The known: Drug‐induced liver injury (DILI) is the most frequent cause of acute liver failure in Western countries. The proportion of cases caused by herbal and dietary supplements has increased overseas.
The new: The proportion of patients admitted to our Sydney quaternary referral centre with non‐paracetamol DILI caused by herbal and dietary supplements has increased since 2009. They had poorer outcomes than patients with DILI caused by paracetamol and prescription medicines. Almost half the patients with supplement‐related DILI had non‐European ethnic backgrounds.
The implications: Rigorous regulatory oversight of herbal and dietary supplements and improved, culturally appropriate community education about their risks are needed in Australia.
Among the increasing range of prescription and non‐prescription pharmacological agents are some that can cause drug‐induced liver injury (DILI) through direct or indirect hepatotoxicity or as the result of an idiosyncratic reaction.1 DILI is responsible for 10% of hospitalisations following abnormal liver function test results2 and for 3–5% of hospitalisations with jaundice.3 It is a leading cause of liver‐related death, and the most common cause of acute liver failure in Western countries.1
Although medication packaging (eg, stronger, standardised warnings in package inserts), design (informed by pre‐clinical toxicity screening models), and safety monitoring have improved,1 paracetamol‐related and non‐paracetamol DILI remain important clinical problems, as their incidence has not declined.4 A recent analysis found that hospitalisations for paracetamol‐related DILI in Australia increased by 108% during 2004–2017.5 There are DILI registries in the United States, Europe, and Asia, but the epidemiology and prognosis of non‐paracetamol DILI have not been investigated in detail in Australia.
DILI caused by herbal and dietary supplements is a growing problem. In the United States, the proportion of supplement‐related DILI cases doubled from 7–9% in 2004–2007 to 19–20% in 2010–2014.6,7 Although cases of severe DILI linked with herbal and dietary supplements are occasionally reported in Australia,8,9 the problem has not been specifically studied here.
We therefore investigated the epidemiology of patients attending our liver transplantation centre who were hospitalised with DILI during 2009–2020. We focused on non‐paracetamol‐related cases, but also compared the clinical features and outcomes of patients with paracetamol‐related or non‐paracetamol DILI.
Methods
We undertook a retrospective analysis of data for all adults (18 years or older) admitted with DILI to the A.W. Morrow Gastroenterology and Liver Centre, Royal Prince Alfred Hospital, Sydney, during 1 January 2009 – 31 August 2020. Cases of DILI were identified by the International Classification of Diseases, tenth revision (ICD‐10) code for “toxic liver disease” (K71) and by searching our liver transplant database for patients who underwent transplantation following acute liver failure secondary to DILI. Patients were excluded if liver injury was not drug‐induced or if they developed DILI during hospital admission for other reasons, as were patients with paracetamol poisoning (ICD‐10 code T39.1) without “toxic liver disease”. All cases of DILI were independently confirmed in patient medical records by two investigators.
Clinical data
Patient data, including baseline laboratory values on admission, were extracted from electronic medical records. Acute liver failure was defined as the presence of hepatic encephalopathy and an international normalised ratio (INR) of more than 1.5 within 26 weeks of symptom onset, in the absence of chronic liver disease. We assessed cases with the following scores and criteria:
- The Roussel–Uclaf causality assessment method (RUCAM) evaluates the likelihood that a medication has caused DILI (highly probable, probable, possible, unlikely, excluded).10
- The model for end‐stage liver disease (MELD) score, calculated from serum creatinine, bilirubin, and INR values, is a validated predictor of survival in patients with cirrhosis or liver failure.11
- The R value — the ratio of alanine aminotransferase to alkaline phosphatase activity — identifies whether a liver function test derangement is hepatocellular, cholestatic, or of mixed pattern in nature.12
- Hy’s law states that patients presenting with hepatocellular DILI and jaundice are at high risk of death or of need for a liver transplant.13
- The King’s College criteria are clinical and laboratory features that identify patients with acute liver failure with poor prognoses and who may therefore require a liver transplant.14
Statistical analysis
Statistical analyses were performed in SPSS 22.0 (IBM). Continuous variables were summarised as means with standard deviations (SDs) or medians with interquartile ranges (IQRs). The statistical significance of differences between groups was assessed in χ2 or Fisher exact tests (categorical parameters) and Student t or Mann–Whitney tests (continuous parameters). The primary outcome was 90‐day transplant‐free survival (time to death or transplantation); factors associated with the primary outcome were assessed in a multivariable Cox regression model. Potential variables were selected on the basis of information in published literature and clinical knowledge, followed by application of the least absolute shrinkage and selection operator (LASSO) method; the tuning parameter was selected to minimise the mean square error in tenfold cross‐validation. Selection‐adjusted confidence intervals (CIs) and P values for the LASSO estimate using the chosen tuning parameter value were estimated. Multicollinearity between covariates was assessed with a variance inflation factor; values exceeding 5 were deemed significant. Cumulative probability of study endpoints was estimated in Kaplan–Meier analyses. Time‐to‐event curves for groups were compared in log‐rank tests.
Ethics approval
The study was conducted according to the Declaration of Helsinki, and was approved by the Sydney Local Health District Human Research Ethics Committee, which waived the requirement for informed consent by patients (X20‐0108 and 2020/ETH00726).
Results
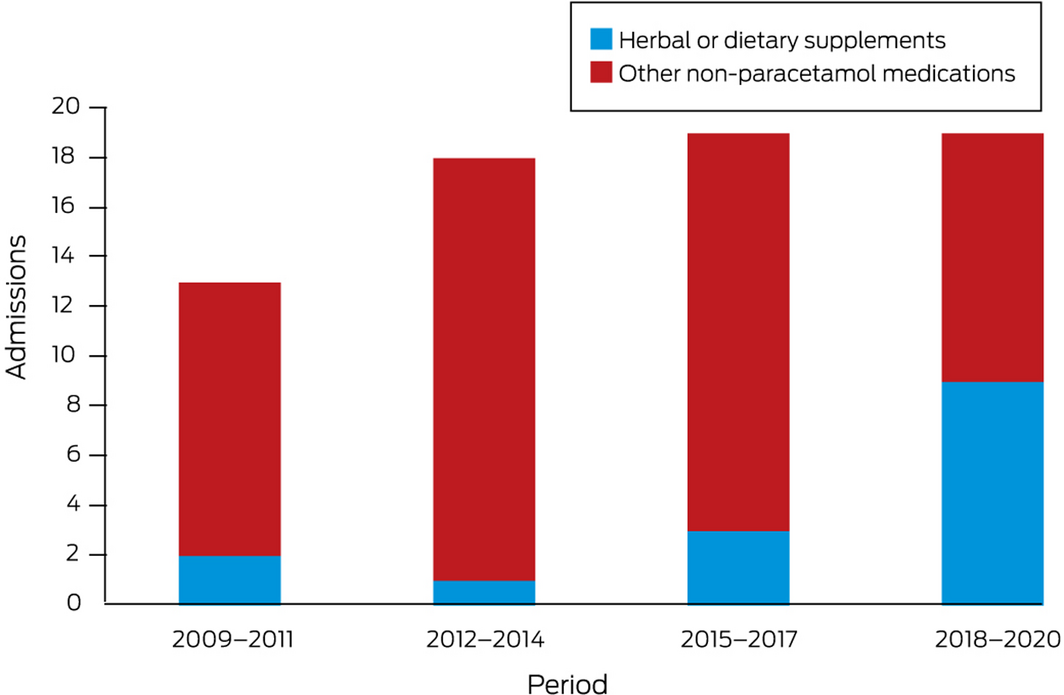
During January 2009 – August 2020, 184 of 294 admitted patients fulfilled study inclusion criteria: 69 with non‐paracetamol and 115 with paracetamol‐related DILI (online Supporting Information). The numbers of non‐paracetamol DILI admissions were similar across the study period, but the proportion linked with herbal and dietary supplements increased from 2 of 13 (15%) during 2009–11 to 9 of 19 (47%) during 2018–20 (linear trend: P = 0.011) (Box 1).
The median age of patients with non‐paracetamol DILI was higher than for those with paracetamol‐related DILI, and smaller proportions were women or had histories of psychiatric disorders or significant alcohol use. Larger proportions of patients with non‐paracetamol DILI presented with jaundice, fever, or rash, or with mixed or cholestatic liver function test derangements. Larger proportions of patients with paracetamol‐related DILI presented with hepatocellular derangement, and their median creatinine and INR values were higher than for patients with non‐paracetamol DILI. The proportions of patients with acute liver failure, or with disease fulfilling King’s College criteria or Hy’s law were similar for the two groups; the median baseline MELD score was higher for people with paracetamol‐related DILI (27.2; IQR, 17.1–34.6) than for those with non‐paracetamol DILI (20.5; IQR, 14.8–30.8) (Box 2).
Among patients with non‐paracetamol DILI, smaller proportions of supplement‐related cases involved people of European ethnic background (8 of 15, 53% v 46 of 54, 85%) or cholestatic liver function test patterns (one of 15, 7% v 18 of 54, 33%) than of cases not linked with supplements; larger proportions fulfilled Hy’s law criteria (14 of 15, 93% v 20 of 54, 37%) (Box 3).
Medication history and treatment
Among the 69 patients with non‐paracetamol‐related DILI, the most frequently implicated medications were antibiotics and antifungal medications (19, 28%), herbal or dietary supplements (15, 22%), anti‐tuberculosis medications (six, 9%), and anti‐cancer medications (five, 7%) (Box 4). Most of these medications are listed in the LiverTox database15 as causing DILI. The implicated medication was initiated a median of 57 days (IQR, 25–84 days) before admission for a median stay of 38.5 days (IQR, 19–76 days). The likelihood of the implicated medication having caused DILI was rated by RUCAM as possible in eleven cases (16%), probable in 57 (83%), and highly probable in one case (1%). Liver biopsy was performed for 36 patients (52%); all had histopathology consistent with DILI. As treatment, 24 patients received corticosteroids (35%) and 12 ursodeoxycholic acid (17%).
Sixty‐two of the 112 cases of paracetamol‐related DILI for which the relevant information was available (55%) were intentional single overdoses (median, 21.5 g; IQR, 13–36 g) and 50 were staggered ingestions (median, 5.5 g per day; IQR, 4–9 g per day). Thirty‐eight of 113 patients had co‐ingested other medications (34%), including 14 who had taken codeine and 12 who had taken non‐steroidal anti‐inflammatory drugs. The likelihood of paracetamol causing DILI was rated as possible for one patient (1%), probable for 46 (43%), and highly probable for 60 patients (56%; RUCAM not available: eight patients). Intravenous N‐acetylcysteine was administered to 113 patients (98%) a median of 16 hours (IQR, 6–39 hours) after paracetamol ingestion.
Admission to intensive care with complications of acute liver failure (84 of 155, 73% v 24 of 69, 35%) and haemodialysis (56 of 115, 49% v 8 of 69, 11%) were each more frequent for patients with paracetamol‐related DILI than for those with non‐paracetamol DILI. Within the latter group, the rates were similar for cases in which herbal or dietary supplements were or were not implicated (data not shown).
Outcomes
During the 90‐day follow‐up, 12 patients with paracetamol‐related DILI died and three received liver transplants; seven patients with non‐paracetamol‐related DILI died and 12 received transplants. Ninety‐day transplant‐free survival was 86% (95% CI, 79–93%) for patients with paracetamol‐related DILI, and 71% (95% CI, 60–82%) for patients with non‐paracetamol DILI (Box 5, A). In both patient groups, survival was greater for patients with low MELD scores than for those with scores above 20 (Box 5, B). Finally, 90‐day transplant‐free survival was 74% (95% CI, 62–87%) for patients with DILI attributed to non‐paracetamol medications other than herbal and dietary supplements, and 59% (95% CI, 34–85%) for patients in which DILI was attributed to supplements; only the difference between the paracetamol‐related and supplement‐related cases was statistically significant (Box 5, C). Of the 19 patients who died without receiving liver transplants, one died intra‐operatively during an attempted transplant, four died after or during preparation for transplantation, and 14 were deemed medically or socially unsuitable for transplantation.
Significant multicollinearity between potential predictors of the primary outcome was not evident. Ninety‐day transplant‐free survival for patients with non‐paracetamol DILI was influenced by admission MELD score (mortality risk, per point increase: adjusted hazard ratio [aHR], 1.24; 95% CI, 1.14–1.36), and that of patients with paracetamol‐related DILI by admission sodium level (per 1.0 mmol/L increase: aHR, 1.26; 95% CI, 1.05–1.54) and admission MELD score (aHR, per point increase: 1.19; 95% CI, 1.09–3.74) (Box 6).
Discussion
DILI is a leading cause of acute liver failure and liver‐related death, particularly in Western countries. Data on its epidemiology in Australia are limited, especially on cases caused by drugs other than paracetamol. Our study is the first to specifically investigate cases of non‐paracetamol DILI causing hospitalisation in Australia over a period of several years.
Patients with paracetamol‐related and non‐paracetamol DILI comprise two distinct clinical groups. Despite higher median MELD scores and more frequent need for intensive care and dialysis, 90‐day transplant‐free survival for patients with paracetamol DILI was higher than for those with non‐paracetamol DILI. Indeed, the prognosis for patients with paracetamol‐related DILI and acute liver failure is good, as these patients are generally younger and have fewer other medical conditions, and an antidote for paracetamol poisoning is available.16 Higher admission MELD scores were independently associated with poorer survival for patients with paracetamol‐related or non‐paracetamol DILI. Although the MELD score was primarily derived and validated to predict survival for patients with cirrhosis, its prognostic value in acute liver failure has been described, especially in non‐paracetamol cases.17
In our study, the proportion of DILI cases linked with herbal and dietary supplements increased over time. The overall proportion of non‐paracetamol DILI cases attributed to supplements (22%) was similar to that reported in the United States (20% in 2013)7 and Iceland18 (2010–2011: 16%). As in other Western countries, bodybuilding and weight loss supplements were implicated in half of these cases, but the number in which traditional Chinese medicines were implicated was higher than in other reports,6,19 perhaps unsurprising in light of the strong demand for traditional Chinese medicines in Australia.20 Most patients with supplement‐related DILI had hepatocellular patterns of liver injury, as previously reported for similar cases.21,22 Such patients have a poorer prognosis than people with DILI caused by conventional medicines, and they have higher rates of liver transplantation.1,7,23 A larger proportion of patients with supplement‐related DILI in our study met Hy’s law criteria (93%) than of patients with DILI of other aetiology, reflecting the greater severity of liver injury. However, we found no significant difference in transplant‐free survival between cases in which supplements were implicated and those involving other non‐paracetamol medications, perhaps because the numbers of patients were relatively small. Further, we found no difference in survival between cases of paracetamol‐related DILI and those of non‐paracetamol DILI in which herbal and dietary supplements were not implicated.
Our findings have several important clinical implications. First, paracetamol‐related DILI remains a problem and public health measures, including further reductions of pack sizes, should be considered.5
Second, the rise in the proportion of non‐paracetamol DILI cases in which herbal and dietary supplements were implicated reflects the rise in supplement use in Australia over the past two decades. During 1993–2007, the prevalence of complementary medicines use increased from 49% to 70%; many people use them without knowing the evidence (or lack thereof) for their therapeutic claims.20 The lack of regulatory oversight in the preparation and marketing of herbal and dietary supplements has attracted criticism.1,24 In Australia, the Therapeutic Goods Administration (TGA) classifies them as “listed” (more than 12 000 lower risk products) or “registered” (about 200 higher risk products).20 However, overseas herbal and dietary supplements purchased online evade Australian regulatory oversight. While the TGA assesses the safety and quality of the ingredients of all supplements, only high risk medicines are assessed for efficacy. The post‐marketing surveillance of adverse reactions to medicines required by the TGA is critical to maintaining safe use. However, reporting adverse reactions is voluntary, relying on the diligence of physicians and pharmacists.25 Although the regulation of herbal and dietary supplements has improved, it remains imperfect.
Third, community education is important. Australia has a culturally diverse population, and this is reflected by its use of herbal and dietary supplements.20 About half of the patients in our study with supplement‐related DILI had non‐European ethnic backgrounds, compared with fewer than 20% of patients with non‐paracetamol DILI caused by other medications. Rates of herbal and dietary supplement use are higher in Asia, Africa, and Latin America (as high as 80%) than in Australia, the United States, or Europe,22 but cases of acute liver failure secondary to DILI are less frequent in these areas than those caused by acute viral hepatitis (types A, B, E). Migrants transport their cultural beliefs and traditions about supplement use to their new homes, contributing to the their rising use in Western countries.20,22 Culturally and linguistically diverse communities should therefore be targeted for appropriate education about the potential dangers of herbal and dietary supplements.
Limitations
First, the retrospective nature of our study means our conclusions rely on the accuracy and completeness of the data we analysed. However, RUCAM assessments indicated at least “probable” causality for the drugs implicated in 84% of cases of non‐paracetamol DILI without re‐challenging. Our study design was the most feasible for assessing DILI patterns over a 12‐year period. Further, all collected data were independently verified by two investigators, and hard endpoints were chosen as study outcomes to minimise subjectivity. Second, we examined data for patients hospitalised with DILI at a single quaternary referral centre, skewing our sample toward more severe DILI, and our findings may not be generalisable to the community or to non‐transplantation medical centres. Nonetheless, changes over time were similar to those reported by national DILI registries overseas.5,6,7,19 Finally, our patient sample was relatively small, limiting our ability to detect statistically significant differences. Larger DILI databases are needed. The Australian Drug Induced Liver Injury Network (AusDILIN) was established in 2020, and currently links 16 tertiary referral hospitals across Australia; it facilitates the prospective entry of non‐paracetamol DILI cases into a database for research purposes and to provide regular feedback on their incidence and prevalence.
Conclusion
The number of people hospitalised in our Sydney liver transplant referral centre with DILI caused by herbal and dietary supplements increased during 2009–2020. Ninety‐day transplant‐free survival for patients with non‐paracetamol DILI, especially cases caused by supplements, was poorer than for people hospitalised with paracetamol‐related DILI. Further, survival was poorer for patients with higher MELD scores, irrespective of the medication implicated. DILI remains an important clinical problem that requires more investigation, regulatory changes for the substances involved, and patient and community education.
Box 1 – Numbers of admissions with non‐paracetamol‐related drug‐induced liver injury to the A.W. Morrow Gastroenterology and Liver Centre, Royal Prince Alfred Hospital, January 2009 – August 2020
Box 2 – Baseline characteristics of patients with paracetamol or non‐paracetamol‐related drug‐induced liver injury
|
|
Patients with drug‐induced liver injury |
|
|||||||||||||
|
Characteristic |
All |
Paracetamol‐related |
Non‐paracetamol |
P |
|||||||||||
|
|
|||||||||||||||
|
Patients |
184 |
115 |
69 |
|
|||||||||||
|
Sex (women) |
126 (69%) |
88 (77%) |
38 (55%) |
0.002 |
|||||||||||
|
Age (years), median (IQR) |
42.5 (29–56) |
36 (27–52) |
52 (37–63) |
< 0.001 |
|||||||||||
|
Ethnic background |
|
|
|
|
|||||||||||
|
European |
148 (80%) |
94 (82%) |
54 (78%) |
0.57 |
|||||||||||
|
Asian |
21 (11%) |
9 (8%) |
12 (17%) |
0.048 |
|||||||||||
|
Other |
15 (8%) |
12 (11%) |
3 (4%) |
0.14 |
|||||||||||
|
Body mass index (kg/m2), median (IQR) |
24.5 (22.1–28.8) |
24.5 (22.2–28.9) |
24.7 (22.1–28.3) |
0.95 |
|||||||||||
|
History of chronic liver disease |
25 (14%) |
11 (10%) |
14 (20%) |
0.05 |
|||||||||||
|
History of psychiatric disorder |
75 (43%) |
57 (53%) |
18 (26%) |
< 0.001 |
|||||||||||
|
History of significant alcohol use* |
63 (35%) |
52 (47%) |
11 (16%) |
< 0.001 |
|||||||||||
|
Source of admission |
|
|
|
|
|||||||||||
|
From home |
57 (31%) |
34 (30%) |
23 (33%) |
0.62 |
|||||||||||
|
From general practitioner |
11 (6%) |
4 (4%) |
7 (10%) |
0.07 |
|||||||||||
|
From gastroenterology clinic |
10 (6%) |
0 |
10 (15%) |
< 0.001 |
|||||||||||
|
Inter‐hospital transfer |
105 (57%) |
76 (67%) |
29 (42%) |
0.001 |
|||||||||||
|
Clinical presentation |
|
|
|
|
|||||||||||
|
Symptomatic |
163 (90%) |
99 (88%) |
64 (93%) |
0.27 |
|||||||||||
|
Fever |
19 (11%) |
7 (7%) |
12 (17%) |
0.026 |
|||||||||||
|
Jaundice |
56 (32%) |
8 (8%) |
48 (70%) |
< 0.001 |
|||||||||||
|
Rash |
10 (6%) |
0 |
10 (15%) |
< 0.001 |
|||||||||||
|
Encephalopathy |
59 (33%) |
37 (33%) |
22 (32%) |
0.87 |
|||||||||||
|
Drug‐induced liver injury pattern (R value) |
|
|
|
|
|||||||||||
|
Hepatocellular |
143 (78%) |
105 (91%) |
38 (55%) |
< 0.001 |
|||||||||||
|
Cholestatic |
24 (13%) |
5 (4%) |
19 (28%) |
< 0.001 |
|||||||||||
|
Mixed |
17 (9%) |
5 (4%) |
12 (17%) |
0.003 |
|||||||||||
|
Baseline serum laboratory values, median (IQR) |
|
|
|
|
|||||||||||
|
Eosinophils (× 109/L; RI, 0.0–0.5 × 109/L) |
0.0 (0.0–0.1) |
0.0 (0.0–0.1) |
0.1 (0.0–0.2) |
< 0.001 |
|||||||||||
|
Sodium (mmol/L; RI, 135–145mmol/L) |
137 (135–140) |
137 (135–140) |
137 (135–140) |
0.96 |
|||||||||||
|
Creatinine (μmol/L; RI, 45–90 µmol/L [women], 60–110 µmol/L [men]) |
82 (62–135) |
93 (67–173) |
68 (51–99) |
< 0.001 |
|||||||||||
|
Albumin (g/L; RI, 33–48 g/L) |
35 (30–40) |
34 (30–41) |
35 (28–40) |
0.57 |
|||||||||||
|
Bilirubin (μmol /L; RI, < 20µmol/L) |
59 (30–132) |
51 (26–82) |
164 (58–379) |
< 0.001 |
|||||||||||
|
Alkaline phosphatase (U/L; RI, 30–110 U/L) |
124 (96–202) |
108 (79–136) |
208 (141–414) |
< 0.001 |
|||||||||||
|
‐Glutamyltransferase (U/L; RI, 5–35 U/L) |
161 (79–336) |
136 (76–281) |
211 (121–421) |
0.005 |
|||||||||||
|
Alanine aminotransferase (U/L; RI, 10–35 U/L) |
2618 (536–4947) |
3807 (1704–7372) |
819 (209–2665) |
< 0.001 |
|||||||||||
|
Aspartate aminotransferase (U/L; RI, 10–35 U/L) |
2165 (463–7779) |
4934 (1557–10 100) |
510 (122–1675) |
< 0.001 |
|||||||||||
|
International normalised ratio (RI, 0.9–1.2) |
2.2 (1.4–4.4) |
3.0 (1.7–5.2) |
1.4 (1.1–2.7) |
< 0.001 |
|||||||||||
|
Baseline MELD score, median (IQR) |
24.2 (15.7–33.6) |
27.2 (17.1–34.6) |
20.5 (14.8–30.8) |
0.026 |
|||||||||||
|
Acute liver failure |
54 (29%) |
36 (31%) |
18 (26%) |
0.44 |
|||||||||||
|
Fulfilled King’s College criteria for liver transplantation |
45 (83%) |
31 (86%) |
14 (78%) |
0.44 |
|||||||||||
|
Fulfilled Hy’s law criteria |
99 (54%) |
65 (57%) |
34 (49%) |
0.34 |
|||||||||||
|
|
|||||||||||||||
|
IQR = interquartile range; MELD = model for end‐stage liver disease; RI = reference interval. * More than 14 standard drinks (140 g alcohol) per week. |
|||||||||||||||
Box 3 – Baseline characteristics of patients with non‐paracetamol‐related drug‐induced liver injury, by herbal and dietary supplement involvement
|
|
Patients with non‐paracetamol‐related drug‐induced liver injury |
|
|||||||||||||
|
Characteristic |
All |
Herbal or dietary supplement‐related |
Not herbal or dietary supplement‐related |
P |
|||||||||||
|
|
|||||||||||||||
|
Patients |
69 |
15 |
54 |
|
|||||||||||
|
Sex (women) |
38 (55%) |
6 (40%) |
32 (59%) |
0.19 |
|||||||||||
|
Age (years), median (IQR) |
52 (37–63) |
52 (27–58) |
52 (38–63) |
0.32 |
|||||||||||
|
Ethnic background |
|
|
|
|
|||||||||||
|
European |
54 (78%) |
8 (53%) |
46 (85%) |
0.008 |
|||||||||||
|
Asian |
12 (17%) |
5 (33%) |
7 (13%) |
0.07 |
|||||||||||
|
Other |
3 (4%) |
2 (13%) |
1 (2%) |
0.05 |
|||||||||||
|
Body mass index (kg/m2), median (IQR) |
24.7 (22.1–28.3) |
25.3 (23.8–29.4) |
24.3 (21.6–27.0) |
0.26 |
|||||||||||
|
History of chronic liver disease |
14 (20%) |
3 (20%) |
11 (20%) |
0.98 |
|||||||||||
|
History of psychiatric disease |
18 (26%) |
3 (20%) |
15 (28%) |
0.54 |
|||||||||||
|
History of significant alcohol use* |
11 (16%) |
4 (27%) |
7 (13%) |
0.20 |
|||||||||||
|
Source of admission |
|
|
|
|
|||||||||||
|
From home |
23 (33%) |
2 (13%) |
21 (39%) |
0.06 |
|||||||||||
|
From general practitioner |
7 (10%) |
3 (20%) |
4 (7%) |
0.15 |
|||||||||||
|
From gastroenterology clinic |
10 (15%) |
3 (20%) |
7 (13%) |
0.49 |
|||||||||||
|
Inter‐hospital transfer |
29 (42%) |
7 (47%) |
22 (41%) |
0.68 |
|||||||||||
|
Clinical presentation |
|
|
|
|
|||||||||||
|
Symptomatic |
64 (93%) |
14 (93%) |
50 (93%) |
0.92 |
|||||||||||
|
Fever |
12 (17%) |
1 (7%) |
11 (20%) |
0.22 |
|||||||||||
|
Jaundice |
48 (70%) |
13 (87%) |
35 (65%) |
0.10 |
|||||||||||
|
Rash |
10 (15%) |
0 |
10 (19%) |
0.07 |
|||||||||||
|
Encephalopathy |
22 (32%) |
2 (13%) |
20 (37%) |
0.08 |
|||||||||||
|
Drug‐induced liver injury pattern (R value) |
|
|
|
|
|||||||||||
|
Hepatocellular |
38 (55%) |
10 (67%) |
28 (52%) |
0.31 |
|||||||||||
|
Cholestatic |
19 (28%) |
1 (7%) |
18 (33%) |
0.041 |
|||||||||||
|
Mixed |
12 (17%) |
4 (27%) |
8 (15%) |
0.28 |
|||||||||||
|
Baseline serum laboratory values, median (IQR) |
|
|
|
|
|||||||||||
|
Eosinophils (× 109/L; RI, 0.0–0.5 × 109/L) |
(0.0–0.2) |
0.1 (0.1–0.2) |
0.1 (0.0–0.2) |
0.48 |
|||||||||||
|
Sodium (mmol/L; RI, 135–145mmol/L) |
137 (135–140) |
137 (136–139) |
137 (135–140) |
0.88 |
|||||||||||
|
Creatinine (μmol /L; RI, 45–90 µmol/L [women], 60–110 µmol/L [men]) |
68 (51–99) |
69 (62–102) |
66 (48–99) |
0.62 |
|||||||||||
|
Albumin (g/L; RI, 33–48 g/L) |
35 (28–40) |
36 (28–42) |
35 (28–39) |
0.60 |
|||||||||||
|
Bilirubin (μmol /L; RI, < 20µmol/L) |
164 (58–379) |
344 (164–461) |
122 (36–293) |
0.009 |
|||||||||||
|
Alkaline phosphatase (U/L; RI, 30–110 U/L) |
208 (141–414) |
168 (110–213) |
227 (157–462) |
0.010 |
|||||||||||
|
‐Glutamyltransferase (U/L; RI, 5–35 U/L) |
211 (121–421) |
137 (89–212) |
272 (124–551) |
0.037 |
|||||||||||
|
Alanine aminotransferase (U/L; RI, 10–35 U/L) |
819 (209–2665) |
921 (229–1919) |
745 (190–2934) |
0.72 |
|||||||||||
|
Aspartate aminotransferase (U/L; RI, 10–35 U/L) |
510 (122–1675) |
673 (260–1065) |
480 (110–1781) |
0.83 |
|||||||||||
|
International normalised ratio (RI, 0.9–1.2) |
1.4 (1.1–2.7) |
1.6 (1.2–3.2) |
1.4 (1.1–2.2) |
0.29 |
|||||||||||
|
Baseline MELD score, median (IQR) |
20.5 (14.8–30.8) |
24.1 (16.5–32.6) |
20.4 (14.1–28.7) |
0.24 |
|||||||||||
|
Acute liver failure |
18 (26%) |
2 (13%) |
16 (30%) |
0.20 |
|||||||||||
|
Fulfilled King’s College criteria for liver transplantation |
14 (78%) |
2 (100%) |
12 (75%) |
0.42 |
|||||||||||
|
Fulfilled Hy’s law criteria |
34 (49%) |
14 (93%) |
20 (37%) |
< 0.001 |
|||||||||||
|
|
|||||||||||||||
|
IQR = interquartile range; MELD = model for end‐stage liver disease; RI = reference interval. * More than 14 standard drinks (140 g alcohol) per week. |
|||||||||||||||
Box 4 – Medications implicated in 69 cases of patients hospitalised with non‐paracetamol‐related drug‐induced liver injury
|
Medications, by class |
Number |
||||||||||||||
|
|
|||||||||||||||
|
Antibiotics, antifungals |
19 (28%) |
||||||||||||||
|
Dicloxacillin/flucloxacillin |
6 (9%) |
||||||||||||||
|
Amoxicillin (with or without clavulanic acid) |
5 (7%) |
||||||||||||||
|
Cephalexin |
2 (3%) |
||||||||||||||
|
Nitrofurantoin |
2 (3%) |
||||||||||||||
|
Metronidazole |
1 (1%) |
||||||||||||||
|
Erythromycin |
1 (1%) |
||||||||||||||
|
Voriconazole |
1 (1%) |
||||||||||||||
|
Clindamycin |
1 (1%) |
||||||||||||||
|
Herbal and dietary supplements |
15 (22%) |
||||||||||||||
|
Anabolic steroids/bodybuilding supplement |
5 (7%) |
||||||||||||||
|
Traditional Chinese medicine |
4 (6%) |
||||||||||||||
|
Weight loss supplement |
3 (4%) |
||||||||||||||
|
Kava |
1 (1%) |
||||||||||||||
|
Cassia cinnamon |
1 (1%) |
||||||||||||||
|
Curcumin |
1 (1%) |
||||||||||||||
|
Anti‐tuberculosis medications |
6 (9%) |
||||||||||||||
|
Pyrazinamide |
2 (3%) |
||||||||||||||
|
Isoniazid |
1 (1%) |
||||||||||||||
|
Rifampicin |
1 (1%) |
||||||||||||||
|
Pyrazinamide/isoniazid |
1 (1%) |
||||||||||||||
|
Rifampicin/isoniazid/pyrazinamide/ethambutol |
1 (1%) |
||||||||||||||
|
Anti‐cancer medications |
5 (7%) |
||||||||||||||
|
Immune checkpoint inhibitors |
4 (6%) |
||||||||||||||
|
Pazopanib |
1 (1%) |
||||||||||||||
|
Other medications |
24 (35%) |
||||||||||||||
|
Anti‐inflammatory drugs |
3 (4%) |
||||||||||||||
|
Disulfiram |
3 (4%) |
||||||||||||||
|
Statins |
2 (3%) |
||||||||||||||
|
Phenytoin |
2 (3%) |
||||||||||||||
|
Antidepressants |
2 (3%) |
||||||||||||||
|
Amphetamines |
2 (3%) |
||||||||||||||
|
Interferons |
2 (3%) |
||||||||||||||
|
Immunomodulators (methotrexate, azathioprine) |
2 (3%) |
||||||||||||||
|
Carbimazole |
1 (1%) |
||||||||||||||
|
Rivaroxaban |
1 (1%) |
||||||||||||||
|
Methoxyflurane |
1 (1%) |
||||||||||||||
|
Allopurinol |
1 (1%) |
||||||||||||||
|
Buprenorphine |
1 (1%) |
||||||||||||||
|
Norethisterone |
1 (1%) |
||||||||||||||
|
|
|||||||||||||||
|
|
|||||||||||||||
Box 5 – Transplant‐free survival in patients with drug‐induced liver injury: Kaplan–Meier analyses
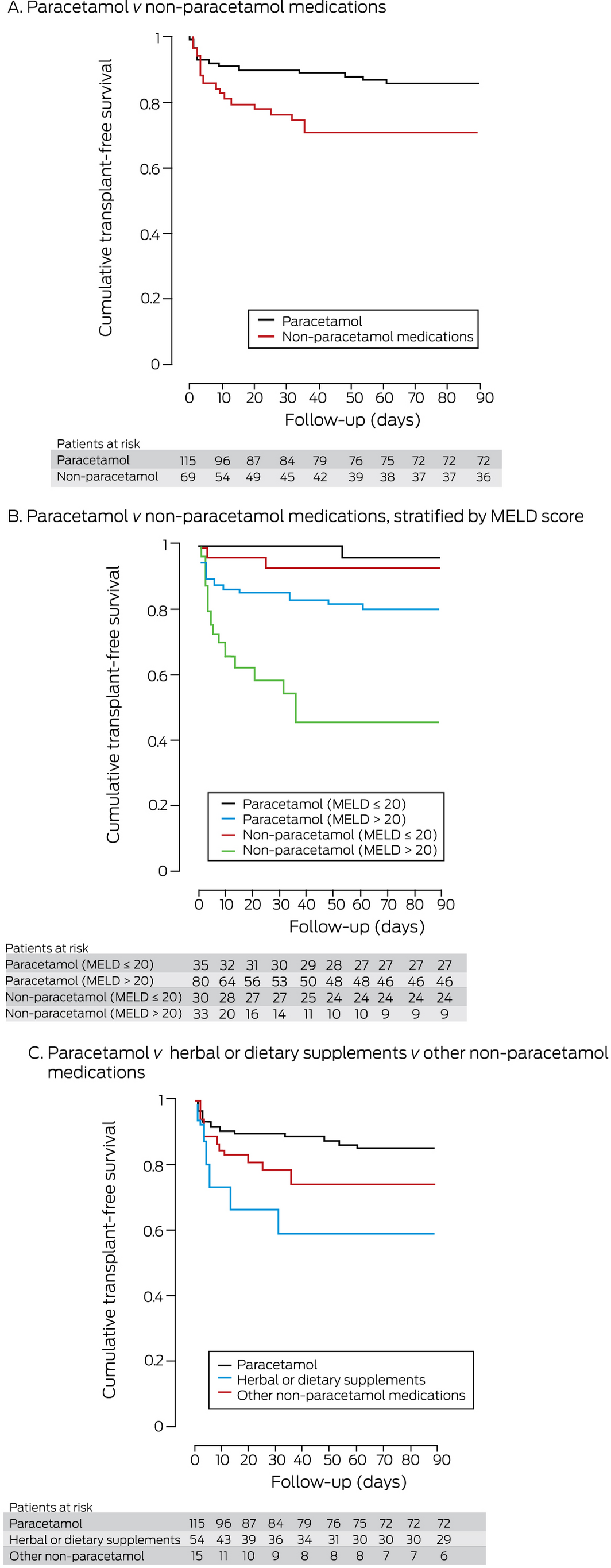
MELD = model for end‐stage liver disease.
Log‐rank analyses: A. Paracetamol v non‐paracetamol medications: P = 0.019. B. Paracetamol v non‐paracetamol (MELD ≤ 20): P = 0.49; paracetamol v non‐paracetamol (MELD > 20): P = 0.001; paracetamol, MELD ≤ 20 v MELD > 20: P = 0.029; paracetamol, MELD ≤ 20 v MELD > 20: P < 0.001. C. Paracetamol v dietary and herbal supplements: P = 0.007; paracetamol v other non‐paracetamol medications: P = 0.09; dietary and herbal supplements v other non‐paracetamol medications: P = 0.23.
Box 6 – Admission factors (selected by LASSO) associated with death or transplantation by 90 days for people hospitalised with drug‐induced liver injury: uni‐ and multivariable analyses
|
Factor |
Hazard ratio |
Adjusted hazard ratio (95% CI) |
|||||||||||||
|
|
|||||||||||||||
|
Non‐paracetamol drug‐induced liver injury |
|
|
|||||||||||||
|
Alkaline phosphatase level (per U/L) |
1.00 (1.00–1.00) |
1.00 (1.00–1.01) |
|||||||||||||
|
‐Glutamyltransferase (per U/L) |
1.00 (1.00–1.00) |
1.00 (0.99–1.00) |
|||||||||||||
|
Alanine aminotransferase (per U/L) |
1.00 (1.00–1.00) |
1.00 (1.00–1.00) |
|||||||||||||
|
Aspartate aminotransferase (per U/L) |
1.00 (1.00–1.00) |
1.00 (1.00–1.00) |
|||||||||||||
|
MELD score (per point) |
1.14 (1.08–1.22) |
1.24 (1.14–1.36) |
|||||||||||||
|
Paracetamol‐related drug‐induced liver injury |
|
|
|||||||||||||
|
Age (per year) |
1.01 (0.98–1.04) |
1.06 (0.98–1.17) |
|||||||||||||
|
Admission sodium level (per mmol/L) |
1.22 (1.10–1.35) |
1.26 (1.05–1.54) |
|||||||||||||
|
Admission albumin level (per g/L) |
0.93 (0.86–1.00) |
0.96 (0.82–1.12) |
|||||||||||||
|
‐Glutamyltransferase (per U/L) |
1.00 (1.00–1.00) |
0.99 (0.99–1.01) |
|||||||||||||
|
Alanine aminotransferase (per U/L) |
1.00 (1.00–1.00) |
1.00 (1.00–1.00) |
|||||||||||||
|
Aspartate aminotransferase (per U/L) |
1.00 (1.00–1.00) |
1.00 (1.00–1.00) |
|||||||||||||
|
MELD score (per point) |
1.12 (1.04–1.19) |
1.19 (1.09–3.74) |
|||||||||||||
|
|
|||||||||||||||
|
CI = confidence interval; LASSO = least absolute shrinkage and selection operator method; MELD = model for end‐stage liver disease. |
|||||||||||||||
Received 19 December 2020, accepted 4 June 2021
- Emily Nash1
- Abdul‐Hamid Sabih1
- John Chetwood1
- Georgette Wood2
- Keval Pandya1
- Terry Yip3
- Avik Majumdar1,2
- Geoffrey W McCaughan1,2
- Simone I Strasser1,2
- Ken Liu1,2
- 1 AW Morrow Gastroenterology and Liver Centre, Royal Prince Alfred Hospital, Sydney, NSW
- 2 Sydney Medical School, University of Sydney, Sydney, NSW
- 3 Medical Data Analytic Centre and Institute of Digestive Disease, Chinese University of Hong Kong, Hong Kong
No relevant disclosures.
- 1. Hoofnagle JH, Björnsson ES. Drug‐induced liver injury: types and phenotypes. N Engl J Med 2019; 381: 264–273.
- 2. Sistanizad M, Peterson GM. Drug‐induced liver injury in the Australian setting. J Clin Pharm Ther 2013; 38: 115–120.
- 3. Vuppalanchi R, Liangpunsakul S, Chalasani N. Etiology of new‐onset jaundice: how often is it caused by idiosyncratic drug‐induced liver injury in the United States? Am J Gastroenterol 2007; 102: 558–562.
- 4. European Association for the Study of the Liver. EASL clinical practice guidelines: drug induced liver injury. J Hepatol 2019; 70: 1222–1261.
- 5. Cairns R, Brown JA, Wylie CE, et al. Paracetamol poisoning‐related hospital admissions and deaths in Australia, 2004–2017. Med J Aust 2019; 211: 218–223. https://www.mja.com.au/journal/2019/211/5/paracetamol-poisoning-related-hospital-admissions-and-deaths-australia-2004-2017
- 6. Navarro VJ, Khan I, Björnsson E, et al. Liver injury from herbal and dietary supplements. Hepatology 2017; 65: 363–373.
- 7. Navarro VJ, Barnhart H, Bonkovsky HL, et al. Liver injury from herbals and dietary supplements in the US Drug‐Induced Liver Injury Network. Hepatology 2014; 60: 1399–1408.
- 8. Gow PJ, Connelly NJ, Hill RL, et al. Fatal fulminant hepatic failure induced by a natural therapy containing kava. Med J Aust 2003; 180: 198–199. https://www.mja.com.au/journal/2003/178/9/fatal-fulminant-hepatic-failure-induced-natural-therapy-containing-kava
- 9. Smith RJ, Bertilone C, Robertson AG. Fulminant liver failure and transplantation after use of dietary supplements. Med J Aust 2016; 204: 30–32. https://www.mja.com.au/journal/2016/204/1/fulminant-liver-failure-and-transplantation-after-use-dietary-supplements
- 10. Danan G, Benichou C. Causality assessment of adverse reactions to drugs. I. A novel method based on the conclusions of International Consensus Meeting: application to drug‐induced liver injuries. J Clin Epidemiol 1993; 46: 1323–1330.
- 11. Kamath PS, Kim WR; Advanced Liver Disease Study Group. The model for end‐stage liver disease (MELD). Hepatology 2007; 45: 797–805.
- 12. Bénichou C. Criteria of drug‐induced liver disorders. Report of an international consensus meeting. J Hepatol 1990; 11: 272–276.
- 13. Robles‐Díaz M, Lucena MI, Kaplowitz N, et al; Spanish DILI Registry; SLatinDILI Network; Safer and Faster Evidence‐based Translation Consortium. Use of Hy’s law and a new composite algorithm to predict acute liver failure in patients with drug‐induced liver injury. Gastroenterology 2014; 147: 109–118.
- 14. O’Grady JG, Alexander GJ, Hayllar KM, Williams R. Early indicators of prognosis in fulminant hepatic failure. Gastroenterology 1989; 97: 439–445.
- 15. National Institute of Diabetes and Digestive and Kidney Diseases. LiverTox. Clinical and research information on drug‐induced liver injury. Updated June 2021. https://www.ncbi.nlm.nih.gov/books/NBK547852 (viewed June 2021).
- 16. Katarey D, Verma S. Drug‐induced liver injury. Clin Med (Lond) 2016; 16 (Suppl 6): s104–s109.
- 17. McPhail MJW, Farne H, Senvar N, et al. Ability of King’s College criteria and model for end‐stage liver disease scores to predict mortality of patients with acute liver failure: a meta‐analysis. Clin Gastroenterol Hepatol 2016; 14: 516–525.
- 18. Björnsson ES, Bergmann OM, Björnsson HK, et al. Incidence, presentation, and outcomes in patients with drug‐induced liver injury in the general population of Iceland. Gastroenterology 2013; 144: 1419–1425.
- 19. Medina‐Caliz I, Garcia‐Cortes M, Gonzalez‐Jimenez A, et al; Spanish DILI Registry. Herbal and dietary supplement‐induced liver injuries in the Spanish DILI registry. Clin Gastroenterol Hepatol 2018; 16: 1495–1502.
- 20. Barnes J, McLachlan AJ, Sherwin CMT, Enioutina EY. Herbal medicines: challenges in the modern world. Part 1. Australia and New Zealand. Expert Rev Clin Pharmacol 2016; 9: 905–915.
- 21. de Boer YS, Sherker AH. Herbal and dietary supplement induced liver injury. Clin Liver Dis 2017; 21: 135–149.
- 22. Grewal P, Ahmad J. Severe liver injury due to herbal and dietary supplements and the role of liver transplantation. World J Gastroenterol 2019; 25: 6704–6712.
- 23. Hillman L, Gottfried M, Whitsett M, et al. Clinical features and outcomes of complementary and alternative medicine induced acute liver failure and injury. Am J Gastroenterol 2017; 111: 958–965.
- 24. Cohen PA. Assessing supplement safety: the FDA’s controversial proposal. N Engl J Med 2012; 366: 389–391.
- 25. Mitchell SJ, Hilmer SN. Drug‐induced liver injury in older adults. Ther Adv Drug Saf 2010; 1: 65–77.





Abstract
Objective: To compare the characteristics and outcomes of drug‐induced liver injury (DILI) caused by paracetamol and non‐paracetamol medications, particularly herbal and dietary supplements.
Design: Retrospective electronic medical record data analysis.
Setting, participants: Adults admitted with DILI to the Gastroenterology and Liver Centre at the Royal Prince Alfred Hospital, Sydney (a quaternary referral liver transplantation centre), 2009–2020.
Main outcome measures: 90‐day transplant‐free survival; drugs implicated as causal agents in DILI.
Results: A total of 115 patients with paracetamol‐related DILI and 69 with non‐paracetamol DILI were admitted to our centre. The most frequently implicated non‐paracetamol medications were antibiotics (19, 28%), herbal and dietary supplements (15, 22%), anti‐tuberculosis medications (six, 9%), and anti‐cancer medications (five, 7%). The number of non‐paracetamol DILI admissions was similar across the study period, but the proportion linked with herbal and dietary supplements increased from 2 of 13 (15%) during 2009–11 to 9 of 19 (47%) during 2018–20 (linear trend: P = 0.011). Despite higher median baseline model for end‐stage liver disease (MELD) scores, 90‐day transplant‐free survival for patients with paracetamol‐related DILI was higher than for patients with non‐paracetamol DILI (86%; 95% CI, 79–93% v 71%; 95% CI, 60–82%) and herbal and dietary supplement‐related cases (59%; 95% CI, 34–85%). MELD score was an independent predictor of poorer 90‐day transplant‐free survival in both paracetamol‐related (per point increase: adjusted hazard ratio [aHR], 1.19; 95% CI, 1.09–3.74) and non‐paracetamol DILI (aHR, 1.24; 95% CI, 1.14–1.36).
Conclusion: In our single centre study, the proportion of cases of people hospitalised with DILI linked with herbal and dietary supplements has increased since 2009. Ninety‐day transplant‐free survival for patients with non‐paracetamol DILI, especially those with supplement‐related DILI, is poorer than for those with paracetamol‐related DILI.