During 2010, more than 47 000 hospitalisations for ST-segment-elevation myocardial infarction (STEMI) are expected in Australia.1 Morbidity and mortality will be significant — at 12 months, 8.9% will have had a repeat myocardial infarction or stroke and 8% will have died.2 The estimated economic cost of acute coronary syndromes (ACS) amounts to $17.9 billion, comprising $1.8 billion in direct hospital costs, $3.8 billion indirect costs, and $12.3 billion from loss in the value of health.1 Fortunately, trial evidence suggests that expeditious and effective reperfusion reduces morbidity and mortality.3
There is a dearth of recent information on local delivery of this proven life-saving treatment in Australia.4-6 Without data and international comparisons, it is difficult to address issues of evidence implementation at the national level and to identify priorities for local action.7
The ACACIA registry was a nationwide prospective audit of ACS conducted between 1 November 2005 and 31 July 2007.8 The registry involved 39 remote, rural, and metropolitan hospitals across all states and territories of Australia. Hospitals were asked to enrol 100–150 consecutive patients presenting with suspected ACS. Larger hospitals were presumed to have shorter, and smaller hospitals longer, recruitment periods. Ethics committee approval was obtained from each site. Consent to access medical records was obtained from all patients. For patients who died before giving consent, local ethics committees granted access to medical records.
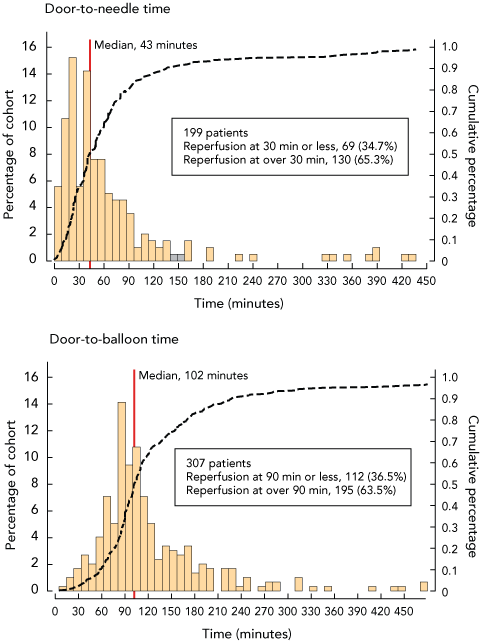
ACACIA focused on demographic, clinical procedural and logistical factors involved in the management of patients with ACS. In-hospital and 12-month outcomes were recorded. Late outcomes were documented by re-examining case notes, telephone interview, and examination of the National Death Index. Detailed descriptions of the clinical variables recorded have been previously published.8 Time to presentation was defined as time from symptom onset to first medical (doctor) contact. Time to reperfusion therapy was time from presentation until the time reperfusion therapy was administered, either by thrombolysis (door-to-needle), or primary percutaneous intervention (primary PCI; door-to-balloon). Reperfusion was considered timely if the patient received either thrombolysis within 30 minutes of presentation, or underwent angiography (and primary PCI if appropriate) within 90 minutes of presentation.9 Patients presenting to remote (17) and rural hospitals (114) were combined and referred to as “rural”.
Cox proportional hazards models accounting for the cluster design were used to determine outcomes by the type of reperfusion therapy and timeliness of reperfusion. Each model adjusted for individual calculated Global Registry of Acute Coronary Events (GRACE) risk score and factors significantly associated with use of reperfusion to adjust for baseline risk.10 Kaplan–Meier survival curves were plotted for use and timeliness of reperfusion. Statistical analysis was performed using Stata 11.0 (StataCorp, College Station, Tex, USA).
A total of 3402 patients were enrolled in the ACACIA registry, of whom 755 met the criteria of suspected STEMI at admission. Of these 755 patients, 707 (93.6%) were discharged with a final diagnosis of STEMI. Further, 684 patients (90.6%) presented within 12 hours of symptom onset. Demographic and clinical characteristics of the study cohort according to rural or metropolitan region are shown in Box 1. Overall, there were few differences between baseline characteristics of metropolitan and rural patient groups.
Door-to-needle and door-to-balloon times are detailed in Box 1 and Box 2. Timely reperfusion was achieved in 23.1% of the total population (174 patients), representing 34.5% of the 505 who were treated with any form of reperfusion therapy. For those treated with reperfusion therapy, there was no difference in the likelihood of timely reperfusion between rural and metropolitan patients for either the provision of primary PCI (7 of 23 rural patients [30.4%] v 98 of 284 metropolitan patients [34.5%]; P = 0.69) or thrombolysis (26 of 63 rural patients [41.3%] v 44 of 135 metropolitan patients [32.6%]; P = 0.23). Overall, angiography was performed on 671 of 755 patients (88.9%) during the initial admission; however, metropolitan patients were more likely than rural patients to receive angiography (575 of 624 metropolitan patients [92.2%] v 96 of 131 rural patients [73.3%]; P < 0.001). Stepwise forward logistic regression clustered by hospital site revealed the factors that were associated with use of reperfusion therapy, and the adjusted odds ratios for these factors are shown in Box 3. Box 4 shows the adjusted odds ratios for factors associated with timely reperfusion therapy for those who were treated with reperfusion.
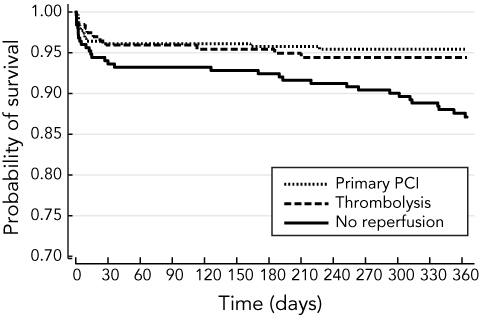
After adjusting for baseline risk using the GRACE score and adjusting for potential confounders, reperfusion of any kind was associated with a reduction in mortality throughout 12 months of follow-up (hazard ratio [HR], 0.44; 95% CI, 0.25–0.78; P < 0.01). This benefit remained evident for primary PCI compared individually with those receiving no reperfusion therapy (HR, 0.33; 95% CI, 0.18–0.61; P < 0.001), but not for thrombolysis (HR, 0.66; 95% CI, 0.26–1.68; P = 0.38; Box 5).
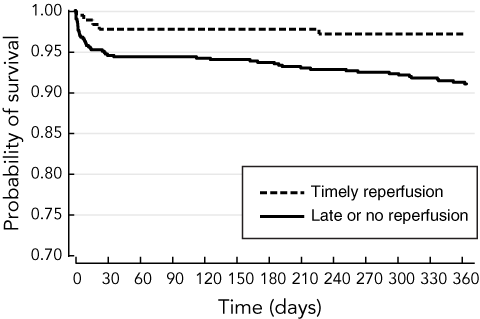
Timely reperfusion, compared with late or no reperfusion, was associated with a decrease in the odds of death throughout the 12-month follow-up period of 78.3% (HR, 0.22; 95% CI, 0.05–0.95; P = 0.04), even after adjustment by baseline GRACE score (Box 6). The difference in the combined rate of death, recurrent MI, and stroke throughout the 12 months did not reach statistical significance (HR, 0.66; 95% CI, 0.36–1.20, P = 0.17).
First, we have again observed the benefits of reperfusion therapy, which have previously been demonstrated in various randomised controlled trials and registries. Although there have been no randomised trials comparing any reperfusion therapy to no reperfusion therapy, it is known that 30-day mortality rates for medical treatment alone are around 13%, compared with 6%–7% for optimal thrombolytic therapy.11,12 Previous randomised controlled trial mortality rates were 3%–5% for optimal primary PCI.13,14 Of note, these results were from clinical trials where patient selection resulted in a lower overall risk profile. Our contemporary Australian 30-day mortality rate of 4.8% (36 of 755 patients) among those receiving reperfusion therapy is congruent with clinical trial mortality rates. When the modes of reperfusion are compared, mortality benefits persisted for primary PCI but not for thrombolysis. This is likely due to the small sample size, but is consistent with clinical trials demonstrating decreased mortality with primary PCI versus thrombolysis.15
Our second important finding relates to the contemporary patterns of use of reperfusion therapy in Australia. Although all patients in our cohort were suspected of presenting with STEMI on admission and 93.6% were discharged with a final diagnosis of STEMI, only 66.9% received any form of reperfusion therapy. This is despite a median time to presentation of only 105 minutes, and 90.6% of patients presenting within 12 hours of symptom onset. The fact that these rates are similar to those documented by various registries, with no improvement over the past two decades, is of concern.16,17 The GRACE registry (reporting on 1763 patients presenting with STEMI within 12 hours of symptom onset in 94 hospitals from 14 countries) found that 30% of eligible patients with STEMI did not receive reperfusion therapy.18 Therefore, despite the existence of guidelines, their implementation remains a challenge, and this evidence suggests that there is an ongoing need for strategies that facilitate this implementation.
Third, our study reconfirms, in a routine clinical practice setting, the advantages of early reperfusion. Contrasting rural and metropolitan populations reiterates this point. When these two groups were compared, there was no difference in long-term outcomes. Although the rates of reperfusion were similar in both groups, the rural group achieved that rate mainly through thrombolysis, while the metropolitan group achieved a similar rate from PCI. This underscores that time to reperfusion is more important than the modality of reperfusion. In this sense, our findings support current guideline recommendations.9 The Australian guidelines are slightly more specific than those of the American College of Cardiology (ACC) and American Heart Association (AHA) guidelines on this point,3 in that while patients presenting with STEMI should be considered for primary PCI if the time from first medical contact to balloon time is less than 90 minutes, an estimated time delay beyond 90 minutes should lead thrombolysis to be considered as the primary treatment.
A recent consensus recommendation from the National Heart Foundation for translating knowledge into action in ACS outlined specific recommendations and actions for early reperfusion for STEMI.19 These included empowering and supporting appropriately trained health care workers to initiate thrombolysis in regional and remote settings, to set up protocols for single-call activation of PCI systems that operate 24 hours a day, 7 days a week, and to optimise the use of existing infrastructure for reperfusion through regional collaborations and protocols. Each recommendation is backed by sound evidence,20,21 and early initiatives both in Australia22,23 and elsewhere have shown quantifiable outcome improvements.24
Various ST-segment criteria have been proposed for reperfusion therapy.25 Current Australian guidelines reflect the Minnesota Code and previous trials of thrombolysis, which classified STEMI as persistent ST-segment elevation ≥ 1 mm in two contiguous limb leads or ≥ 2 mm in two contiguous chest leads (or presumed new LBBB).26,27 Some more recent trials of thrombolysis and PCI have used ≥ 1 mm in any two contiguous leads, while the 2007 consensus document of the European Society of Cardiology, the ACC, the AHA and the World Heart Federation on the universal definition of myocardial infarction uses the Minnesota Code definition for men and changes the criteria for praecordial leads in women to ≥ 1.5 mm.28-30 In our study, 8.6% of all patients with suspected STEMI on admission had praecordial ST-segment elevation of 1–2 mm. Of these, 80% went on to have a discharge diagnosis of STEMI, and may therefore have benefitted from reperfusion therapy, which was only used in 26.2% of this group. A strategy of urgent coronary angiography with intervention when required in such patients with “borderline” ST-segment elevation may improve the rate of reperfusion in this group. In hospitals without PCI facilities, patients in this subgroup who are not considered candidates for thrombolysis initially may require close monitoring for progressive ECG changes or may need to be considered for transfer to a PCI-capable hospital where feasible.
Finally, we demonstrated factors associated with not using or delaying reperfusion therapy. Our findings add to the existing factors identified in other Australian cohort studies that included older age (≥ 70 years), past stroke, and past hypertension, and reinforces other factors such as diabetes and past ACS.31 Similarly, it was observed that more patient complexity was associated with less reperfusion. This is not surprising, given the greater uncertainty with respect to relative weighing of risk and benefit. Objective estimates of risk and benefit provided through decision support may help to improve outcomes.
Factors associated with not using reperfusion therapy are the same factors associated with greatest benefit from reperfusion. For example, patients who present with normal electrocardiograms and develop ST-segment elevation in hospital are often those who present very early in the course of their acute syndrome, which represents a “window of opportunity” in which a threatened STEMI may be treated before myocardial damage becomes irreversible.32 Similarly, among patients presenting with acute pulmonary oedema, data from both the SHOCK (Should We Emergently Revascualize Occluded Coronaries for Cardiogenic Shock) trial33 and the Duke registry34 suggest that these patients experience better inpatient and long-term outcomes when the infarct-related artery is reopened, either by thrombolysis or PCI. Although numbers in our study were small, about 70% of patients with LBBB on presentation were not reperfused despite an undisputed indication for therapy (in the presence of typical chest pain), representing one clear area where improvements in care may be made.
Treating unit specialty appeared to have an impact on use of reperfusion therapy. Similar findings have been previously reported with referral for coronary angiography.35 This finding has implications for rural regions, where access to specialist cardiac services are limited, and argues for widespread adaptation of clinical decision support systems,36 as well as specialist clinical networks in cardiology.37-39
The main limitation of our study was that it was observational by design. As such, it is suboptimal for estimating treatment effects. However, the data were not used to estimate new treatment effects, but simply to confirm previously robust clinical trial results. A second limitation was the limited representation of rural and remote patients. Census data indicate that roughly 14% of Australians live in rural and remote regions.40 Although these patients represented 17.4% of our cohort, this sample was derived from a few select centres. It is clear that future registries will need to focus more on country patients, and that data infrastructure should match clinical infrastructure.
1 Demographic and clinical characteristics of the 755 patients enrolled in the ACACIA registry who had suspected ST-segment-elevation myocardial infarction at admission to participating metropolitan and rural hospitals
Received 25 October 2009, accepted 11 June 2010
- Luan T Huynh1
- Jamie M Rankin2
- Phil Tideman3
- David B Brieger4
- Matthew Erickson2
- Andrew J Markwick3
- Carolyn Astley1,3
- David J Kelaher5
- Derek P B Chew3
- 1 Flinders University, Adelaide, SA.
- 2 Royal Perth Hospital, Perth, WA.
- 3 Flinders Medical Centre, Adelaide, SA.
- 4 University of Sydney and Concord Hospital, Sydney, NSW.
- 5 Sanofi-aventis Australia, Sydney, NSW.
The ACACIA study was sponsored by Sanofi-aventis Australia.
Luan Huynh, Derek Chew and David Brieger have in the past received modest speaker fees and travel assistance to attend academic meetings from Sanofi-aventis and its competitors. Sanofi-aventis had a representative on the steering committee and was given 30 days to review the article, but did not make any changes.
- 1. Access Economics. The economic costs of heart attack and chest pain (acute coronary syndrome). Sydney: Eli Lilly, 2009. http://www.accesseconomics.com.au/publicationsreports/showreport.php?id=204 (accessed Aug 2010).
- 2. Chew DP, Amerena JV, Coverdale SG, et al. Invasive management and late clinical outcomes in contemporary Australian management of acute coronary syndromes: observations from the ACACIA registry. Med J Aust 2008; 188: 691-697. <MJA full text>
- 3. Antman EM, Anbe DT, Armstrong PW, et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Revise the 1999 Guidelines for the Management of Patients with Acute Myocardial Infarction). Circulation 2004; 110: e82-292.
- 4. Birkhead JS, Walker L, Pearson M, et al. Improving care for patients with acute coronary syndromes: initial results from the National Audit of Myocardial Infarction Project (MINAP). Heart 2004; 90: 1004-1009.
- 5. Carruthers KF, Dabbous OH, Flather MD, et al. Contemporary management of acute coronary syndromes: does the practice match the evidence? The global registry of acute coronary events (GRACE). Heart 2005; 91: 290-298.
- 6. Gibson CM. NRMI and current treatment patterns for ST-elevation myocardial infarction. Am Heart J 2004; 148: S29-S33.
- 7. Scott IA. Why we need a national registry in interventional cardiology. Med J Aust 2008; 189: 223-227. <MJA full text>
- 8. Chew D, Amerena J, Coverdale S, et al. Current management of acute coronary syndromes in Australia: observations from the acute coronary syndromes prospective audit. Intern Med J 2007; 37: 741-748.
- 9. Acute Coronary Syndromes Working Group. Guidelines for the management of acute coronary syndromes 2006. Med J Aust 2006; 184 (8 Suppl): S9-29. <MJA full text>
- 10. Eagle KA, Lim MJ, Dabbous OH, et al. A validated prediction model for all forms of acute coronary syndrome: estimating the risk of 6-month postdischarge death in an international registry. JAMA 2004; 291: 2727-2733.
- 11. GUSTO Investigators. An international randomized trial comparing four thrombolytic strategies for acute myocardial infarction. N Engl J Med 1993; 329: 673-682.
- 12. Van de Werf F, Adgey J, Ardissino D, et al. Single-bolus tenecteplase compared with front-loaded alteplase in acute myocardial infarction: the ASSENT-2 double-blind randomised trial. Lancet 1999; 354: 716-722.
- 13. Cannon CP, Gibson CM, Lambrew CT, et al. Relationship of symptom-onset-to-balloon time and door-to-balloon time with mortality in patients undergoing angioplasty for acute myocardial infarction. JAMA 2000; 283: 2941-2947.
- 14. Stone GW, Grines CL, Cox DA, et al. Comparison of angioplasty with stenting, with or without abciximab, in acute myocardial infarction. N Engl J Med 2002; 346: 957-966.
- 15. Andersen HR, Nielsen TT, Rasmussen K, et al. A comparison of coronary angioplasty with fibrinolytic therapy in acute myocardial infarction. N Engl J Med 2003; 349: 733-742.
- 16. Rogers WJ, Bowlby LJ, Chandra NC, et al. Treatment of myocardial infarction in the United States (1990 to 1993). Observations from the National Registry of Myocardial Infarction. Circulation 1994; 90: 2103-2114.
- 17. French WJ. Trends in acute myocardial infarction management: use of the National Registry of Myocardial Infarction in quality improvement. Am J Cardiol 2004; 85: 5B-9B.
- 18. Eagle KA, Goodman SG, Avezum A, et al. Practice variation and missed opportunities for reperfusion in ST-segment-elevation myocardial infarction: findings from the Global Registry of Acute Coronary Events (GRACE). Lancet 2002; 359: 373-377.
- 19. Brieger D, Kelly A, Aroney C, et al. Acute coronary syndromes: consensus recommendations for translating knowledge into action. Med J Aust 2009; 191: 334-338. <MJA full text>
- 20. Terkelsen CJ, Nørgaard BL, Lassen JF, et al. Potential significance of spontaneous and interventional ST-changes in patients transferred for primary percutaneous coronary intervention: observations from the ST-MONitoring in Acute Myocardial Infarction study (The MONAMI study). Eur Heart J 2006; 27: 267-275.
- 21. Carstensen S, Nelson GC, Hansen PS, et al. Field triage to primary angioplasty combined with emergency department bypass reduces treatment delays and is associated with improved outcome. Eur Heart J 2007; 28: 2313-2319.
- 22. Victorian Government Department of Health. Safer systems, saving lives. Improving care for acute myocardial infarction. http://www.health.vic.gov.au/sssl/interventions/acute.htm (accessed Aug 2010).
- 23. Queensland Health. Statewide cardiac clinical network. http://www.health.qld.gov.au/cpic/service_improve/clin_net_cardiac.asp (accessed Aug 2010).
- 24. Krumholz HM, Bradley EH, Nallamothu BK, et al. A campaign to improve the timeliness of primary percutaneous coronary intervention: Door-to-balloon: an alliance for quality. JACC Cardiovasc Interv 2008; 1: 97-104.
- 25. Menown IB, Mackenzie G, Adgey AA. Optimizing the initial 12-lead electrocardiographic diagnosis of acute myocardial infarction. Eur Heart J 2000; 21: 275-283.
- 26. Blackburn H, Keys A, Simonson E, et al. The electrocardiogram in population studies: a classification system. Circulation 1960; 21: 1160-1175.
- 27. Effectiveness of intravenous thrombolytic treatment in acute myocardial infarction. Gruppo Italiano per lo Studio della Streptochinasi nell'Infarto Miocardico (GISSI). Lancet 1986; 1: 397-402.
- 28. Grines CL, Cox DA, Stone GW, et al. Coronary angioplasty with or without stent implantation for acute myocardial infarction. Stent Primary Angioplasty in Myocardial Infarction Study Group. N Engl J Med 1999; 341: 1949-1956.
- 29. Montalescot G, Barragan P, Wittenberg O; ADMIRAL Investigators. Platelet glycoprotein IIb/IIIa inhibition with coronary stenting for acute myocardial infarction. N Engl J Med 2001; 344: 1895-1903.
- 30. Thygesen K, Alpert JS, White HD, et al. Universal definition of myocardial infarction. Circulation 2007; 116: 2634-2653.
- 31. Scott IA, Derhy PH, O’Kane D, et al. Discordance between level of risk and intensity of evidence-based treatment in patients with acute coronary syndromes. Med J Aust 2007; 187: 153-159. <MJA full text>
- 32. Verheugt FW, Gersh BJ, Armstrong PW. Aborted myocardial infarction: a new target for reperfusion therapy. Eur Heart J 2006; 27: 901-904.
- 33. Wong SC, Sanborn T, Sleeper LA, et al. Angiographic findings and clinical correlates in patients with cardiogenic shock complicating acute myocardial infarction: a report from the SHOCK Trial Registry. J Am Coll Cardiol 2000; 36: 1077-1083.
- 34. Bengtson JR, Kaplan AJ, Pieper KS, et al. Prognosis in cardiogenic shock after acute myocardial infarction in the interventional era. J Am Coll Cardiol 1992; 20: 1482-1489.
- 35. Borowsky SJ, Kravitz RL, Laouri M, et al. Effect of physician specialty on use of necessary coronary angiography. J Am Coll Cardiol 1995; 26: 1484-1491.
- 36. Kawamoto K, Houlihan CA, Balas EA, Lobach DF. Improving clinical practice using clinical decision support systems: a systematic review of trials to identify features critical to success. BMJ 2005; 330: 765.
- 37. Jong P, Gong Y, Liu PP, et al. Care and outcomes of patients newly hospitalized for heart failure in the community treated by cardiologists compared with other specialists. Circulation 2003; 108: 184-191.
- 38. Jollis JG, DeLong ER, Peterson ED, et al. Outcome of acute myocardial infarction according to the specialty of the admitting physician. N Engl J Med 1996; 335: 1880-1887.
- 39. Goodacre S, Nicholl J, Dixon S, et al. Randomised controlled trial and economic evaluation of a chest pain observation unit compared with routine care. BMJ 2004; 328: 254.
- 40. Australian Bureau of Statistics. Measures of Australia’s progress, 2004. Population distribution. Canberra: ABS, 2004. (ABS Cat. No. 1370.0) http://www.abs.gov.au/Ausstats/abs@.nsf/94713ad445ff1425ca25682000192af2/282e34ea888d4eccca256e7d0000263e!OpenDocument (accessed Jul 2009).





Abstract
Objective: To describe the contemporary management and outcomes of patients presenting with ST-segment-elevation myocardial infarction (STEMI) in Australia.
Design, participants and setting: Observational analysis of data for patients who presented with suspected STEMI and enrolled in the Australian Acute Coronary Syndrome Prospective Audit from 1 November 2005 to 31 July 2007.
Main outcome measures: Factors associated with use of reperfusion therapy and timely use of reperfusion therapy, and the effects of reperfusion on mortality.
Results: In total, 755 patients had suspected STEMI. Median time to presentation was 105 minutes (IQR, 60–235 minutes). Reperfusion therapy was used in 66.9% of patients (505/755), and timely reperfusion therapy in 23.1% (174/755). Thombolysis was administered in 39.2% of those who received reperfusion therapy (198/505), while 60.8% (307/505) received primary percutaneous intervention. Cardiac arrest (OR, 2.83; P = 0.001) and treatment under the auspices of a cardiology unit (OR, 2.14; P = 0.02) were associated with use of reperfusion therapy. A normal electrocardiogram on presentation (OR, 0.42; P = 0.01), left bundle branch block (OR, 0.18; P = 0.001), acute pulmonary oedema (OR, 0.34; P < 0.01), history of diabetes (OR, 0.54; P < 0.01), and previous lesion on angiogram of > 50% (OR, 0.51; P = 0.001) were associated with not using reperfusion. Inhospital mortality was 4.0% (30/755), mortality at 30 days was 4.8% (36/755), and mortality at 1 year was 7.8% (59/755). Receiving reperfusion therapy of any kind was associated with decreased 12-month mortality (hazard ratio [HR], 0.44; 95% CI, 0.25–0.78; P < 0.01). Timely reperfusion was associated with a reduction in mortality of 78% (HR, 0.22; P = 0.04). There were no significant differences in early and late mortality in rural patients compared with metropolitan patients (P = 0.66).
Conclusion: Timely reperfusion, not the modality of reperfusion, was associated with significant outcome benefits. Australian use of timely or any reperfusion remains poor and incomplete.