The nematode Angiostrongylus cantonensis has spread down the eastern coast of Australia over recent decades. A healthy 21-year-old man developed life-threatening eosinophilic meningoencephalitis following ingestion of a slug in Sydney. We describe the first case of this severity in which the patient survived.
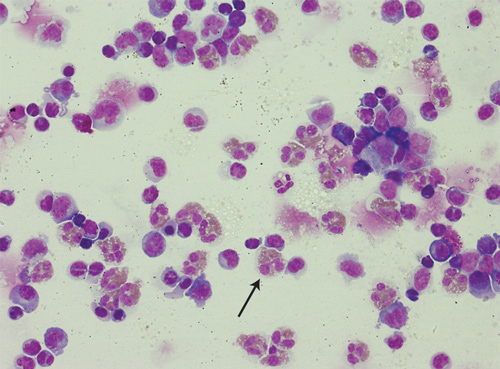
Progress computed tomography (CT) brain imaging results remained normal. His peripheral eosinophil count had risen, later peaking at 1.9 × 109/L on Day 24. A third lumbar puncture was performed. His CSF protein remained elevated at 0.71g/L, CSF red cell count was 216 × 109/L and CSF white cell count was 504 × 109/L. Specific staining for eosinophils was performed, showing 37% of the leukocytes to be eosinophils (RI, < 10%, Box 1).
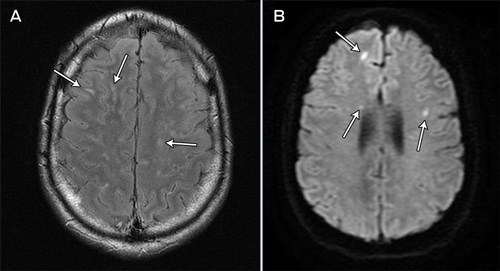
By this stage it had emerged that the patient had eaten a slug from a Sydney garden, as a dare, 7 days before presentation. An enzyme immunoassay for Angiostrongylus IgG performed on the CSF was positive. A progress MRI scan, performed on Day 26 after admission, revealed multiple foci of hyperintensity in the cerebral hemispheres, brainstem and cerebellum as well as within the spinal cord (Box 2). Several of the lesions showed restricted diffusion and some showed contrast enhancement. Pial enhancement was seen within the posterior fossa and over the spinal cord.
Angiostrongylus cantonensis, also known as the rat lungworm, is the most common cause of eosinophilic meningitis globally. This condition generally follows a benign, self-limited course.1 Rarely, the parasite causes meningoencephalitis, which should be considered a related but distinct clinical entity with a dramatically poorer prognosis. The mortality rate has been reported at 79%2 and, of patients who become comatose, at least 90% do not survive.3
A. cantonensis is endemic in South-East Asia and the Pacific region, and has spread down the eastern coast of Australia over the past 50 years.4 In Australia, it has been observed that cases tend to be particularly severe. This reflects the higher total larval load ingested from terrestrial hosts, which feed on rat faecal pellets harbouring thousands of larvae. In comparison, aquatic snails, which commonly cause the disease in South-East Asia, generally carry a smaller larval load.5,6 The first reported human case acquired in Sydney occurred in 2001,7 in the remarkably similar circumstances of a young man accepting a dare to eat a slug, highlighting the importance of specific questioning in the patient’s history. Our patient’s case is only the second reported case acquired in Sydney and, internationally, our patient is the first with the disease of this severity to have survived.
Recent investigations have sought to identify factors associated with the development of clinically severe angiostrongyliasis. In one study, clinical features including headache, abnormal CSF pressure and abnormal peripheral blood eosinophil count were associated with severe disease.8 An Activation Criteria for Angiostrongyliasis (ACA) scoring system, incorporating these factors, was proposed and validated in a population of Chinese patients, with a score of ≥ 7 predictive of severe disease. If we had used the presenting eosinophil count, our patient would only have had an ACA score maximum of 5, and most likely lower than this if his CSF opening pressure had been recorded at the first lumbar puncture. He would have scored 8 if his peak peripheral eosinophil count and his highest recorded CSF pressure had been used.
A second study investigated factors specifically associated with the development of the encephalitic form of the disease.2 In a cohort of 94 patients with angiostrongyliasis, of whom 14 developed encephalitis, it was found that the clinical factors predictive of encephalitis were temperature > 38°C at presentation, older age and longer duration of headache. Fever at presentation was associated with a remarkable 37-fold risk of encephalitis. Interestingly, other variables such as CSF opening pressure, peripheral or CSF eosinophil counts or paraesthesia were not predictive of encephalitis in this study. Our case indicates that caution should be used when applying the predictive factors reported in these studies, and suggests that peak eosinophil count and delayed CSF pressure results may be more useful when calculating the ACA.
The optimal treatment for Angiostrongylus meningoencephalitis remains poorly defined. Corticosteroids are commonly used, with the rationale of dampening the inflammatory reaction to the nematode, and have been shown in a double-blind, placebo-controlled trial to provide symptomatic relief in eosinophilic meningitis.9 However, studies of patients with the encephalitic form of the disease have not found corticosteroids to be effective.3 Anthelmintics are generally not used due to the theoretical possibility of exacerbating cerebral inflammation and damage as a result of larval death in the central nervous system (CNS), and the lack of evidence of their efficacy.9,10 We used albendazole when there was little to lose and, perhaps as expected, it did not lead to any appreciable benefit. In the absence of effective treatment of angiostrongyliasis, it is important in endemic areas that the public understand the small but very serious risks associated with ingestion of uncooked molluscs.
It is known that time spent in a minimally conscious state following traumatic brain injury does not correlate with the chance of functional recovery.11 This observation may extend to patients with diffuse brain injury caused by severe cerebral infection or inflammation. This case shows the potential for the CNS to recover following a severe, generalised insult in a young patient with supportive care. It is important for doctors to appreciate this capacity when wrestling with difficult decisions about continuation of care for critically unwell patients.
- 1. Wang QP, Lai DH, Zhu XQ, et al. Human angiostrongyliasis. Lancet Infect Dis 2008; 8: 621-630.
- 2. Sawanyawisuth K, Takahashi K, Hoshuyama T, et al. Clinical factors predictive of encephalitis caused by Angiostrongylus cantonensis. Am J Trop Med Hyg 2009; 81: 698-701.
- 3. Chotmongkol V, Sawanyawisuth K. Clinical manifestations and outcome of patients with severe eosinophilic meningoencephalitis presumably caused by Angiostrongylus cantonensis. Southeast Asian J Trop Med Public Health 2002; 33: 231-234.
- 4. Prociv P, Carlisle MS. The spread of Angiostrongylus cantonensis in Australia. Southeast Asian J Trop Med Public Health 2001; 32 Suppl 2: 126-128.
- 5. Cooke-Yarborough CM, Kornberg AJ, Hogg GG, et al. A fatal case of angiostrongyliasis in an 11-month-old infant. Med J Aust 1999; 170: 541-543.
- 6. Prociv P. Parasitic meningitis. Med J Aust 1999; 170: 517-518.
- 7. Pryor DS, Konecny P, Senanayake SN, Walker J. First report of human angiostrongyliasis acquired in Sydney. Med J Aust 2003; 179: 430-431. <MJA full text>
- 8. Diao Z, Xiao H, Wang J, et al. Identification of factors associated with clinically severe angiostrongyliasis. Am J Trop Med Hyg 2010; 83: 1352-1355.
- 9. Chotmongkol V, Sawanyawisuth K, Thavornpitak Y. Corticosteroid treatment of eosinophilic meningitis. Clin Infect Dis 2000; 31: 660-662.
- 10. Kliks MM, Kroenke K, Hardman JM. Eosinophilic radiculomyeloencephalitis: an angiostrongyliasis outbreak in American Samoa related to ingestion of Achatina fulica snails. Am J Trop Med Hyg 1982; 31: 1114-1122.
- 11. Lammi MH, Smith VH, Tate RL, Taylor CM. The minimally conscious state and recovery potential: a follow-up study 2 to 5 years after traumatic brain injury. Arch Phys Med Rehabil 2005; 86: 746-754.





We thank Dennis Hokin and Jennifer Posen at San Pathology, Sydney Adventist Hospital for assistance with histopathology.
No relevant disclosures.