The known Australia trails other countries with regard to organ donation rates. The potential for organ donation after circulatory death (DCD), which is expanding in importance overseas, is unknown.
The new Many potential DCD donors are not recognised. Organ donation is frequently not discussed with the families of potential donors. As many as two-thirds of potential DCD kidney donors are not identified. Older patients, those dying of non-neurological causes, and patients with chronic single organ disease are often not identified as potential donors.
The implications Systems for reviewing the medical suitability for organ donation of all patients undergoing end-of-life care in critical care environments should be established.
Organ donation rates in Australia increased from about 200 donors per year in the early 2000s to 379 in 2014; that is, from 10.2 to 16.1 donors per million population (dpmp).1 Although approaching the national target of 25 dpmp by 2018,2 Australia still lags behind countries such as Spain, where the overall deceased donation rate is 39.7 dpmp.3 In many countries, donation after circulatory death (DCD) accounts for an increasing proportion of donor numbers, although comparisons are limited by variations in DCD practices.4 Within Australia, where it is undertaken in a controlled manner according to Maastricht category III,5 DCD resulted in 107 donors (28% of all deceased organ donations; 4.6 dpmp)1 in 2014.
Outcomes of DCD renal and lung transplantation6,7 are similar to those for donation after brain death. Although DCD liver transplantation has been associated with primary graft dysfunction and ischaemic cholangiopathy,8,9 rates of hepatic DCD have also risen.10 Further, the first DCD heart donation programs commenced in Australia in July 2014, with good early outcomes.11,12 The full potential for increasing transplantation numbers through DCD remains unknown.
Several studies have estimated the donor potential in Australia and elsewhere,13-17 but have not specifically investigated DCD. Lack of familiarity with DCD in Australia18 and the difficulty of predicting time to death after withdrawal of cardio-respiratory support may limit consideration of DCD by clinicians. In addition, there are no standardised DCD donor suitability criteria or definitive measurement methodology; as a result, the potential for DCD in Australian hospitals is unknown.
The DonateLife Audit was established as a monitoring tool by the Australian Organ and Tissue Authority and DonateLife Network. It is designed to identify missed donation opportunities and barriers to donation, and to measure rates of request, consent and conversion of potential into actual donations. Our aim was to apply ideal and expanded organ suitability criteria to determine the potential for DCD at contributing hospitals on the basis of DonateLife Audit data, and to compare this potential with actual DCD rates. We hypothesised that there is a large unrealised potential for DCD in Australian hospitals, and that identifying unrecognised candidate donors could increase overall donation rates.
Methods
Setting
We identified data in the DonateLife Audit database for all patients who died during July 2012 – December 2014 at the 75 contributing hospitals, and who had been intubated and were neither confirmed as being brain-dead nor likely to have met brain death criteria at the official time of death. The Audit retrospectively recorded data for all patients aged 28 days to 80 years who had died in an intensive care unit or emergency department, or who had died because of an irrecoverable brain injury within 24 hours of discharge from either type of department. Data collected by funded, dedicated, trained staff at each hospital included baseline patient characteristics, donation discussions and outcomes, brain death status, organs donated, time and location of the withdrawal of cardio-respiratory support and death, and laboratory and imaging results related to cardiac, renal, pancreatic, hepatic and respiratory functions.
Development of organ suitability criteria
We applied a hierarchical process in developing ideal and expanded criteria for assessing potential DCD donors. Overall and organ-specific criteria for donor suitability were derived from national guidelines19 and, when more information was required, from international guidelines.20 In the absence of published guidelines, opinions were sought from directors of regional transplant programs, and finally from local transplantation and donation experts (Box 1).
Specific values recorded in the DonateLife Audit were the lower of the blood creatinine level measurements recorded during hospitalisation (at admission and the final level before death), the highest Pao2/Fio2 ratio from the final three arterial blood gas measurements, the final alanine aminotransferase (ALT) level, and the findings from the final chest x-ray.
Patients were classified into three mutually exclusive groups:
-
Actual DCD donors had died a circulatory death, and organ retrieval for the purpose of transplantation had commenced in the operating room (with surgical incision); this included patients whose organs were deemed medically unsuitable during surgery, prior to or after organ removal.
-
Potential ideal DCD donors had died a circulatory death after withdrawal of cardio-respiratory support, and satisfied the ideal criteria for donation of one or more organs listed in Box 1, but did not actually become donors.
-
Potential expanded criteria DCD donors met the expanded criteria (Box 1) but not the criteria for ideal DCD donors, and had not actually become donors.
Patients who did not die within the specified time period for donating an organ were not deemed to be potential donors. The internal validity of the definitional criteria was tested by assessing how many actual donors satisfied them.
To estimate the unrealised potential pool of additional donors for DCD in Australia, we identified patients in each of the ideal and expanded potential donor groups whose families had not been approached with regard to organ donation. An identification rate of 100% and a consent rate of 60% (the consent rate in Australia in 20142) were then applied. These steps were repeated for each specific organ.
When assessing variability in donor practices, five smaller regions (the Australian Capital Territory, the Northern Territory, South Australia, Tasmania, and Western Australia) were collapsed into one group. Donation rates were compared in a de-identified manner with those in the three larger states (New South Wales, Queensland and Victoria) and across different hospital types.
For kidney and lung donation (organs potentially more resistant to longer periods of ischaemia), the impact of increasing the acceptable time to death after withdrawal of cardio-respiratory support on the numbers of DCD donors was also evaluated.
Statistical analysis
Data were analysed in Stata 13 (StataCorp) and Excel (Microsoft). Data are reported as absolute numbers and proportions, and as means (with standard deviations) or medians (with interquartile ranges); they were compared in χ2, Wilcoxon or Kruskal–Wallis tests as appropriate for the distribution of the data and the numbers of groups compared.
Ethics approval
The project was approved as a low risk study by the Human Research and Ethics Committee of Alfred Health (study number, 336/14).
Results
During the 30-month study period, there were 87 158 deaths at the 75 participating hospitals; data for 16 477 deaths were entered into the DonateLife Audit. Of these, 10 293 patients had been intubated prior to death, including 1513 who were confirmed as or were likely to have been brain-dead (606 donors). Of the remaining 8780 patients, 202 were actual DCD donors, 318 were deemed to be potential ideal donors, and 435 were potential expanded criteria donors.
Actual DCD donors
Organs were retrieved for transplantation in 185 cases from 202 actual DCD donors (Box 2): 178 kidney, 69 lung, 35 liver, three pancreas, and three heart donors (note: cardiac DCD began in July 2014). Most donors (83%) were at tertiary centres (Box 2, Box 3). The largest number of DCD donors were in Victoria (90 of 202, 45%), even after indexing against the number of deaths (Box 4). One hundred actual donors (50%) also met at least one of the ideal DCD organ criteria, 56 (28%) met expanded DCD criteria, and 46 actual donors (23%) met neither criterion (online Appendix, Box 5).
Potential DCD donors: overall numbers and effect on overall donation rates
Of the 318 potential ideal and 435 potential expanded criteria donors, organ donation had not been discussed with the families of 193 (61%) and 313 (72%) respectively (Box 2, Box 5). Ideal and expanded criteria potential donors were more likely to have died of non-neurological causes than actual donors, and also had more comorbidities, less favourable clinical and laboratory results, and higher noradrenaline requirements (Box 2). The median age of potential ideal donors was less than that of actual DCD donors, while the median age of expanded criteria donors was higher (Box 2). Potential donors were predominantly located at tertiary hospitals (369 of 506, 73%; Box 3). Consent was obtained for 35 potential ideal (11%) and 47 potential expanded criteria donors (11%); reasons for non-donation included medical contraindications (43% and 51% respectively), failed physiological support (14% and 9%), and unsuitable organs (6% and 17%).
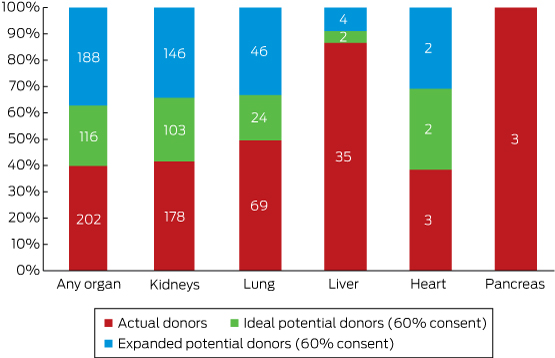
Applying a notional consent rate of 60%, 116 potential ideal donors and 188 potential expanded criteria donors could be considered as the potential for additional DCD in DonateLife hospitals (Box 6). This is equivalent to 46 extra ideal donors and 75 extra expanded criteria donors per year, increasing the overall donation rate from 16.1 to 18.1 dpmp (12% increase; DCD, 36% of all deceased organ donations) if ideal donors were included, and to 21.3 dpmp if both ideal and expanded criteria donors were included (32% increase; DCD, 46%).
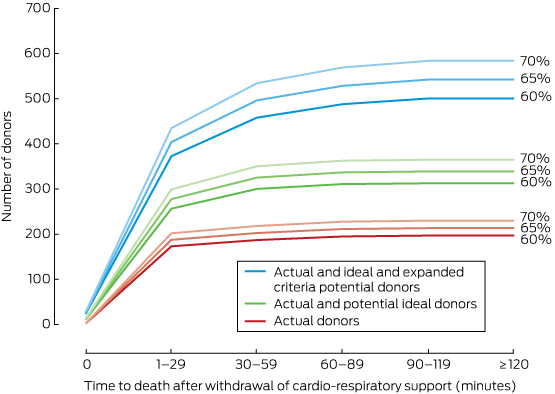
Most potential donors (271, 89%) died within 60 minutes of cardio-respiratory support being withdrawn, and 213 potential donors (70%) were under 60 years of age (Box 7).
Potential DCD donors: organ-specific information
Organ donation had been discussed with the families of 30% of potential kidney donors; donation had not been discussed for 172 ideal and 244 expanded criteria potential kidney donors. Forty ideal and 77 expanded criteria potential lung donors and nine potential liver donors had not had organ donation discussed; there were eight potential heart donors and no potential pancreas donors.
Effect of increasing the acceptable time between withdrawal of cardio-respiratory support and death, and of increasing the consent rate on donation of kidneys and lungs
Accepting organs from ideal and extended criteria donors declared dead up to 120 minutes after withdrawal of cardio-respiratory support (keeping other criteria constant and assuming a consent rate of 60%) would lead to an 18% increase in potential kidney donors (from 394 at 60 min to 467) and a 5% increase in lung donors (from 138 at 90 min to 145) compared with numbers expected in standard donation time frames; extending the acceptable time to 180 minutes would achieve a 32% increase in kidney donors (from 394 to 522) and a 16% increase in lung donors (from 138 to 160) (Box 8).
Modelling the effect of increasing consent rates suggested that identifying potential ideal and expanded criteria donors could achieve a greater overall rise in donor numbers than increasing the consent rate from 60% to 70% alone (Box 9).
Discussion
Our review of the DonateLife Audit database identified a large untapped potential for DCD donations. Many patients with criteria compatible with organ donation did not become donors, nor had the opportunity for donation been discussed with their families; most were potential kidney or lung donors.
Donation was not discussed with the families of several key subgroups of potential donors: those who died of non-neurological causes, patients with chronic organ disease, and older patients. Half the potential donors died of non-neurological causes, compared with 8% of actual DCD donors. They may not be recognised as potential donors because of historic reliance on brain death criteria for donation, the recent uptake of DCD, and lesser awareness among clinical staff that patients without brain injury can be donors. Such patients may have more comorbidities and greater organ dysfunction than donors with a neurological cause of death, and may therefore be dismissed as medically unsuitable, despite the fact that certain organs might be acceptable for donation. Chronic heart, lung or liver disease were each recorded for one in five ideal potential DCD donors, compared with 5–15% of actual DCD donors. Including older patients with good organ function would appear to greatly increase the DCD potential (Box 7); proceeding with older donors after brain death is already undertaken in Australia.1
While more than one strategy should be employed to increase donation rates, our results suggest that extending the acceptable time until death after withdrawal of cardio-respiratory support and increasing family consent rates would win relatively fewer additional donors compared with identifying candidates better, ensuring that families are approached for donation, and relaxing age limits. Similar observations have been made by others who have examined the time to death after withdrawal of cardio-respiratory support in a series of attempted DCD.21
The Canadian Institute for Health Information examined conversion rates for possible DCD donors in 2014 by calculating the proportion of suitable donors who donated organs.22 Their conversion rates after circulatory death were one-sixth of those for donations after brain death, consistent with our concept of missed DCD donor potential.
Implications
The potential donors identified would have provided many additional organs for transplantation. Optimal identification of candidate donors and discussions with families at current consent rates would have resulted in a deceased donation rate of 21.3 dpmp, with DCD comprising 46% of all donations in Australia, comparable with rates in the United Kingdom.23 The large gap between DCD potential and actual donor rates warrants systematic review of the potential for donation by all patients undergoing end-of-life care. Organisations such as DonateLife should ensure that critical care and donation specialists are aware of this untapped potential. Although some increase in the number of potential DCD donors can be achieved with current processes, accurately assessing resource levels would be required to manage more donors and the subsequent increase in transplantation activity.
Strengths and limitations
This is the first examination of the national potential for DCD according to actual patient characteristics. The quality of data in the DonateLife Audit is probably high, as it is collected by trained data collectors. However, the data are retrospective and, although donation outcomes and basic demographic information are routinely checked, the accuracy of specific organ function parameters are not audited. Data collection processes may vary between centres. Without consensus regarding DCD donor suitability criteria, we have generated criteria based on expert opinion and published guidelines. Some data types specified in previous guidelines, such as weight, hepatic steatosis and cold ischaemic time, were not available to us.
Our criteria may under- or overestimate the potential for DCD in Australia. We cannot predict how many potential DCD donors might have organs deemed medically unsuitable for transplantation; including these donors would lead to our overestimating the potential. However, given that 23% of actual DCD donors met neither ideal nor expanded criteria, it is likely there are suitable donors who also fail to meet our criteria. The low numbers of potential liver and heart DCD donors may also have been attributable to our conservative criteria. In 2014, 97% of actual donors were located in DonateLife hospitals; however, 62 adult intensive care units in Australia were not part of the network. These were predominantly small regional and private intensive care units, but also included one tertiary unit; together, they accounted for about one-third of available intensive care unit beds.24 The number of potential donors in these hospitals is unknown, possibly leading to our underestimating the full potential of DCD.
Conclusion
There is an untapped potential in Australia for DCD donation, particularly of kidneys and lungs. Patients dying of non-neurological conditions, who are older, or who have chronic illnesses may be missed as donor candidates. Processes should be developed in hospital critical care environments that facilitate evaluating every patient undergoing end-of-life care for their medical suitability and potential to donate; the validity and cost of such routine evaluation should be examined. Assessing the potential for donation in hospitals outside the DonateLife network should also be attempted. Research into long term organ outcomes from ideal and expanded DCD donors would allow more informed donor criteria.
Box 1 – Criteria for classification of patients as ideal or expanded criteria potential donation after circulatory death (DCD) donors*
|
Organ |
Ideal DCD criteria |
Expanded DCD criteria |
|||||||||||||
|
|
|||||||||||||||
|
All organs |
|
|
|||||||||||||
|
Kidney |
|
|
|||||||||||||
|
Lung |
|
|
|||||||||||||
|
Liver |
|
|
|||||||||||||
|
Heart |
|
|
|||||||||||||
|
Pancreas |
|
|
|||||||||||||
|
|
|||||||||||||||
|
HIV = human immunodeficiency virus; WCRS = time to death after withdrawal from cardio-respiratory support. * Groups are mutually exclusive; ie, patients who met ideal potential donor criteria were excluded from the expanded group. |
|||||||||||||||
Box 2 – Characteristics of potential ideal donation after circulatory death (DCD) donors, potential expanded criteria DCD donors, and actual DCD donors
|
|
Potential ideal DCD donors |
Potential expanded DCD donors |
Actual DCD donors |
P |
|||||||||||
|
|
|||||||||||||||
|
Total number of patients |
318 |
435 |
202* |
|
|||||||||||
|
No discussion about donation (ie, patients who represent additional unrecognised potential for DCD) |
193 (61%) |
313 (72%) |
2 (1%) |
|
|||||||||||
|
Donation discussed with family |
125 (39%) |
122 (28%) |
200* (99%) |
< 0.001 |
|||||||||||
|
Consented to organ donation |
35 (11%) |
47 (11%) |
200 (99%) |
|
|||||||||||
|
Age (years), median (IQR) |
44 (29–53) |
57 (43–65) |
52 (31–60) |
< 0.001 |
|||||||||||
|
Sex (men) |
190 (60%) |
255 (59%) |
139 (69%) |
0.045 |
|||||||||||
|
Length of stay in intensive care unit (days), median (IQR) |
5 (3–12) |
4 (2–10) |
4 (2–6) |
0.020 |
|||||||||||
|
Number at tertiary centres (%) |
241 (76%) |
327 (75%) |
167 (83%) |
0.085 |
|||||||||||
|
Time to death after WCRS (minutes), median (IQR) |
17 (10–30) |
20 (10–45) |
18 (13–22) |
0.003 |
|||||||||||
|
Failure of physiologic support |
0 |
0 |
0 |
|
|||||||||||
|
Cause of death |
|
|
|
< 0.001 |
|||||||||||
|
Non-neurological |
149 (47%) |
218 (50%) |
16 (7.9%) |
|
|||||||||||
|
Cerebral hypoxia |
88 (28%) |
104 (24%) |
86 (43%) |
|
|||||||||||
|
Intracranial haemorrhage |
32 (10%) |
63 (15%) |
48 (24%) |
|
|||||||||||
|
Traumatic brain injury |
28 (8.8%) |
15 (3.5%) |
36 (18%) |
|
|||||||||||
|
Cerebral infarction |
11 (3.5%) |
19 (4.4%) |
12 (5.9%) |
|
|||||||||||
|
Other neurological |
9 (3%) |
15 (3.5%) |
4 (2%) |
|
|||||||||||
|
Chronic medical conditions |
|
|
|
|
|||||||||||
|
Chronic heart disease |
58 (18%) |
138 (32%) |
21 (10%) |
< 0.001 |
|||||||||||
|
Chronic lung disease |
54 (17%) |
90 (21%) |
30 (15%) |
0.16 |
|||||||||||
|
Chronic liver disease |
63 (20%) |
88 (20%) |
11 (5.5%) |
< 0.001 |
|||||||||||
|
Chronic renal disease |
3 (1%) |
20 (4.6%) |
0 |
< 0.001 |
|||||||||||
|
Peripheral vascular disease |
6 (2%) |
35 (8.0%) |
1 (1%) |
< 0.001 |
|||||||||||
|
Diabetes |
37 (12%) |
67 (15%) |
11 (5.5%) |
0.002 |
|||||||||||
|
Hypertension |
22 (6.9%) |
109 (25%) |
45 (22%) |
< 0.001 |
|||||||||||
|
Hepatitis C |
0 |
80 (18%) |
2 (1%) |
< 0.001 |
|||||||||||
|
Recent intravenous drug use |
22 (6.9%) |
57 (13%) |
3 (2%) |
< 0.001 |
|||||||||||
|
Laboratory results, median (IQR) |
|
|
|
|
|||||||||||
|
Alanine aminotransferase level (IU/L) |
68 (33–157) |
70 (29–214) |
51 (29–96) |
0.01 |
|||||||||||
|
Highest Pao2/Fio2 ratio |
305 (171–390) |
290 (167–383) |
420 (311–477) |
< 0.001 |
|||||||||||
|
Lower serum creatinine level during admission (μmol/L) |
75 (54–100) |
85 (60–119) |
64 (50–81) |
< 0.001 |
|||||||||||
|
Chest x-ray: major changes |
94/244 (39%) |
102/298 (34%) |
34/170 (20%) |
< 0.001 |
|||||||||||
|
Current or ex-smoker |
15/202 (7.4%) |
21/275 (7.6%) |
23/109 (21%) |
0.003 |
|||||||||||
|
Renal replacement therapy |
75 (24%) |
107 (25%) |
8 (4%) |
< 0.001 |
|||||||||||
|
Noradrenaline or adrenaline administered |
189/221 (86%) |
257/313 (82%) |
107/129 (83%) |
0.57 |
|||||||||||
|
Noradrenaline dose (μg/min), median (IQR) |
13 (0.4–33) |
10 (0.05–30) |
5 (0.5–12) |
0.001 |
|||||||||||
|
|
|||||||||||||||
|
* Procedural consent for donation was obtained in the absence of family or next of kin who could provide consent. |
|||||||||||||||
Box 3 – Numbers of actual donation after circulatory death (DCD) donors and of potential ideal and expanded criteria DCD donors with whose families organ donation had not been discussed, by hospital type
|
|
Overall |
Hospital type |
P |
||||||||||||
|
Tertiary |
Metropolitan |
Rural |
Private |
||||||||||||
|
|
|||||||||||||||
|
Number of hospitals |
75 |
34 |
15 |
22 |
4 |
|
|||||||||
|
Deaths (non-brain deaths) |
8780 |
6125 |
1446 |
1136 |
73 |
|
|||||||||
|
Actual DCD donors |
|
|
|
|
|
|
|||||||||
|
Number |
202 |
167 |
26 |
9 |
0 |
|
|||||||||
|
Number per 1000 non-brain deaths |
23.0 |
27.3 |
18.0 |
7.9 |
0 |
< 0.001 |
|||||||||
|
Potential ideal DCD donors without discussion of organ donation |
|
|
|
|
|
|
|||||||||
|
Number |
193 |
141 |
24 |
28 |
0 |
|
|||||||||
|
Number per 1000 non-brain deaths |
22.5 |
23.7 |
16.9 |
24.8 |
0 |
< 0.001 |
|||||||||
|
Number (60% consent) |
116 |
85 |
14 |
17 |
0 |
|
|||||||||
|
Number per 1000 non-brain deaths (60% consent) |
13.5 |
14.2 |
10.1 |
14.9 |
0 |
0.002 |
|||||||||
|
Potential expanded criteria DCD donors without discussion of organ donation |
|
|
|
|
|
|
|||||||||
|
Number |
313 |
228 |
42 |
43 |
0 |
|
|||||||||
|
Number per 1000 non-brain deaths |
36.5 |
38.3 |
29.6 |
38.2 |
0 |
< 0.001 |
|||||||||
|
Number (60% consent) |
188 |
137 |
25 |
26 |
0 |
|
|||||||||
|
Number per 1000 non-brain deaths (60% consent) |
21.9 |
23.0 |
17.7 |
22.9 |
0 |
< 0.001 |
|||||||||
|
|
|||||||||||||||
|
|
|||||||||||||||
Box 4 – Numbers of actual donation after circulatory death (DCD) donors and of potential ideal and expanded criteria DCD donors with whose families organ donation had not been discussed, by region
|
|
Overall |
Region |
P |
||||||||||||
|
Victoria |
NSW |
Queensland |
Other* |
||||||||||||
|
|
|||||||||||||||
|
Number of hospitals |
75 |
23 |
20 |
12 |
20 |
|
|||||||||
|
Deaths (non-brain deaths) |
8780 |
2452 |
3021 |
1349 |
1958 |
|
|||||||||
|
Actual DCD donors |
|
|
|
|
|
|
|||||||||
|
Number |
202 |
90 |
47 |
41 |
24 |
|
|||||||||
|
Number per 1000 non-brain deaths |
23.0 |
36.7 |
15.6 |
30.4 |
12.3 |
< 0.001 |
|||||||||
|
Potential ideal DCD donors without discussion of organ donation |
|
|
|
|
|
|
|||||||||
|
Number |
193 |
71 |
28 |
45 |
49 |
|
|||||||||
|
Number per 1000 non-brain deaths |
22.5 |
30.1 |
9.4 |
34.4 |
25.3 |
0.002 |
|||||||||
|
Number (60% consent) |
116 |
43 |
17 |
27 |
29 |
|
|||||||||
|
Number per 1000 non-brain deaths (60% consent) |
13.5 |
18.0 |
5.6 |
20.6 |
15.2 |
0.035 |
|||||||||
|
Potential expanded criteria DCD donors without discussion of organ donation |
|
|
|
|
|
|
|||||||||
|
Number |
313 |
120 |
80 |
60 |
53 |
|
|||||||||
|
Number per 1000 non-brain deaths |
36.5 |
50.8 |
26.9 |
45.9 |
27.4 |
0.007 |
|||||||||
|
Number (60% consent) |
188 |
72 |
48 |
36 |
32 |
|
|||||||||
|
Number per 1000 non-brain deaths (60% consent) |
21.9 |
30.5 |
16.1 |
27.5 |
16.4 |
0.062 |
|||||||||
|
|
|||||||||||||||
|
* Australian Capital Territory, Northern Territory, South Australia, Tasmania and Western Australia. |
|||||||||||||||
Box 5 – Numbers of actual donors and of potential donation after circulatory death donors*
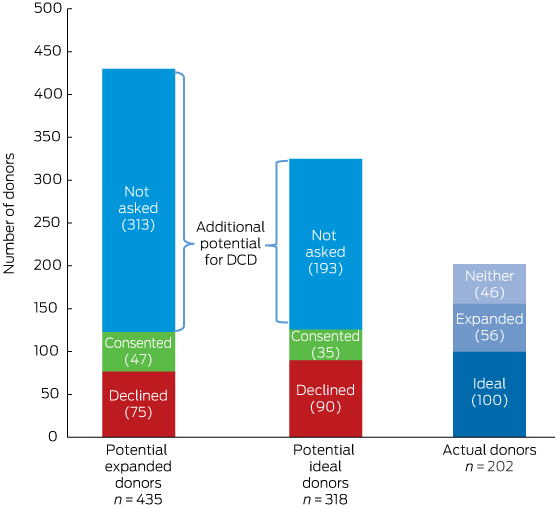
* The three groups are mutually exclusive: potential donors did not become donors but met specific donor organ suitability criteria (ideal or expanded). For actual donors, the numbers who satisfied the ideal and expanded donation criteria are shown.
Box 6 – Total potential of donation after circulatory death: additional organs that would be available were ideal and expanded criteria potential donors included
Box 7 – Cumulative numbers of actual and potential donors by time between withdrawal of cardio-respiratory support and death (A) and by age of donor (B)*
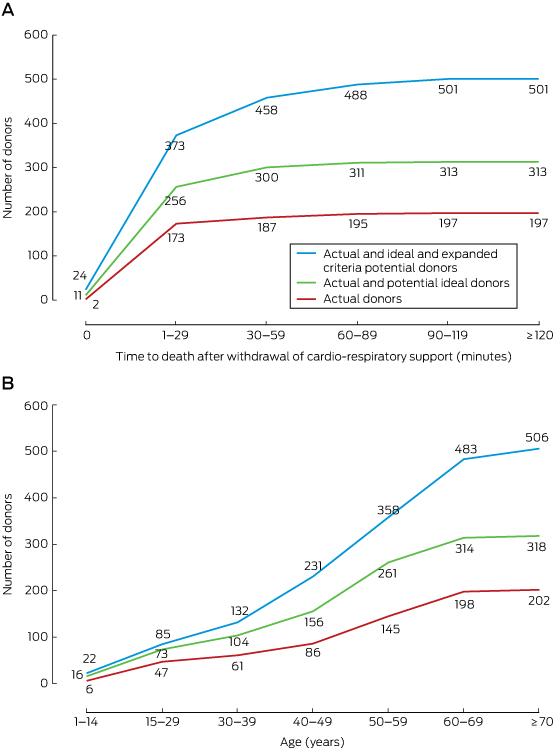
* A 60% consent rate and satisfaction of the organ suitability criteria in are assumed. The numbers indicate the cumulative numbers of actual and potential donors to that point on the graph.
Box 8 – The effect of increasing the acceptable time between withdrawal of cardio-respiratory support and death on the cumulative numbers of potential kidney (A) and lung (B) donors (donations after cardio-circulatory death)*
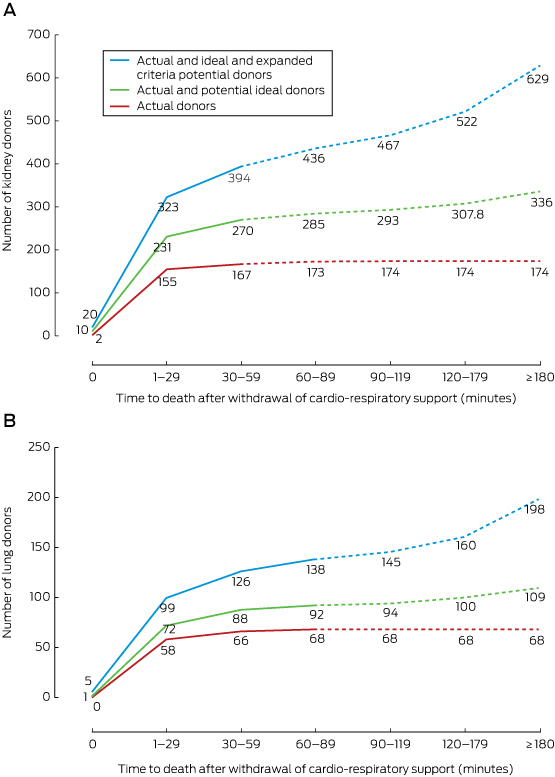
* Dotted lines indicate donation at a time point later than the ideal cut-off for time between withdrawal of support and death (60 minutes for kidneys, 90 minutes for lungs; assumed consent rate, 60%). The numbers indicate the cumulative numbers of actual and potential donors to that point on the graph.
Received 8 December 2016, accepted 31 March 2017
- Sandeep S Rakhra1
- Helen I Opdam2,3
- Laura Gladkis3
- Byron Arcia3
- Michael A Fink2,4
- John Kanellis5,6
- Peter S Macdonald7
- Gregory I Snell1
- David V Pilcher1,8
- 1 Alfred Health, Melbourne, VIC
- 2 Austin Health, Melbourne, VIC
- 3 Australian Organ and Tissue Authority, Canberra, ACT
- 4 University of Melbourne, Melbourne, VIC
- 5 Monash Health, Melbourne, VIC
- 6 Centre for Inflammatory Diseases, Monash University, Melbourne, VIC
- 7 St Vincent's Hospital, Sydney, NSW
- 8 Monash University, Melbourne, VIC
We acknowledge and thank all data collectors and audit officers who contribute to the DonateLife Audit. The DonateLife organisation is funded by the Australian Government. This particular research project was not funded.
No relevant disclosures.
- 1. Australia and New Zealand Organ Donation Registry. ANZOD Registry annual report 2015. Adelaide: ANZOD Registry, 2015. http://www.anzdata.org.au/anzod/ANZODReport/2015/2015ANZOD_annrpt.pdf (accessed Nov 2015).
- 2. Australian Organ and Tissue Donation and Transplantation Authority. Annual report 2014–15. Canberra: AOTDTA, 2015. http://www.donatelife.gov.au/sites/default/files/OTA_AR2015.pdf (accessed Dec 2015).
- 3. International Registry in Organ Donation and Transplantation. Final numbers 2014. IRODT, 2015. http://www.irodat.org/img/database/pdf/NEWSLETTER2015_December2.pdf (accessed Feb 2017).
- 4. Manara AR, Murphy PG, O’Callaghan G. Donation after circulatory death. Br J Anaesth 2012; 108 (Suppl 1): i108-i121.
- 5. Kootstra G, Daemen JH, Oomen AP. Categories of non-heart-beating donors. Transplant Proc 1995; 27: 2893-2894.
- 6. Snell GI, Esmore DS, Westall GP, et al. The Alfred Hospital lung transplant experience. Clin Transpl 2007: 131-144.
- 7. Doyle MBM, Collins K, Vachharajani N, et al. Outcomes using grafts from donors after cardiac death. J Am Coll Surg 2015; 221: 142-152.
- 8. Suárez F, Otero A, Solla M, et al. Biliary complications after liver transplantation from Maastricht category-2 non-heart-beating donors. Transplantation 2008; 85: 9-14.
- 9. Callaghan CJ, Charman SC, Muiesan P, et al. Outcomes of transplantation of livers from donation after circulatory death donors in the UK: a cohort study. BMJ Open 2013; 3: e003287.
- 10. Agopian VG, Petrowsky H, Kaldas FM, et al. The evolution of liver transplantation during 3 decades: analysis of 5347 consecutive liver transplants at a single center. Ann Surg 2013; 258: 409-421.
- 11. Macdonald P, Verran D, O’Leary M, et al. Heart transplantation from donation after circulatory death donors. Transplantation 2015; 99: 1101-1102.
- 12. Dhital KK, Iyer A, Connellan M, et al. Adult heart transplantation with distant procurement and ex-vivo preservation of donor hearts after circulatory death: a case series. Lancet 2015; 385: 2585-2591.
- 13. Pilcher D, Gladkis L, Arcia B, et al. Estimating the number of organ donors in Australian hospitals — implications for monitoring organ donation practices. Transplantation 2015; 99: 2203-2209.
- 14. Opdam HI, Silvester W. Potential for organ donation in Victoria: an audit of hospital deaths. Med J Aust 2006; 185: 250-254. <MJA full text>
- 15. Opdam HS, Silvester W. Identifying the potential organ donor: an audit of hospital deaths. Intensive Care Med 2004; 30: 1390-1397.
- 16. Kirchner C, Raduenz S, Fruehauf NR, et al. Estimated organ donor potential in German maximum care hospitals. Transplant Proc 2013; 45: 1310-1312.
- 17. Sheehy E, Conrad SL, Brigham LE, et al. Estimating the number of potential organ donors in the United States. N Engl J Med 2003; 349: 667-674.
- 18. Coulson TG, Pilcher DV, Graham SM, et al. Single-centre experience of donation after cardiac death. Med J Aust 2012; 197: 166-169. <MJA full text>
- 19. Transplantation Society of Australia and New Zealand. Clinical guidelines for organ transplantation from deceased donors; version 1.0. Apr 2015. http://www.donatelife.gov.au/sites/default/files/TSANZ%20Clinical%20Guidelines%20for%20Organ%20Transplantation%20from%20Deceased%20Donors_Version%201.0_April%202016.pdf (accessed Dec 2015).
- 20. British Transplantation Society. Transplantation from deceased donors circulatory death. 2013. https://bts.org.uk/wp-content/uploads/2016/09/15_BTS_Donors_DCD-1.pdf (accessed Oct 2015).
- 21. Scalea JR, Redfield RR, Rizzari MD, et al. When do DCD donors die? Outcomes and implications of DCD at a high-volume, single-center OPO in the United States. Ann Surg 2016; 263: 211-216.
- 22. Canadian Institute for Health Information. Deceased organ donor potential in Canada: report. Dec 2014. https://www.cihi.ca/sites/default/files/organdonorpotential_2014_en_0.pdf (accessed Feb 2016).
- 23. NHS Blood and Transplant. Organ donation and transplantation activity report 2014/15. http://nhsbtmediaservices.blob.core.windows.net/organ-donation-assets/pdfs/activity_report_2014_15.pdf (accessed Feb 2016).
- 24. Australia and New Zealand Intensive Care Society. 2015 Annual report. Melbourne: ANZICS, 2015. http://www.anzics.com.au/Downloads/ANZICS Annual Report 2015.pdf (accessed Mar 2017).





Abstract
Objective: To determine the potential for organ donation after circulatory death (DCD) in Australia by applying ideal and expanded organ suitability criteria, and to compare this potential with actual DCD rates.
Design: Retrospective cohort study.
Setting, methods: We analysed DonateLife audit data for patients aged 28 days to 80 years who died between July 2012 and December 2014 in an intensive care unit or emergency department, or who died within 24 hours of discharge from either, in the 75 Australian hospitals contributing data to DonateLife. Ideal and expanded organ donation criteria were derived from international and national guidelines, and from expert opinion. Potential DCD organ donors were identified by applying these criteria to patients who had been intubated and were neither confirmed as being brain-dead nor likely to have met brain death criteria at the official time of death.
Results: 8780 eligible patients were identified, of whom 202 were actual DCD donors. For 193 potential ideal (61%) and 313 potential expanded criteria DCD donors (72%), organ donation had not been discussed with their families; most were potential donors of kidneys (416 potential donors) or lungs (117 potential donors). Potential donors were typically older, dying of non-neurological causes, and more frequently had chronic organ disease than actual donors. Identifying all these potential donors, assuming a consent rate of 60%, would have increased Australia’s donation rate from 16.1 to 21.3 per million population in 2014.
Conclusions: The untapped potential for DCD in Australia, particularly of kidneys and lungs, is significant. Systematic review of all patients undergoing end-of-life care in critical care environments for donor suitability could result in significant increases in organ donation rates.