- The known: People who have experienced an acute coronary syndrome (ACS) are at high risk of further events. High blood cholesterol is an important modifiable factor that increases the risks of both initial and subsequent ACS events.
- The new: Only 55% of patients treated in Australia for ACS were undergoing intensive lipid‐lowering therapy 6 or 12 months after their hospitalisation. The major predictor of not receiving such therapy at follow‐up was its not being prescribed at hospital discharge.
- The implications: Improving oral lipid‐lowering therapy for people who have had an ACS should prevent recurrent coronary events.
Acute coronary syndromes (ACS) are a major cause of morbidity and mortality in Australia;1 more than 120 000 hospitalisations for ACS are recorded each year, and about 6% end with the patient dying in hospital.1
Patients who have experienced an ACS are at high risk of further events, so that more than one‐third of all ACS admissions are of patients who have had previous events.2 Reducing the cholesterol levels of patients who have presented with an ACS is an important secondary preventive goal. Treatment with statins reduces event rates among patients with coronary disease by 25–40%,3 and these benefits are apparent within a few months of an ACS.4 Higher intensity therapy is more effective than lower dose therapy, and providing high intensity statin therapy before hospital discharge is accorded the highest grade of recommendation in most clinical practice guidelines.5,6 A further modest improvement in outcomes for patients receiving statin therapy is achieved by adding non‐statin lipid‐lowering therapies, such as the cholesterol absorption inhibitor ezetimibe,7 which can also be prescribed for patients who do not tolerate statins. Adherence is one of the most important determinants of outcomes for people who have had an ACS,8 but only 50–60% of patients adhere to statin therapy one year after starting.9,10
Patterns of high intensity lipid‐lowering therapy for patients who have had an ACS and adherence to therapy during the year after discharge from acute care have not previously been well described in Australia. Our aim was to determine the prevalence and predictors of people hospitalised with ACS receiving high intensity lipid‐lowering therapy during the 12 months after their discharge from hospital.
Methods
The Cooperative National Registry of Acute Coronary care, Guideline Adherence and Clinical Events (CONCORDANCE) is a prospective, Australian investigator‐initiated clinical quality registry.11 Initiated in 2009, it collects detailed clinical data from 42 representative hospitals around Australia on the management of and outcomes for patients with ACS, including medications during the acute hospital admission and at 6‐ and 12‐month follow‐ups. Data are collected on electronic case report forms and detail the patients’ pre‐hospital assessment and management, admission diagnosis, demographic information and medical history, and in‐hospital investigations and management. Patients are followed up 6 and 12 months after discharge from hospital for information on clinical events and medication compliance. Informed patient consent, required for collecting medical and post‐discharge information, is obtained during hospitalisation in an opt‐out process.
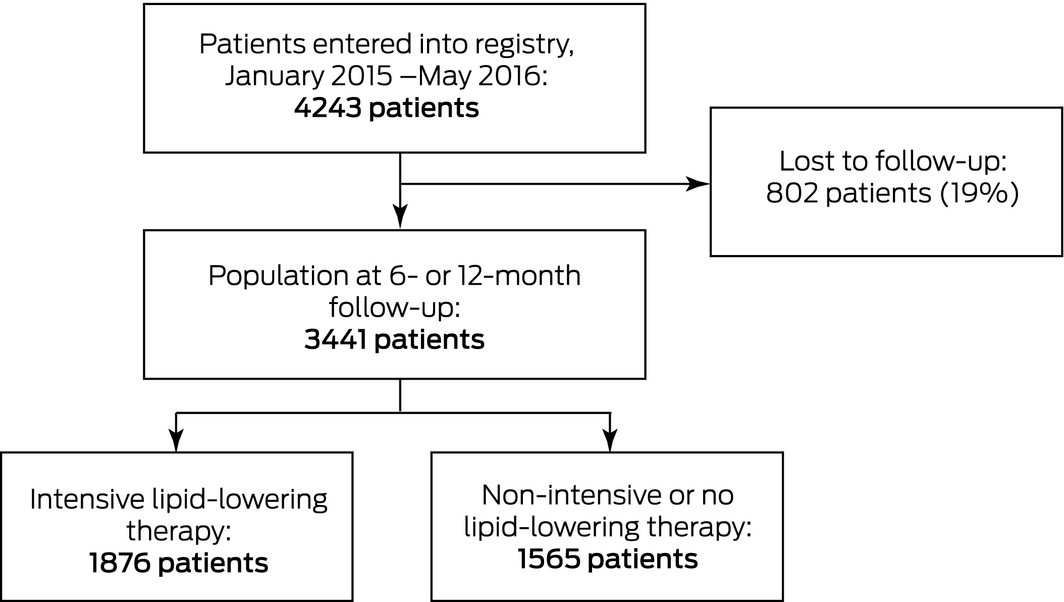
The data for our analysis were extracted from the CONCORDANCE database. The study sample comprised patients who were enrolled in CONCORDANCE between January 2015 and May 2016 with a confirmed diagnosis of ACS and survived to hospital discharge, and for whom data on the prescribing of lipid‐lowering therapy were available. To be eligible for inclusion in our study, information on lipid‐lowering therapy at the most recent follow‐up (6 or 12 months after the index admission) was required.
The endpoint of interest was whether a patient received intensive lipid‐lowering therapy, defined as treatment with atorvastatin (≥ 40 mg/day), rosuvastatin (≥ 20 mg/day), or simvastatin (≥ 80 mg/day), with or without ezetimibe; lower intensity statin therapy with ezetimibe; or ezetimibe alone. The cited statin doses were based on the American College of Cardiology/American Heart Association classification of intensity,12 with the exception of simvastatin at 80 mg/day or more; although it has been evaluated in randomised controlled trials, simvastatin is not recommended in the United States because of the increased risk of myopathy, including rhabdomyolysis. The advice in Australian prescribing information is that simvastatin at 80 mg/day should be prescribed only for patients at high risk of coronary events who have not achieved their treatment goals on lower doses.
Statistical analysis
Values for categorical variables were summarised as numbers and percentages for patients receiving intensive or non‐intensive lipid‐lowering therapy at their most recent follow‐up. The significance of differences between values for the two groups, after correcting for correlation resulting from the clustering effect of hospitals, was assessed in Rao–Scott χ2 tests.
Variables for which P < 0.05 in univariate analyses and variables deemed to be clinically important were included in generalised linear regression models for predicting the odds of the binary outcomes “intensive lipid‐lowering therapy at follow‐up” and “non‐intensive lipid‐lowering therapy at follow‐up”. The regression was conducted in the generalised estimating equation (GEE) framework13 to correct for correlation associated with the clustering effects of hospitals. An exchangeable correlation structure was assumed. Odds ratios with 95% confidence intervals (CIs) and P values for the type 3 analysis are presented. In a sensitivity analysis, subjects who were prescribed simvastatin (≥ 80 mg) at follow‐up were re‐classified as receiving non‐intensive lipid‐lowering therapy.
Independent variables from the final generalised linear model in the GEE framework were also included in a multilevel mixed model for determining the variance partition coefficient, which describes the proportion of the residual variation in the propensity to not being prescribed intensive lipid‐lowering therapy at the most recent follow‐up that is attributable to unmeasured hospital characteristics. This was depicted in a caterpillar plot of hospital residuals with 95% CIs. The hospital residuals represent departures from the overall mean; a hospital with a 95% CI that does not include zero (the mean log‐odds of not being prescribed intensive lipid‐lowering therapy at 6–12 months after the index admission for ACS) was deemed to differ significantly from the overall mean for all hospitals.
P < 0.05 was deemed statistically significant; all analyses were conducted in SAS 9.3 (SAS Statistical Institute).
Ethics approval
The Concord Repatriation General Hospital Human Ethics Research Committee granted ethics approval for this sub‐analysis (reference, HREC/08/CRGH/180). Each participating site obtained written local Human Research Ethics Committee approval before submitting data to CONCORDANCE.
Results
At the time of our analysis, CONCORDANCE included data for 8792 patients. As therapeutic dosage information was not initially collected by the registry, we selected data for the 4243 patients who had entered the study on or after the date their hospital started supplying this information to the registry. The final study population comprised 3441 patients who were alive at discharge and for whom information on lipid‐lowering therapy at discharge and at 6‐ (1431 patients) or 12‐month (2010 patients) follow‐ups were available. About half the population (1876 of 3441, 55%) were receiving intensive lipid‐lowering therapy, 826 (24%) some form of lipid‐lowering therapy other than intensive lipid‐lowering therapy, and 739 (22%) no lipid‐lowering therapy at the time of their most recent follow‐up (Box 1).
The mean age of patients not undergoing intensive lipid‐lowering therapy at follow‐up was higher and the proportion of women greater than those of people receiving intensive therapy, as was the proportion with historical evidence of coronary disease. However, their mean lipid levels at admission were not statistically different from those for the non‐intensive therapy group (Box 2). A smaller proportion of patients treated in rural locations received high intensity therapy than those treated in urban centres (45% v 61%). The socio‐economic status of the hospital location, the size of the hospital, and the presence of a catheterisation laboratory were not significantly different between the two patient groups (Box 3).
Patients not receiving intensive lipid‐lowering therapy at follow‐up had more frequently been medically managed in hospital (ie, without revascularisation) and had less frequently received intensive lipid‐lowering therapy prior to their admission. When discharged from hospital, they were less frequently prescribed intensive lipid‐lowering therapy, received fewer evidence‐based medications, and were less often referred to cardiac rehabilitation (Box 4).
In the regression model, not being prescribed intensive therapy at hospital discharge was the strongest independent predictor of not receiving high intensity lipid‐lowering therapy at follow‐up (odds ratio [OR], 7.24; 95% CI, 4.37–12.0). Not being prescribed intensive therapy prior to admission was also a significant predictor (OR, 1.53; 95% CI, 1.26–1.85), as were being female, being medically managed, undergoing coronary artery bypass grafting, and not being referred to a cardiac rehabilitation program (Box 5). Similar results were obtained in a sensitivity analysis in which 31 patients receiving simvastatin at dosages of at least 80 mg/day were re‐classified as receiving non‐intensive therapy (online Supporting Information).
The propensity to not receiving intensive lipid‐lowering therapy, adjusted for factors significant in the GEE model, differed significantly between the hospitals to which patients had been admitted, indicating that unmeasured hospital factors contributed some of the variation in receiving high dose lipid‐lowering therapy at follow‐up (Box 6). According to the GEE model, about 17% of residual variation in propensity to not be receiving intensive therapy at follow‐up was attributable to unmeasured hospital characteristics (hospital‐level variance = 0.680, individual‐level variance = 3.29; therefore variance partition coefficient = 0.680/[0.680 + 3.29] = 0.171).
Discussion
We found that only 55% of Australian patients reported receiving intensive lipid‐lowering therapy 6–12 months after an ACS. The most powerful predictor of not receiving such treatment was its not being prescribed at the time of hospital discharge. In addition, receiving high intensity therapy prior to admission was an independent positive predictor of therapy at follow‐up, consistent with previous reports.15 Re‐classifying treatment with simvastatin at doses of at least 80 mg as non‐intensive therapy did not substantially alter these findings.
Nearly one‐fifth of patients were not prescribed high intensity lipid‐lowering therapy at the time of their discharge from hospital. We did not investigate reasons for not prescribing it at discharge, but found that intensive therapy prescribing rates at follow‐up varied according to the hospitals to which the patients had presented with an ACS; the rates were significantly lower for patients from six of 23 hospitals and significantly higher for those from eight of 23 hospitals than the mean prescription rate for all hospitals. This suggests that hospital‐related factors had a significant effect on follow‐up therapy. There were no differences in the limited descriptive data on hospital characteristics we collected (location, size, catheterisation laboratory) between the intensive and non‐intensive therapy groups. However, we did not have information about practices likely to drive prescribing and adherence at the clinical level, such as effective implementation of evidence‐guided pathways or linkage with post‐discharge care. Our quantitative variance analysis indicated that these unmeasured hospital factors were responsible for 17% of variation in treatment at follow‐up, underscoring the important contribution of the health system to longer term adherence to evidence‐based therapy.
The 25 percentage point decline in the use of high intensity lipid‐lowering therapy between discharge and follow‐up is higher than reported from randomised trials (1.2–17.2% per year3), but at the lower end of the range reported by other observational registry studies, which have reported non‐adherence rates as high as 50%.16 In our study, not using appropriate high intensity therapy at follow‐up was independently associated with not being revascularised or undergoing coronary artery bypass grafting (each compared with receiving a percutaneous coronary intervention) and being female. Awareness of the importance of long term pharmacological therapy (in addition to dual antiplatelet therapy) for maintaining the benefits of a percutaneous coronary intervention may promote prescribing of and adherence to other secondary prevention therapies, including intensive lipid‐lowering therapy, among patients who have undergone such interventions.17 On the other hand, being female is associated with an increased risk of statin intolerance;18 if this was the reason for not providing high intensity lipid‐lowering therapy to women in our study, our data clearly suggest that it was not adequately compensated by the prescribing of the statin‐sparing agent ezetimibe.
A recent simulation model based on data from a large US claims database indicated that providing high intensity statin therapy and ezetimibe to the entire population of people with atherosclerotic cardiovascular disease would achieve target lipid levels for 86% of patients, and that adjunctive proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitor therapy would allow the remaining 14% to also reach recommended levels.19 PCSK9 inhibitors are potent additions to the therapeutic armamentarium, achieving low levels of low‐density lipoprotein cholesterol (LDL‐C) in a subset of patients at high risk of coronary events for whom target LDL‐C levels have previously been unattainable.20 Their expense, however, mean that these agents are appropriate only for patients with lipid levels that remain persistently high despite statin therapy at the maximally tolerated intensity, with or without ezetimibe.
Limitations
We did not specifically determine why patients were not receiving intensive lipid‐lowering therapy at hospital discharge or at follow‐up. Adherence to a prescribed therapy is determined by complicated socio‐demographic factors, individual statin tolerance, access to and the nature of medical care, and the complexity and expense of medication regimens.21 We collected limited data on these factors; importantly, we could not determine whether the decision not to undertake intensive therapy was driven by the treating doctor not prescribing the medication or by the patient choosing not to use it. Further, data on receiving statin therapy relied on patient reports and were not objectively validated. We did not have reliable data on lipid measurements during the follow‐up period and could not determine the contribution of low LDL‐C levels to the setting of statin dosage during the follow‐up phase.
Conclusion
Only 55% of patients in Australia receive intensive lipid‐lowering therapy during the 12 months after an ACS. Not receiving such therapy is most strongly linked with its not being prescribed at hospital discharge, and adherence to therapy declines during the subsequent 12 months. Optimising oral lipid‐lowering therapy for people who have had an ACS should help prevent recurrent coronary events among people at high risk.
Box 1 – The selection of patients for inclusion in our analysis of statin therapy for patients with data in the CONCORDANCE registry
Box 2 – Characteristics of patients receiving or not receiving intensive lipid‐lowering therapy at their most recent follow‐up, 6 or 12 months after the index admission for treatment of acute coronary syndrome (ACS)
|
Variable |
Receiving intensive lipid‐lowering therapy at most recent follow‐up |
P |
|||||||||||||
|
Yes |
No |
||||||||||||||
|
|
|||||||||||||||
|
Number of patients |
1876 |
1565 |
|
||||||||||||
|
Demographic information |
|||||||||||||||
|
Age (years), mean (SD) |
63.4 (12.1) |
64.5 (13.9) |
0.009 |
||||||||||||
|
Sex (men) |
1438 (77%) |
1045 (67%) |
< 0.001 |
||||||||||||
|
Medical history/comorbid conditions |
|||||||||||||||
|
Previous myocardial infarction |
477 (25%) |
493 (32%) |
< 0.001 |
||||||||||||
|
Exertional angina pectoris |
318 (17%) |
369 (24%) |
0.006 |
||||||||||||
|
Congestive heart failure |
98 (5%) |
137 (9%) |
< 0.001 |
||||||||||||
|
Coronary artery disease on angiogram |
587 (31%) |
588 (38%) |
< 0.001 |
||||||||||||
|
Previous coronary intervention |
381 (20%) |
294 (19%) |
0.40 |
||||||||||||
|
Previous coronary artery bypass graft |
177 (9%) |
187 (12%) |
0.004 |
||||||||||||
|
Previous positive stress test |
107 (6%) |
116 (7%) |
0.07 |
||||||||||||
|
Previous atrial fibrillation |
123 (7%) |
175 (11%) |
< 0.001 |
||||||||||||
|
Previous deep vein thrombosis/pulmonary embolism |
52 (3%) |
67 (4%) |
0.023 |
||||||||||||
|
Previous major bleeding |
27 (1%) |
26 (2%) |
0.63 |
||||||||||||
|
Previous metal valve replacement |
11 (1%) |
23 (1%) |
< 0.001 |
||||||||||||
|
Permanent pacemaker |
41 (2%) |
44 (3%) |
0.24 |
||||||||||||
|
Implantable defibrillator |
16 (1%) |
19 (1%) |
0.20 |
||||||||||||
|
Chronic renal failure |
93 (5%) |
170 (11%) |
< 0.001 |
||||||||||||
|
Chronic renal failure, on dialysis |
11 (1%) |
30 (2%) |
0.26 |
||||||||||||
|
Previous stroke/transient ischaemic attack |
81 (4%) |
104 (7%) |
0.003 |
||||||||||||
|
Very high cardiovascular risk before ACS event* |
889 (47%) |
913 (58%) |
< 0.001 |
||||||||||||
|
Diabetes |
476 (25%) |
456 (29%) |
< 0.001 |
||||||||||||
|
Hypertension |
1127 (60%) |
973 (62%) |
0.40 |
||||||||||||
|
Dyslipidaemia |
1043 (56%) |
862 (55%) |
0.84 |
||||||||||||
|
Positive smoking history |
573 (31%) |
434 (28%) |
0.52 |
||||||||||||
|
Family history of coronary heart disease |
716 (38%) |
512 (33%) |
0.05 |
||||||||||||
|
Lipid levels at index admission |
|||||||||||||||
|
Low‐density lipoprotein cholesterol (mmol/L), mean (SD)† |
2.69 (1.06) |
2.64 (1.10) |
0.40 |
||||||||||||
|
Low‐density lipoprotein cholesterol > 1.81 mmol/L† |
1012 (76%) |
728 (74%) |
0.18 |
||||||||||||
|
Total cholesterol (mmol/L), mean (SD)‡ |
4.71 (2.01) |
4.69 (2.69) |
0.81 |
||||||||||||
|
Total cholesterol > 4 mmol/L‡ |
987 (66%) |
696 (63%) |
0.35 |
||||||||||||
|
|
|||||||||||||||
|
SD = standard deviation. * Defined as having any of the following: previous myocardial infarction; exertional angina pectoris; percutaneous coronary intervention; coronary artery bypass graft; peripheral arterial disease; stroke/transient ischaemic attack; chronic kidney disease AND diabetes; diabetes AND aged more than 60 years; diabetes AND Aboriginal or Torres Strait Islander. Intensive lipid‐lowering therapy group: n = 1323; non‐intensive or no statin therapy group: n = 986. Intensive lipid‐lowering therapy group: n = 1504, non‐intensive or no statin therapy group: n = 1097. |
|||||||||||||||
Box 3 – Characteristics of hospitals to which patients presented with acute coronary syndrome (ACS), according to whether patients were receiving intensive lipid‐lowering therapy at their most recent follow‐up (6 or 12 months after the index admission)
|
Variable |
Patient receiving intensive lipid‐lowering therapy at follow‐up |
P |
|||||||||||||
|
Yes |
No |
||||||||||||||
|
|
|||||||||||||||
|
Number of patients |
1876 |
1565 |
|
||||||||||||
|
Socio‐economic status, by quintile* |
|
|
0.29 |
||||||||||||
|
Quartile 1 (most disadvantaged) (n = 2) |
1/2 (50%) |
1/2 (50%) |
|
||||||||||||
|
Quartile 2 (n = 675) |
349/675 (52%) |
326/675 (48%) |
|
||||||||||||
|
Quartile 3 (n = 849) |
459/849 (54%) |
390/849 (46%) |
|
||||||||||||
|
Quartile 4 (n = 908) |
412/908 (45%) |
496/908 (55%) |
|
||||||||||||
|
Quartile 5 (least disadvantaged) (n = 779) |
523/779 (67%) |
256/779 (33%) |
|
||||||||||||
|
Size of hospital (number of beds) |
0.28 |
||||||||||||||
|
0–349 (n = 871) |
464/871 (53%) |
407/871 (47%) |
|
||||||||||||
|
350–499 (n = 948) |
416/948 (44%) |
532/948 (56%) |
|
||||||||||||
|
500–649 (n = 922) |
588/922 (64%) |
334/922 (36%) |
|
||||||||||||
|
≥ 650 (n = 700) |
408/700 (58%) |
292/700 (42%) |
|
||||||||||||
|
Location of hospital |
|
|
0.013 |
||||||||||||
|
Rural (n = 1409) |
630/1409 (45%) |
779/1409 (55%) |
|
||||||||||||
|
Urban (n = 2032) |
1246/2032 (61%) |
786/2032 (39%) |
|||||||||||||
|
Catheterisation laboratory in hospital |
0.09 |
||||||||||||||
|
Yes (n = 2923) |
1675/2923 (56%) |
1284/2923 (44%) |
|
||||||||||||
|
No (n = 482) |
201/482 (42%) |
281/482 (58%) |
|
||||||||||||
|
|
|||||||||||||||
|
* Index of Relative Socio‐Economic Disadvantage (IRSD),14 based on the postcode of the hospital. |
|||||||||||||||
Box 4 – In‐hospital treatment and medications associated with prescribing of intensive lipid‐lowering therapy at latest follow‐up (6 or 12 months) after index admission for treatment of acute coronary syndrome
|
Variable |
Patient receiving intensive lipid‐lowering therapy at follow‐up |
P |
|||||||||||||
|
Yes |
No |
||||||||||||||
|
|
|||||||||||||||
|
Number of patients |
1876 |
1565 |
|
||||||||||||
|
In‐hospital treatment |
|
|
< 0.001 |
||||||||||||
|
Coronary artery bypass grafting |
158 (8%) |
135 (9%) |
|
||||||||||||
|
Percutaneous coronary intervention |
1114 (59%) |
526 (34%) |
|
||||||||||||
|
Medically managed |
604 (32%) |
904 (58%) |
|
||||||||||||
|
Discharge from hospital |
|||||||||||||||
|
Referral to cardiac rehabilitation |
1428 (76%) |
911 (58%) |
< 0.001 |
||||||||||||
|
Number of evidence‐based medications prescribed at discharge, mean (SD) |
4.3 (0.9) |
3.8 (1.2) |
< 0.001 |
||||||||||||
|
Lipid‐lowering therapy prescribed prior to hospital admission |
|||||||||||||||
|
Intensive therapy |
|||||||||||||||
|
Intensive statin therapy alone |
512 (27%) |
293 (19%) |
< 0.001 |
||||||||||||
|
Intensive statin therapy and ezetimibe |
55 (3%) |
16 (1%) |
0.005 |
||||||||||||
|
Lower intensity statin therapy with ezetimibe |
32 (2%) |
10 (1%) |
0.007 |
||||||||||||
|
Ezetimibe alone |
35 (2%) |
22 (1%) |
0.46 |
||||||||||||
|
Non‐intensive therapy |
|||||||||||||||
|
Statin therapy other than intensive therapy |
195 (10%) |
350 (22%) |
< 0.001 |
||||||||||||
|
Other lipid‐lowering therapy |
14 (1%) |
20 (1%) |
0.23 |
||||||||||||
|
No lipid‐lowering therapy |
1033 (55%) |
854 (55%) |
0.88 |
||||||||||||
|
Lipid‐lowering therapy prescribed at discharge |
|||||||||||||||
|
Intensive therapy |
|||||||||||||||
|
Intensive statin therapy alone |
1642 (88%) |
907 (58%) |
< 0.001 |
||||||||||||
|
Intensive statin therapy and ezetimibe |
80 (4%) |
22 (1%) |
0.004 |
||||||||||||
|
Lower intensity statin therapy with ezetimibe |
29 (2%) |
13 (1%) |
0.08 |
||||||||||||
|
Ezetimibe alone |
26 (1%) |
22 (1%) |
0.96 |
||||||||||||
|
Non‐intensive therapy |
|||||||||||||||
|
Statin therapy other than intensive therapy |
55 (3%) |
378 (24%) |
< 0.001 |
||||||||||||
|
Other lipid‐lowering therapy |
3 (< 1%) |
14 (1%) |
< 0.001 |
||||||||||||
|
No lipid‐lowering therapy |
41 (2%) |
209 (13%) |
< 0.001 |
||||||||||||
|
|
|||||||||||||||
|
SD = standard deviation. |
|||||||||||||||
Box 5 – Regression analysis: predictors of not receiving intensive lipid‐lowering therapy at 6 or 12 months after index admission for treatment of acute coronary syndrome
|
Parameter |
Odds ratio (95% CI) |
P |
|||||||||||||
|
|
|||||||||||||||
|
Sex |
|||||||||||||||
|
Men |
1 |
— |
|||||||||||||
|
Women |
1.20 (1.02–1.41) |
0.037 |
|||||||||||||
|
Age (years) |
|
0.10 |
|||||||||||||
|
< 55 |
0.86 (0.66–1.12) |
|
|||||||||||||
|
55 to < 65 |
0.88 (0.66–1.18) |
|
|||||||||||||
|
65 to < 74 |
0.73 (0.54–0.99) |
|
|||||||||||||
|
> 74 |
1 |
|
|||||||||||||
|
In‐hospital treatment |
|
0.016 |
|||||||||||||
|
Percutaneous coronary intervention |
1 |
|
|||||||||||||
|
Coronary artery bypass grafting |
1.55 (1.26–1.92) |
|
|||||||||||||
|
Medically managed |
1.54 (1.25–1.91) |
|
|||||||||||||
|
Intensive lipid‐lowering therapy prescribed during admission |
|||||||||||||||
|
Yes |
1 |
— |
|||||||||||||
|
No |
1.53 (1.26–1.85) |
0.026 |
|||||||||||||
|
Intensive lipid‐lowering therapy prescribed at discharge |
|||||||||||||||
|
Yes |
1 |
— |
|||||||||||||
|
No |
7.24 (4.37–12.0) |
< 0.001 |
|||||||||||||
|
Referred to cardiac rehabilitation program |
|||||||||||||||
|
Yes |
1 |
— |
|||||||||||||
|
No |
1.39 (1.09–1.78) |
0.009 |
|||||||||||||
|
|
|||||||||||||||
|
CI = confidence interval. |
|||||||||||||||
Box 6 – Caterpillar plot of hospital residuals (with 95% confidence intervals) for log‐odds of not being prescribed intensive lipid‐lowering therapy at most recent follow‐up (6 or 12 months) after index admission for treatment of acute coronary syndrome
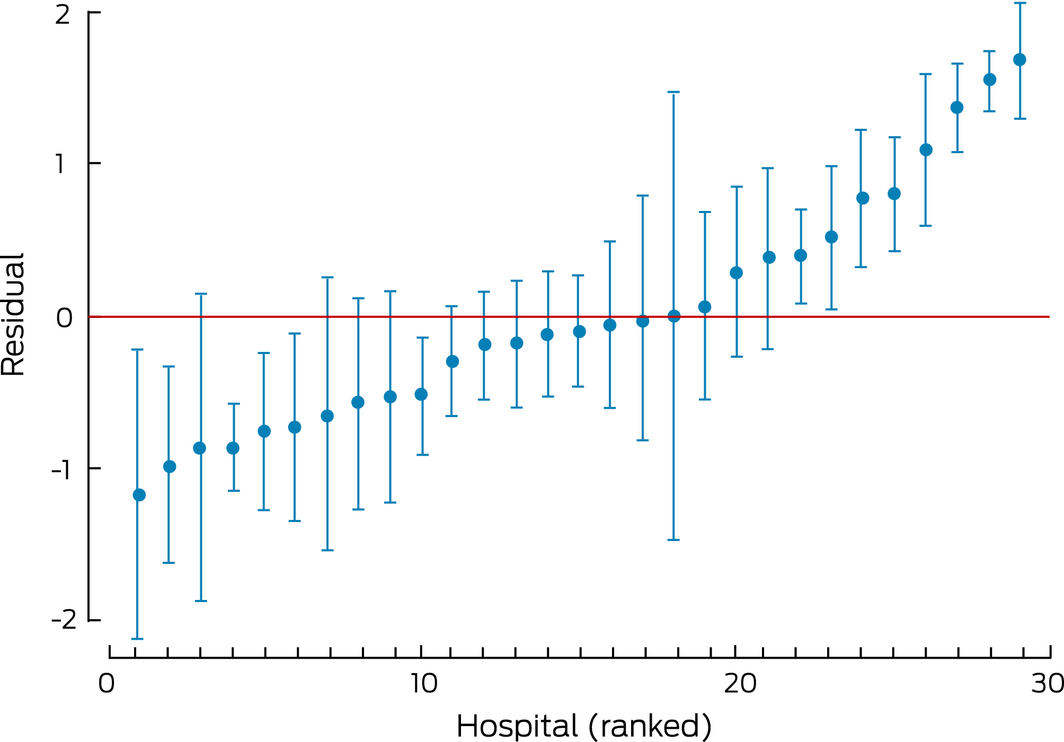
This plot shows the centre effect for all hospitals in the sample. If the confidence interval does not cross the residual line at zero, the odds of not receiving intensive lipid‐lowering therapy at the latest follow‐up is significantly higher or lower than the average for all hospitals.
Received 23 May 2018, accepted 28 September 2018
- David Brieger1
- Mario D'Souza2,3
- Karice Hyun4
- James C Weaver5
- Leonard Kritharides1,4
- 1 Concord Repatriation General Hospital, Sydney, NSW
- 2 University of Sydney School of Public Health, Sydney, NSW
- 3 Clinical Research Centre, Sydney Local Health District, Sydney, NSW
- 4 ANZAC Research Institute, Sydney, NSW
- 5 St George Hospital, Sydney, NSW
We thank Hazel Palmer (Scriptix) for her editorial assistance, which was funded by Sanofi Australia.
CONCORDANCE received funding from Sanofi Australia to support collection of follow‐up lipid data from patients enrolled during 2015–2016.
- 1. Australian Institute of Health and Welfare. Monitoring acute coronary syndrome using national hospital data: an information paper on trends and issues (Cat. no. CVD 57). Canberra: AIHW, 2011.
- 2. Deloitte Access Economics. ACS in perspective: the importance of secondary prevention. Nov 2011. https://www2.deloitte.com/content/dam/Deloitte/au/Documents/Economics/deloitte-au-economics-acs-perspective-importance-secondary-prevention-011111.pdf (viewed Sept 2018).
- 3. Hirsh BJ, Smilowitz NR, Rosenson RS, et al. Utilization of and adherence to guideline‐recommended lipid‐lowering therapy after acute coronary syndrome: opportunities for Improvement. J Am Coll Cardiol 2015; 66: 184–192.
- 4. Schwartz GG, Olsson AG, Ezekowitz MD, et al. Effects of atorvastatin on early recurrent ischemic events in acute coronary syndromes: the MIRACL study: a randomized controlled trial. JAMA 2001; 285: 1711–1718.
- 5. Lloyd‐Jones DM, Morris PB, Ballantyne CM, et al. 2017 Focused update of the 2016 ACC expert consensus decision pathway on the role of non‐statin therapies for LDL‐cholesterol lowering in the management of atherosclerotic cardiovascular disease risk: a report of the American College of Cardiology Task Force on Expert Consensus Decision Pathways. J Am Coll Cardiol 2017; 70: 1785–1822.
- 6. Chew DP, Scott IA, Cullen L, et al. National Heart Foundation of Australia & Cardiac Society of Australia and New Zealand: Australian clinical guidelines for the management of acute coronary syndromes 2016. Heart Lung Circ 2016; 25: 895–951.
- 7. Cannon CP, Blazing MA, Giugliano RP, et al. Ezetimibe added to statin therapy after acute coronary syndromes. N Engl J Med 2015; 372: 2387–2397.
- 8. De Vera MA, Bhole V, Burns LC, Lacaille D. Impact of statin adherence on cardiovascular disease and mortality outcomes: a systematic review. Br J Clin Pharmacol 2014; 78: 684–698.
- 9. Lauffenburger JC, Robinson JG, Oramasionwu C, Fang G. Racial/ethnic and gender gaps in the use of and adherence to evidence‐based preventive therapies among elderly Medicare Part D beneficiaries after acute myocardial infarction. Circulation 2014; 129: 754–763.
- 10. Muntner P, Yun H, Sharma P, et al. Ability of low antihypertensive medication adherence to predict statin discontinuation and low statin adherence in patients initiating treatment after a coronary event. Am J Cardiol 2014; 114: 826–831.
- 11. Aliprandi‐Costa B, Ranasinghe I, Turnbull F, et al. The design and rationale of the Australian Cooperative National Registry of Acute Coronary care, Guideline Adherence and Clinical Events (CONCORDANCE). Heart Lung Circ 2013; 22: 533–541.
- 12. Stone NJ, Robinson JG, Lichtenstein AH, et al. 2013 ACC/AHA guideline on the treatment of blood cholesterol to reduce atherosclerotic cardiovascular risk in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 2014; 63: 2889–2934.
- 13. Liang KY, Zeger SL. Regression analysis for correlated data. Annu Rev Public Health 1993; 14: 43–68.
- 14. Australian Bureau of Statistics. 2033.0.55.001. Census of population and housing: Socio‐Economic Indexes for Areas (SEIFA), Australia, 2011. IRSD. Mar 2013. http://www.abs.gov.au/ausstats/abs@.nsf/Lookup/by%20Subject/2033.0.55.001~2011~Main%20Features~IRSD~10005 (viewed Sept 2018).
- 15. Rosenson RS, Kent ST, Brown TM, et al. Underutilization of high‐intensity statin therapy after hospitalization for coronary heart disease. J Am Coll Cardiol 2015; 65: 270–277.
- 16. Foody JM, Joyce AT, Rudolph AE, et al. Persistence of atorvastatin and simvastatin among patients with and without prior cardiovascular diseases: a US managed care study. Curr Med Res Opin 2008; 24: 1987–2000.
- 17. Hiratzka LF, Eagle KA, Liang L, et al. Atherosclerosis secondary prevention performance measures after coronary bypass graft surgery compared with percutaneous catheter intervention and nonintervention patients in the Get With the Guidelines database. Circulation 2007; 116(11 Suppl): I207–I212.
- 18. Stroes ES, Thompson PD, Corsini A, et al. Statin‐associated muscle symptoms: impact on statin therapy. European Atherosclerosis Society consensus panel statement on assessment, aetiology and management. Eur Heart J 2015; 36: 1012–1022.
- 19. Cannon CP, Khan I, Klimchak AC, et al. Simulation of lipid‐lowering therapy intensification in a population with atherosclerotic cardiovascular disease. JAMA Cardiol 2017; 2: 959–966.
- 20. Krahenbuhl S, Pavik‐Mezzour I, von Eckardstein A. Unmet needs in LDL‐C lowering: when statins won't do! Drugs 2016; 76: 1175–1190.
- 21. Ho PM, Bryson CL, Rumsfeld JS. Medication adherence: its importance in cardiovascular outcomes. Circulation 2009; 119: 3028–3035.





Abstract
Objective: To determine the prevalence and identify predictors of people hospitalised with acute coronary syndrome (ACS) receiving intensive lipid‐lowering therapy during the 12 months after their discharge from hospital.
Design: Retrospective observational analysis.
Setting: Data were extracted from CONCORDANCE, a prospective, Australian investigator‐initiated ACS registry.
Participants: Patients enrolled in CONCORDANCE during January 2015 – May 2016 who survived to hospital discharge, for whom information on lipid‐lowering therapy 6 or 12 months after discharge from hospital were available.
Main outcome measures: Not receiving intensive lipid‐lowering therapy (with or without ezetimibe) at the most recent follow‐up (6 or 12 months); predictors of not receiving intensive lipid‐lowering therapy.
Results: 1876 of 3441 patients (55%) were receiving intensive lipid‐lowering therapy 6 or 12 months after their hospitalisation with an ACS. Predictors of not receiving intensive lipid‐lowering therapy included not been prescribed this treatment prior to their hospital admission (odds ratio [OR], 1.53; 95% CI, 1.26–1.85) or at hospital discharge (aOR, 7.24; 95% CI, 4.37–12.0), being a woman (aOR, 1.20; 95% CI, 1.02–1.41), and not being referred for cardiac rehabilitation (aOR 1.39; 95% CI, 1.09–1.78). Patients who were managed medically in hospital (not revascularised; aOR, 1.54; 95% CI, 1.25–1.91) or underwent coronary artery bypass grafting (aOR 1.55; 95% CI, 1.26–1.92) were less likely to be receiving intensive lipid‐lowering therapy at follow‐up than those with a percutaneous coronary intervention. Unmeasured hospital factors accounted for 17% of the variation in the likelihood of intensive lipid‐lowering therapy.
Conclusions: 45% of patients in Australia are not receiving intensive lipid‐lowering therapy in the 12 months after their ACS. Optimising oral lipid‐lowering therapy would reduce the recurrence of coronary events in this high risk group.