There is a substantial body of literature supporting the view that access to health compensation — notably health care cover and income support — is associated with poor recovery after injury.1-5 While these findings are disturbing, it must be recognised that most of these studies are based on a workers compensation population. It may be that the relationship between work-related injury and health outcomes is mediated by factors other than compensation,6 and these factors may or may not be relevant to other compensation populations.
In one of the few studies to examine the relationship between compensation and recovery in a non-workers-compensation sample, Gabbe and colleagues7 studied a large group of orthopaedic patients in a “no-fault” motor vehicle accident (MVA) compensation scheme. After controlling for differences between groups in factors such as age, injury severity, head injury status, injury group and discharge destination, the study found that compensable patients were more likely than non-compensable patients to report moderate-to-severe disability in both physical and mental health domains at 12 months after injury. Compensable patients were also less likely to have returned to work at 12 months. Thus, the study appears to support the notion that access to compensation results in poorer health outcomes.
The aim of our study was to investigate the effect of access to MVA compensation on recovery outcomes at 24 months after injury. We sought to replicate and extend the study by Gabbe et al7 and other studies by:
Comparing health outcomes of injury patients covered by the Victorian MVA compensation scheme with those of non-compensable patients. We used two different methods for identifying eligibility for compensation: examination of medical records (using a similar method to that of Gabbe et al7), and asking patients at 24-month follow-up to identify any compensation scheme they had accessed;
- Controlling for many factors that could contribute to between-group differences, including pre-injury disability, pre-injury quality of life, prior trauma, psychiatric history, demographic factors, acute psychological response to injury, and injury characteristics; and
- Identifying whether interactions with the compensation agency affected recovery outcomes.
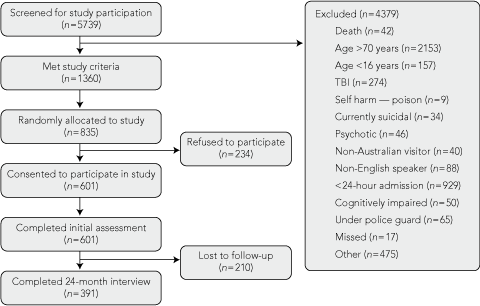
A total of 835 patients were randomly selected for our study, and written consent was obtained from 601 patients. Interview and self-report data were collected from all 601 patients just before discharge. Follow-up telephone assessments were conducted 24 months after the injury. With 210 participants lost to follow-up, there were 391 patients (65%) who completed all assessments (Box 1).
The initial assessment was conducted about 1 week after admission (mean, 7.0 days [SD, 7.0 days]). Clinical researchers administered the Mini-International Neuropsychiatric Interview to assess patients’ psychiatric history.8 Patients then completed a self-report written questionnaire booklet that assessed:
demographic characteristics;
pre-injury quality of life (World Health Organization Quality of Life–Bref [WHOQOL-Bref]);9
pre-injury disability (12-item World Health Organization Disability Assessment Schedule II [WHODAS II]);10
peritrauma distress (Acute Stress Disorders Interview);11
trauma history (Composite International Diagnostic Interview);12
pain intensity at time of assessment (Visual Analogue Scale);13,14 and
acute anxiety and depression severity (Hospital Anxiety and Depression Scale [HADS]).15
Twenty-four months after the injury, participants were contacted by telephone. They were first asked to identify any compensation agency they had accessed since discharge from hospital (including a private health insurance organisation). Participants were asked questions relating to compensation, including their level of stress in dealing with the compensation agency. To assess the severity of post-traumatic stress disorder (PTSD), clinical researchers then administered the Clinician Administered PTSD Scale for DSM-IV (CAPS).16 Participants were also sent self-report booklets assessing quality of life (using the WHOQOL-Bref), disability (using the WHODAS II), and acute anxiety and depression (using the HADS). Participants were also asked if they had returned to work, and if so, whether they had returned to their pre-injury number of work hours.
Baseline characteristics were compared between compensation groups using χ2 tests for dichotomous variables and t-tests for continuous variables. Established cut-off thresholds were used on outcome variables (HADS and CAPS) to create dichotomous variables. Quality-of-life variables were dichotomised using community norms.17 To assess the relationship between compensation and patient outcomes, baseline variables in which significant between-group differences were identified (P < 0.10) were entered as the first step into a binary logistic regression model, with compensation status (MVA-compensable v non-compensable) as the second step. Adjusted odds ratios (AORs) with 95% confidence intervals were calculated. All analyses were conducted using SPSS software, version 17 (SPSS Inc, Chicago, Ill, USA).
The mean age of participants was 39.1 years (SD, 13.4 years). Over two-thirds of participants who completed the study were male (n = 274 [70%]). The mean Injury Severity Score18 was 12.12 (SD, 8.40), indicating that the average injury was of moderate severity. Participants spent a mean of 10.7 days (SD, 9.7 days) in hospital, and 53 patients (14%) required admission to the intensive care unit. Almost half the patients (172 [45%]) met the criteria for mild traumatic brain injury.19 The principal cause of injury was MVAs (284 patients [73%]). Other causes were falls (50 patients [13%]), assault (27 patients [7%]), a non-compensable work injury (one patient [< 1%]), and other accidents (29 patients [7%]). One hundred and seventeen patients (30%) were discharged to a rehabilitation facility and the rest were discharged home.
Post-admission information collected from the medical records of participants who completed the study revealed that 246 patients (63%) were MVA-compensable (under the TAC scheme) and 145 (37%) were non-compensable. Demographic characteristics, pre-injury profiles and injury characteristics for each compensation group are shown in Box 2.
After controlling for factors for which the differences between groups were significant at the 0.1 level (Box 2), MVA-compensable patients were significantly more likely than non-compensable patients to have PTSD (AOR, 2.51 [95% CI, 1.01–6.28]; P = 0.05), depression (AOR, 2.63 [95% CI, 1.14–6.01]; P = 0.02) and anxiety (AOR, 2.24 [95% CI, 1.08–4.63]; P = 0.03) at 24 months after injury. They were also less likely to have returned to their pre-injury number of work hours (AOR, 0.47 [95% CI, 0.27–0.81]; P = 0.006). They did not differ on other variables such as quality of life, disability or return-to-work status.
Differences in demographics and in pre-injury and post-injury variables between the MVA group and the no-compensation group (with other compensation groups removed) are shown in Box 3. The health outcomes at 24 months for these two groups are summarised in Box 4.
After controlling for factors for which the differences between groups were significant at the 0.1 level (Box 4), MVA-compensable patients were significantly more anxious at 24 months than the no-compensation patients (AOR, 2.79 [95% CI, 1.17–6.66]; P = 0.02) (Box 5). They did not differ significantly on any other variable, including PTSD, depression, disability, quality of life, return to work, or return to pre-injury work hours.
In an effort to identify factors that may have contributed to the MVA-compensable group being more anxious than the no-compensation group, we investigated whether stressful interactions with the compensation agency may have played a role. Fifty-two patients (21%) who had accessed MVA compensation reported that they found the process stressful. We conducted a binary logistic regression analysis, controlling for differences between groups at the 0.1 level (Box 3) and for stressful interactions with compensation agencies. There was no significant difference in anxiety between MVA-compensable and no-compensation groups after controlling for stressful interactions with compensation agencies (AOR, 2.04 [95% CI, 0.83–5.01]; P = 0.14) (Box 6).
Initially our findings appeared to replicate those of Gabbe and colleagues7 in suggesting that access to a no-fault MVA compensation scheme was associated with poor health outcomes. When we looked at eligibility for compensation based on hospital medical records, we found that patients who were MVA-compensable had poorer mental health outcomes at 24 months than those who were non-compensable, and were also less likely to have returned to their pre-injury work level.
2 Demographic characteristics and pre-injury status of participants in each compensable group, as identified by medical records
3 Demographic characteristics and pre-injury status of participants in each compensable group, as identified by participants at 24 months
5 Results of a stepwise logistic regression analysis comparing the MVA-compensable group with the non-compensable group with respect to anxiety levels at 24 months
- Meaghan L O’Donnell1,2
- Mark C Creamer1,2
- Alexander C McFarlane3
- Derrick Silove4
- Richard A Bryant5
- 1 Department of Psychiatry, University of Melbourne, Melbourne, VIC.
- 2 Australian Centre for Posttraumatic Mental Health, Melbourne, VIC.
- 3 Centre for Military and Veterans’ Health, Adelaide, SA.
- 4 Department of Psychiatry, University of New South Wales, Sydney, NSW.
- 5 Department of Psychology, University of New South Wales, Sydney, NSW.
Our study was supported by a Victorian Trauma Foundation general grant, a National Health and Medical Research Council (NHMRC) Australian Clinical Research Fellowship and an NHMRC program grant. We gratefully acknowledge all the participants in our study.
None identified.
- 1. Harris I, Mulford J, Solomon M, et al. Association between compensation status and outcome after surgery: a meta-analysis. JAMA 2005; 293: 1644-1652.
- 2. Binder LM, Rohling ML. Money matters: a meta-analytic review of the effects of financial incentives on recovery after closed-head injury. Am J Psychiatry 1996; 153: 7-10.
- 3. Coste J, Lefrançois G, Guillemin F, Pouchot J; French Study Group for Quality of Life in Rheumatology. Prognosis and quality of life in patients with acute low back pain: insights from a comprehensive inception cohort study. Arthritis Rheum 2004; 51: 168-176.
- 4. Zelle BA, Panzica M, Vogt MT, et al. Influence of workers’ compensation eligibility upon functional recovery 10–28 years after polytrauma. Am J Surg 2005; 190: 30-36.
- 5. Rohling ML, Binder LM, Langhinrichsen-Rohling J. Money matters: a meta-analytic review of the association between financial compensation and the experience and treatment of chronic pain. Health Psychol 1995; 14: 537-547.
- 6. Mason S, Wardrope J, Turpin G, Rowlands A. Outcomes after injury: a comparison of workplace and nonworkplace injury. J Trauma 2002; 53: 98-103.
- 7. Gabbe BJ, Cameron PA, Williamson O, et al. The relationship between compensable status and long-term patient outcomes following orthopaedic trauma. Med J Aust 2007; 187: 14-17. <MJA full text>
- 8. Sheehan DV, Lecrubier Y, Harnett-Sheehan K, et al. The Mini-International Neuropsychiatric Interview (MINI): the development and validation of a structured diagnostic psychiatric interview. J Clin Psychiatry 1998; 59 Suppl 20: 22-33.
- 9. World Health Organization Quality of Life Group. WHOQOL-BREF. Introduction, administration, scoring and generic version of the assessment. Geneva: WHO, 1996.
- 10. World Health Organization Disability Assessment Schedule Group. WHODAS Schedule II. Geneva: WHO, 2000.
- 11. Bryant RA, Harvey AG, Dang ST, Sackville T. Assessing acute stress disorder: psychometric properties of a structured clinical interview. Psychol Assess 1998; 10: 215-220.
- 12. World Health Organization Collaborating Centre for Mental Health and Substance Abuse. Composite International Diagnostic Interview, CIDI-Auto 2.1. Administrator’s guide and reference. Sydney: WHO Collaborating Centre for Mental Health and Substance Abuse, 1997.
- 13. Scott JC, Huskisson EC. Graphic representation of pain. Pain 1976; 2: 175-184.
- 14. Huskisson EC. Measurement of pain. Lancet 1974; 2: 1127-1131.
- 15. Zigmond A, Snaith R. The hospital anxiety and depression scale. Acta Psychiatr Scand 1983; 67: 361-370.
- 16. Blake DD, Weathers FW, Nagy LM, et al. Clinician-administered PTSD Scale for DSM-IV. Boston: National Center For Posttraumatic Stress Disorder, 1998: 1-19.
- 17. Hawthorne G, Herrman H, Murphy B. Interpreting the WHOQOL-BREF: preliminary population norms and effect sizes. Soc Indic Res 2006; 77: 37-59.
- 18. Baker SP, O’Neil B, Haddon W, Long WB. The injury severity score: a method for describing patients with multiple injuries and evaluating emergency care. J Trauma 1974; 14: 187-196.
- 19. American Congress of Rehabilitation Medicine. Definition of mild traumatic brain injury. J Head Trauma Rehabil 1993; 8: 86-87.





Abstract
Objective: To conduct a descriptive study investigating the effect of access to motor vehicle accident (MVA) compensation on recovery outcomes at 24 months after injury.
Design and setting: Longitudinal cohort study conducted in two Level 1 trauma hospitals in Victoria, Australia. Participants were 391 randomly selected injury patients with moderate-to-severe injuries. Compensable and non-compensable patients were compared at 24 months after injury on a number of health outcomes.
Main outcome measures: Health outcomes at 24 months, including anxiety and depression severity, quality of life and disability.
Results: Medical records identified two groups of compensation patients: MVA-compensable and non-compensable patients. After controlling for baseline variables, the MVA-compensable patients, at 24 months, had higher levels of post-traumatic stress disorder, anxiety and depression, and were less likely to have returned to their pre-injury number of work hours. However, some patients in the non-compensable group had accessed other forms of compensation (eg, private health care or compensation for victims of crime). When these were removed from the non-compensable group, the differences between MVA-compensable and non-compensable groups all but disappeared.
Conclusion: Our findings do not support previous research showing that access to compensation is associated with poor recovery outcomes. The relationship between access to compensation and health outcomes is complex, and more high-level research is required.