, alirocumab), used as adjuncts to statin or ezetimibe therapy, reduced all‐cause mortality and the recurrence of myocardial infarction at 6‐ and 12‐month follow‐ups.24 These agents were not available during the CONCORDANCE data collection period, but evolocumab has since been approved in Australia, in combination with statins or alone for statin‐intolerant patients, for secondary prevention in people at greatest risk of recurrent events.25,26 Alirocumab is available under the Pharmaceutical Benefits Scheme for patients with familial hypercholesterolaemia.27 Inclisiran, a small interfering RNA that inhibits PCSK9 translation, was found in phase III trials to reduce LDL‐C levels by 50%. This agent, which is administered only twice a year, is not yet approved in Australia, but could revolutionise lipid‐lowering therapy, especially for people at high risk of cardiovascular disease.28
Limitations
As we did not examine why people were not using high‐intensity statin therapy, we could not determine the influence of prescribing patterns and drug intolerance on lipid‐lowering therapy use. Information on use of anti‐lipid therapy at time of follow‐up relied on patient report, and was not objectively validated. Lipid level measurement methods differed between participating centres. Further, we used different criteria for lipid target outcomes (lipid, time point) according to the availability of corresponding data. We did not assess lifestyle factors that may influence LDL‐C levels in some people. Survival bias may have affected our findings, as our analysis included only patients who survived to follow‐up. Unmeasured confounders not reported or not collected from medical records by CONCORDANCE, an observational registry, may have influenced treatment and lipid levels.
Conclusions
During 2009–2018, about 45% of people admitted to hospital in Australia with ACS did not achieve target serum lipid levels within 12 months of discharge, including a considerable proportion of people prescribed intensive statin therapy on discharge. Women, and people not prescribed intensive lipid‐lowering therapy or at least four evidence‐based medications on discharge from hospital were particularly unlikely to meet lipid targets. Optimising lipid levels in people at high risk of recurrent ACS could be achieved by dose escalation, combination therapies, and the adoption of newer lipid‐lowering agents.
Box 1 – Selection of patients enrolled in CONCORDANCE (2009–2018) for our analysis of follow‐up lipid levels
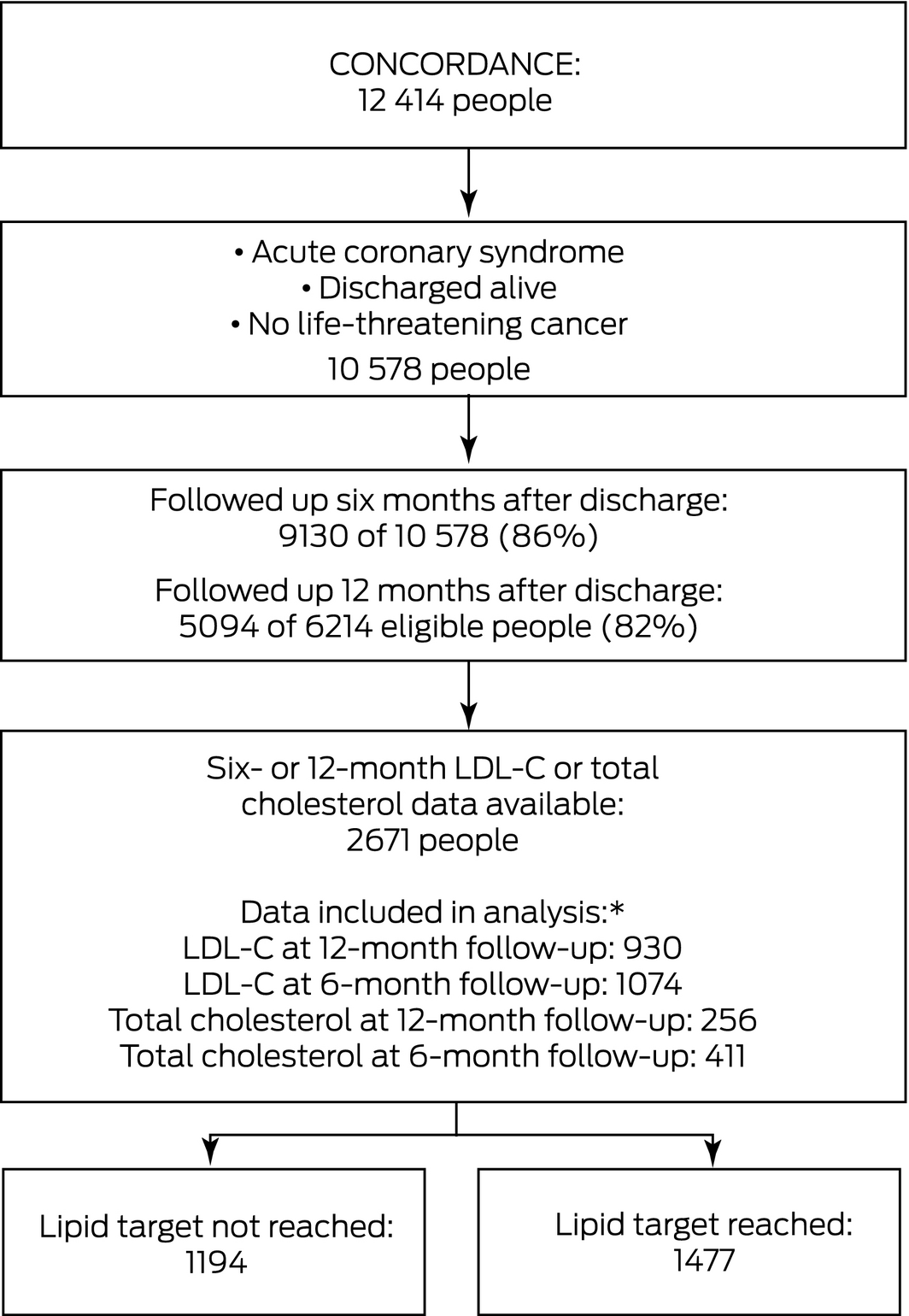
LDL‐C = low‐density lipoprotein cholesterol. * Data types are listed in order of preference for our study; that is, data were included from the highest category for which they were available.
Box 2 – Demographic and medical characteristics of 2671 patients at the time of their admission to hospital with acute coronary syndrome, by achievement of lipid target
|
|
|
Lipid target* |
|
||||||||||||
|
Characteristic |
All patients |
Not achieved |
Achieved |
P |
|||||||||||
|
|
|||||||||||||||
|
Number of patients |
2671 |
1194 [45%] |
1477 [55%] |
|
|||||||||||
|
Demographic characteristics |
|
|
|
|
|||||||||||
|
Age (years), mean (SD) |
63.3 (12.2) |
62.1 (12.2) |
64.3 (12.0) |
0.003 |
|||||||||||
|
Sex (women) |
720 (27%) |
373 (31%) |
347 (23%) |
< 0.001 |
|||||||||||
|
Medical history |
|
|
|
|
|||||||||||
|
Prior myocardial infarction |
722 (27%) |
313 (26%) |
409 (28%) |
0.43 |
|||||||||||
|
Congestive heart failure |
160 (6%) |
57 (5%) |
103 (7%) |
0.002 |
|||||||||||
|
Prior percutaneous coronary intervention |
571 (21%) |
245 (21%) |
326 (22%) |
0.35 |
|||||||||||
|
Prior coronary artery bypass grafting |
305 (11%) |
119 (10%) |
186 (13%) |
0.04 |
|||||||||||
|
Prior atrial fibrillation |
242 (9%) |
97 (8%) |
145 (10%) |
0.14 |
|||||||||||
|
Chronic renal failure |
248 (9%) |
109 (9%) |
139 (9%) |
0.83 |
|||||||||||
|
On dialysis |
48 (19%) |
13 (12%) |
35 (25%) |
0.08 |
|||||||||||
|
Prior stroke/transient ischaemic attack |
156 (6%) |
60 (5%) |
96 (7%) |
0.14 |
|||||||||||
|
Diabetes |
783 (29%) |
288 (24%) |
495 (34%) |
0.001 |
|||||||||||
|
Hypertension |
1639 (61%) |
702 (59%) |
937 (63%) |
0.04 |
|||||||||||
|
Dyslipidaemia |
1534 (58%) |
706 (59%) |
828 (56%) |
0.17 |
|||||||||||
|
History of smoking |
693 (26%) |
365 (31%) |
328 (22%) |
< 0.001 |
|||||||||||
|
Family history of coronary heart disease |
928 (35%) |
422 (35%) |
506 (34%) |
0.61 |
|||||||||||
|
Lipid levels (index admission) |
|
|
|
|
|||||||||||
|
LDL‐C (mmol/L), mean (SD) |
2.7 (1.1) |
3.0 (1.2) |
2.4 (1.0) |
< 0.001 |
|||||||||||
|
LDL‐C > 1.8 mmol/L |
1239 (75%) |
634 (86%) |
605 (66%) |
< 0.001 |
|||||||||||
|
Total cholesterol (mmol/L), mean (SD) |
4.6 (1.3) |
5.0 (1.3) |
4.4 (1.2) |
< 0.001 |
|||||||||||
|
Total cholesterol (> 4 mmol/L) |
1253 (66%) |
667 (77%) |
586 (56%) |
< 0.001 |
|||||||||||
|
|
|||||||||||||||
|
LDL‐C = low‐density lipoprotein cholesterol; SD = standard deviation. * LDL‐C ≤ 1.8 mmol/L (preferred) or total cholesterol ≤ 4 mmol/L six or 12 months after index admission (most recent available data used). |
|||||||||||||||
Box 3 – In‐hospital management and discharge medications prescribed for 2671 patients admitted to hospital with acute coronary syndrome, by achievement of lipid target
|
|
|
Lipid target* |
|
||||||||||||
|
Characteristic |
All patients |
Not achieved |
Achieved |
P |
|||||||||||
|
|
|||||||||||||||
|
Number of patients |
2671 |
1194 |
1477 |
|
|||||||||||
|
In‐hospital treatment |
|
|
|
0.10 |
|||||||||||
|
Coronary artery bypass grafting |
242 (9%) |
107 (9%) |
135 (9%) |
|
|||||||||||
|
Percutaneous coronary intervention |
1364 (51%) |
581(49%) |
783 (53%) |
|
|||||||||||
|
Medical management |
1065 (40%) |
506 (42%) |
559 (38%) |
|
|||||||||||
|
Any lipid‐lowering therapy (admission) |
1271 (48%) |
542 (45%) |
729 (49%) |
0.041 |
|||||||||||
|
Intensive lipid lowering therapy† † (admission) |
770 (29%) |
334 (28%) |
436 (30%) |
0.48 |
|||||||||||
|
Intensive lipid‐lowering therapy† † (discharge) |
2077 (78%) |
876 (73%) |
1201 (81%) |
< 0.001 |
|||||||||||
|
Evidence‐based medications prescribed‡ ‡ (discharge) |
|
|
|
< 0.001 |
|||||||||||
|
0 |
30 (1%) |
22 (2%) |
8 (1%) |
|
|||||||||||
|
1 |
44 (2%) |
31 (3%) |
13 (1%) |
|
|||||||||||
|
2 |
138 (5%) |
78 (7%) |
60 (4%) |
|
|||||||||||
|
3 |
421 (16%) |
200 (17%) |
221 (15%) |
|
|||||||||||
|
4 |
799 (30%) |
341 (29%) |
458 (31%) |
|
|||||||||||
|
5 |
1239 (46%) |
522 (44%) |
717 (49%) |
|
|||||||||||
|
|
|||||||||||||||
|
LDL‐C = low‐density lipoprotein cholesterol; SD = standard deviation. * LDL‐C ≤ 1.80 mmol/L (preferred) or total cholesterol ≤ 4 mmol/L six or 12 months after index admission (most recent available data used). † Atorvastatin (≥ 40 mg/day), rosuvastatin (≥ 20 mg/day), or simvastatin (≥ 80 mg/day), with or without ezetimibe. ‡ Aspirin, P2Y12 receptor antagonists (eg, ticagrelor), beta blockers, statins, angiotensin‐converting‐enzyme inhibitors. |
|||||||||||||||
Box 4 – Lipid‐lowering treatment used at time of most‐recent follow‐up, by achievement of lipid target
|
|
|
Lipid target* |
|
||||||||||||
|
Characteristic† † |
All patients |
Not achieved |
Achieved |
P |
|||||||||||
|
|
|||||||||||||||
|
Number of patients |
2671 |
1194 |
1477 |
|
|||||||||||
|
Intensive lipid‐lowering therapy‡ ‡ |
1172 (44%) |
480 (40%) |
692 (47%) |
0.033 |
|||||||||||
|
High dose statin only |
1096 (41%) |
445 (37%) |
651 (44%) |
0.038 |
|||||||||||
|
High dose statin and ezetimibe |
76 (3%) |
35 (3%) |
41 (3%) |
0.78 |
|||||||||||
|
Non‐intensive lipid‐lowering therapy |
433 (16%) |
195 (16%) |
238 (16%) |
0.92 |
|||||||||||
|
Moderate or low dose statin only |
389 (15%) |
176 (15%) |
213 (14%) |
0.88 |
|||||||||||
|
Moderate or low dose statin and ezetimibe |
44 (2%) |
19 (2%) |
25 (2%) |
0.86 |
|||||||||||
|
Ezetimibe only |
21 (1%) |
14 (1%) |
7 (0.5%) |
0.007 |
|||||||||||
|
Other lipid‐lowering therapy |
268 (10%) |
132 (11%) |
136 (9%) |
0.24 |
|||||||||||
|
No lipid‐lowering therapy |
257 (10%) |
182 (15%) |
75 (5%) |
< 0.001 |
|||||||||||
|
|
|||||||||||||||
|
LDL‐C = low‐density lipoprotein cholesterol; SD = standard deviation. * LDL‐C ≤ 1.80 mmol/L (preferred) or total cholesterol ≤ 4 mmol/L six or 12 months after index admission (most recent available data used). † Missing data on statin type and dose: 789 patients; missing data on other lipid‐lowering therapy at 6‐ or 12‐month follow‐up: 40 patients; missing data on statin and other lipid‐lowering therapy at 6‐ or 12‐month follow‐up: 26 patients. ‡ Atorvastatin (≥ 40 mg/day), rosuvastatin (≥ 20 mg/day), or simvastatin (≥ 80 mg/day), with or without ezetimibe. |
|||||||||||||||
Box 5 – Influence of selected factors on not achieving lipid targets by most recent follow‐up after admission with acute coronary syndrome: multiple logistic regression*
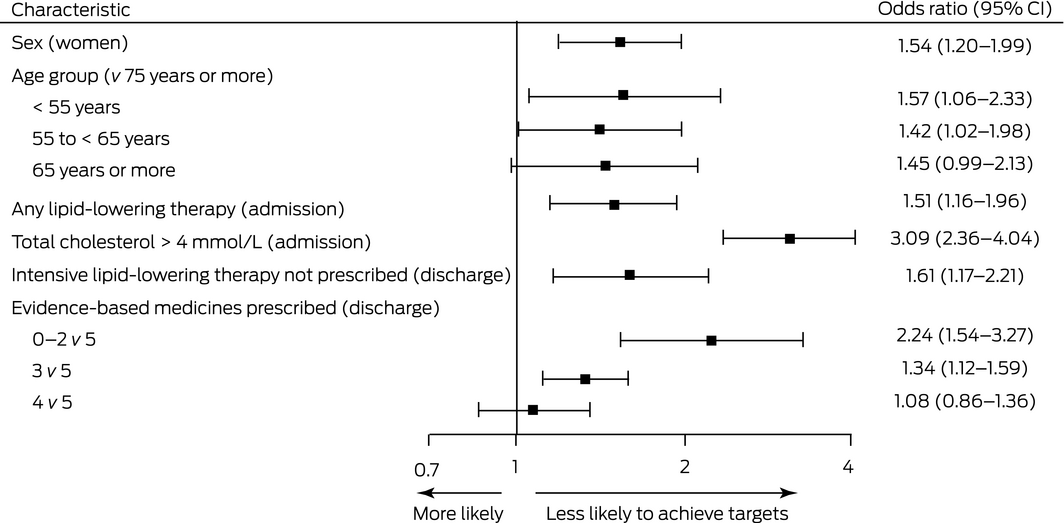
CI = confidence interval. * Based on data for 1039 people who achieved and 867 who did not achieve the lipid target; data for 760 people were missing because total cholesterol level at admission was not available, and for five because age data were not available.
Received 27 April 2021, accepted 11 October 2021
- Noor Alsadat1
- Karice Hyun2,3
- Farzaneh Boroumand4
- Craig Juergens1
- Leonard Kritharides2,4
- David B Brieger2
- 1 Liverpool Hospital, Sydney, NSW
- 2 Concord Repatriation General Hospital, Sydney, NSW
- 3 The University of Sydney, Sydney, NSW
- 4 ANZAC Research Institute, Sydney, NSW
The CONCORDANCE national registry was funded by unrestricted grants from Sanofi Aventis, Astra Zeneca, Eli Lilly, Boehringer Ingelheim, MSD/Schering Plough Joint Venture, and the National Heart Foundation of Australia.
No relevant disclosures.
- 1. Australian Institute of Health and Welfare. Cardiovascular disease (Cat. no. CVD 83). 15 July 2020. https://www.aihw.gov.au/getmedia/36485187‐b5a4‐4db3‐b4b5‐e79da76dde48/Cardiovascular‐disease.pdf.aspx?inline=true (viewed Sept 2020).
- 2. Fujisue K, Tsujita K. Current status of lipid management in acute coronary syndrome. J Cardiol 2017; 70: 101–106.
- 3. Cholesterol Treatment Trialists Collaboration; Baigent C, Blackwell L, Emberson J, et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta‐analysis of data from 170 000 participants in 26 randomised trials. Lancet 2010; 376: 1670–1681.
- 4. Hirsh BJ, Smilowitz NR, Rosenson RS, et al. Utilization of and adherence to guideline‐recommended lipid‐lowering therapy after acute coronary syndrome: opportunities for improvement. J Am Coll Cardiol 2015; 66: 184–192.
- 5. Schwartz GG, Olsson AG, Ezekowitz MD, et al; Myocardial Ischemia Reduction with Aggressive Cholesterol Lowering (MIRACL) Study Investigators. Effects of atorvastatin on early recurrent ischemic events in acute coronary syndromes: the MIRACL study: a randomized controlled trial. JAMA 2001; 285: 1711–1718.
- 6. Ray KK, Cannon CP, McCabe CH, et al; PROVE IT‐TIMI 22 Investigators. Early and late benefits of high‐dose atorvastatin in patients with acute coronary syndromes: results from the PROVE IT‐TIMI 22 trial. J Am Coll Cardiol 2005; 46: 1405–1410.
- 7. Cannon CP, Blazing MA, Giugliano RP, et al; IMPROVE‐IT Investigators. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med 2015; 372: 2387–2397.
- 8. Grundy SM, Stone NJ, Bailey AL, et al. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. J Am Coll Cardiol 2019; 73: e285–e350.
- 9. Stone NJ, Robinson JG, Lichtenstein AH, et al; American College of Cardiology, American Heart Association Task Force on Practice Guidelines. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association task force on practice guidelines. J Am Coll Cardiol 2014; 63: 2889–2934.
- 10. Chew DP, Scott IA, Cullen L, et al. National Heart Foundation of Australia and Cardiac Society of Australia and New Zealand: Australian clinical guidelines for the management of acute coronary syndromes 2016. Med J Aust 2016; 205: 128–133. https://www.mja.com.au/journal/2016/205/3/national‐heart‐foundation‐australia‐and‐cardiac‐society‐australia‐and‐new
- 11. Therapeutic Guidelines. Lipid modification: target lipid levels. In: Cardiovascular. 2018. https://www.tg.org.au (viewed Feb 2021).
- 12. Brieger D, D’Souza M, Hyun K, et al. Intensive lipid‐lowering therapy in the 12 months after an acute coronary syndrome in Australia: an observational analysis. Med J Aust 2019; 210: 80–85. https://www.mja.com.au/journal/2019/210/2/intensive‐lipid‐lowering‐therapy‐12‐months‐after‐acute‐coronary‐syndrome
- 13. Aliprandi‐Costa B, Ranasinghe I, Turnbull F, et al. The design and rationale of the Australian Cooperative National Registry of Acute Coronary care, Guideline Adherence and Clinical Events (CONCORDANCE). Heart Lung Circ 2013; 22: 533–541.
- 14. Rao JNK, Scott AJ. A simple method for the analysis of clustered binary data. Biometrics 1992; 48: 577–585.
- 15. Dallongevillle J, De Bacquer D, Heidrich J, et al; EUROASPIRE Study Group. Gender differences in the implementation of cardiovascular prevention measures after an acute coronary event. Heart 2010; 96: 1744–1749.
- 16. Victor BM, Teal V, Ahedor L, Karalis DG. Gender differences in achieving optimal lipid goals in patients with coronary artery disease. Am J Cardiol 2014; 113: 1611–1615.
- 17. Ahmad Z. Statin intolerance. Am J Cardiol 2014; 113: 1765–1771.
- 18. Cooney MT, Dudina A, De Bacquer D, et al; SCORE investigators. HDL cholesterol protects against cardiovascular disease in both genders, at all ages and at all levels of risk. Atherosclerosis 2009; 206: 611–616.
- 19. Li M, Huang Y, Du X, et al. Impact of prior use of four preventive medications on outcomes in patients hospitalized for acute coronary syndrome: results from CPACS‐2 Study. PLoS One 2016; 11: e0163068.
- 20. Spencer FA, Allegrone J, Goldberg RJ, et al; GRACE Investigators. Association of statin therapy with outcomes of acute coronary syndromes: the GRACE study. Ann Intern Med 2004; 140: 857–866.
- 21. Yan AT, Yan RT, Tan M, et al; Canadian ACS Registries Investigators. Optimal medical therapy at discharge in patients with acute coronary syndromes: temporal changes, characteristics, and 1‐year outcome. Am Heart J 2007; 154: 1108–1115.
- 22. Bagnall AJ, Yan AT, Yan RT, et al; Canadian Acute Coronary Syndromes Registry II Investigators. Optimal medical therapy for non‐ST‐segment‐elevation acute coronary syndromes: exploring why physicians do not prescribe evidence‐based treatment and why patients discontinue medications after discharge. Circ Cardiovasc Qual Outcomes 2010; 3: 530–537.
- 23. Roe MT, Peterson ED, Newby LK, et al. The influence of risk status on guideline adherence for patients with non‐ST‐segment elevation acute coronary syndromes. Am Heart J 2006; 151: 1205–1213.
- 24. Schmidt AF, Carter JPL, Pearce LS, et al. PCSK9 monoclonal antibodies for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst Rev 2020; 10: CD011748.
- 25. Scherer DJ, Nelson AJ, O’Brien R, et al. Status of PCSK9 monoclonal antibodies in Australia. Heart Lung Circ 2019; 28: 1571–1579.
- 26. Australian Department of Health. Evolocumab. Pharmaceutical Benefits Scheme, 2020. https://www.pbs.gov.au/medicine/item/11193D‐11485L‐11972D‐11986W (viewed Jan 2021).
- 27. Australian Department of Health. Alirocumab. Pharmaceutical Benefits Scheme, 2020. https://www.pbs.gov.au/medicine/item/12608n (viewed Feb 2022).
- 28. Ray KK, Wright RS, Kallend D, et al; ORION‐10 and ORION‐11 Investigators. Two phase 3 trials of Inclisiran in patients with elevated LDL cholesterol. N Engl J Med 2020; 382: 1507–1519.





Abstract
Objectives: To assess lipid levels in people six or 12 months after hospitalisation with acute coronary syndrome (ACS); to identify factors associated with not achieving lipid level targets.
Design, setting: Retrospective cohort study; analysis of data from CONCORDANCE, an Australian ACS registry, 2009–2018.
Participants: Adult patients who had experienced confirmed ACS of cardiovascular origin, for whom serum lipid levels had been assessed on admission and six or 12 months after discharge.
Main outcome measures: Not achieving lipid targets by most recent follow‐up (in order of priority: low‐density lipoprotein cholesterol [LDL‐C] ≤ 1.8 mmol/L or total cholesterol ≤ 4 mmol/L); factors associated with not achieving target lipid levels.
Results: Lipid levels measured at 6‐ or 12‐month follow‐up were available for 2671 of 10 578 people discharged from hospital alive; 1194 (45%) had not achieved lipid targets at their most recent follow‐up, including 876 (73%) who had been prescribed intensive lipid‐lowering therapy at discharge. People under 65 years of age, those using lipid‐lowering therapy or with higher cholesterol levels on admission, patients prescribed fewer than four evidence‐based therapies or not prescribed intensive lipid‐lowering therapy on discharge, and women were more likely to not reach lipid level targets.
Conclusion: Almost half the patients did not achieve target lipid levels within 12 months of an admission to hospital with ACS. These people are at elevated risk of recurrent cardiovascular disease, and therapy could be optimised (eg, dose escalation, drug combinations, novel therapies) to improve outcomes.