Primary percutaneous coronary intervention (PCI) is the preferred strategy for reperfusion in patients with ST-elevation myocardial infarction (STEMI).1,2 The use of PCI for STEMI has been widely embraced in Australia. Over 23 randomised controlled trials have provided Level I, class A evidence that PCI is superior to fibrinolysis for management of STEMI, and time to reperfusion is directly related to mortality.1,2
Current American and European guidelines recommend a “door-to-balloon” time (see below) of ≤ 90 minutes for patients with STEMI.3,4 For patients presenting to a non-PCI-capable unit, the European guidelines also recommend PCI if it can be performed within 120 minutes of first medical contact.4
Unlike American and European guidelines, the Cardiac Society of Australia and New Zealand/National Heart Foundation of Australia (CSANZ/NHFA) guidelines for management of patients with STEMI5 are centred around “symptom-to-door” time (see below) based on evidence from two multicentre randomised trials.6,7 The CSANZ/NHFA guidelines recommend PCI over fibrinolysis, if (i) the time from symptom onset to presentation is ≤ 60 minutes and PCI is available in ≤ 60 minutes; or (ii) the time from symptom onset to presentation is > 60 minutes and PCI is available in ≤ 90 minutes. For patients with STEMI presenting to a non-PCI-capable hospital, the guidelines recommend a first-door-to-balloon time of ≤ 120 minutes (including transfer time), if the time from symptom onset to presentation is between 3 and 12 hours.5 The DANAMI-2 (Danish Multicenter Randomized Study of Fibrinolysis versus Primary Angioplasty in Acute Myocardial Infarction-2) trial showed that 30-day clinical outcomes were superior for patients transferred to a PCI site from a peripheral hospital compared with patients who received onsite fibrinolysis, despite the time delay to reperfusion (median door-to-balloon time, 118 minutes).8 The feasibility and outcomes for such transfer programs have not been formally tested in Australia. We report our 6-year experience of onsite PCI and transfer for PCI. Our aim was to determine whether the current CSANZ/NHFA guidelines could be met effectively in a real hospital situation.
The following definitions were used:
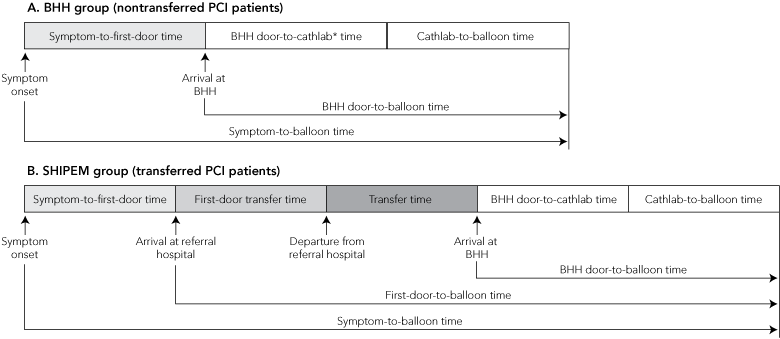
Symptom-to-first-door time. The time from symptom onset to arrival at the first hospital (Box 1).
First-door-to-balloon time. The time from arrival at the first hospital to inflation of the angioplasty balloon.
Symptom-to-balloon time. The time from symptom onset to inflation of angioplasty balloon.
Procedural success. Less than 30% residual stenosis, with no procedural complications (death, recurrent myocardial infarction [MI] or emergency coronary artery bypass graft [CABG] surgery).9
Major adverse cardiac event (MACE). The composite of mortality, recurrent MI and target vessel revascularisation (TVR).
Recurrent myocardial infarction. The recurrence of chest pain for ≥ 30 minutes, with new ST elevation, or a rise in creatine phosphokinase (to three or more times the upper limit of normal or to at least 50% higher than the pre-PCI level).
Target vessel revascularisation. PCI or CABG involving the target vessel.
Other procedural complications included haemorrhage (defined according to TIMI [thrombolysis in myocardial infarction] criteria),10 stroke, CABG, cardiogenic shock and groin complications.
Following coronary angiography, PCI was performed. If the patient was unsuitable for PCI, they were referred for CABG or treated medically. All patients received a loading dose of aspirin and clopidogrel before or during PCI. Use of glycoprotein IIb/IIIa inhibitors or a thrombus aspiration device was at the discretion of the interventional cardiologist. Drug-eluting stents were used in patients at high risk of restenosis according to Victorian Department of Human Services criteria.11 Duration of clopidogrel treatment was at the discretion of the cardiologist.
The BHH patients were significantly older than the SHIPEM patients and had a higher prevalence of hypercholesterolaemia (Box 2). There was no difference in procedural details between the groups (Box 3), and no patients required “bail-out” CABG for a procedural complication.
Fewer than a third of patients in either group presented within 60 minutes of symptom onset: 180 of 598 BHH patients (30.1%), and 48 of 189 SHIPEM patients (25.4%) (P = 0.214). The majority of patients presented within 3 hours: 484 of 598 (80.9%) BHH patients and 139 of 189 SHIPEM patients (73.5%) (P = 0.029). Patients in the SHIPEM group had significantly longer median symptom-to-first-door, first-door-to-balloon and symptom-to-balloon times than the BHH patients (Box 4). Median first-door-to-balloon times did not change significantly over the 6-year study period (data not shown). The median transfer time for the SHIPEM group was 36 minutes (interquartile range 25–48 minutes).
There was no significant difference in 30-day mortality, recurrent MI, TVR or MACE between the BHH and SHIPEM groups. There were also no differences in bleeding, stroke or groin complications. There was no significant difference in 12-month mortality, MI, TVR or MACE for BHH and SHIPEM patients. Six patients died of non-cardiac causes within 12 months in the BHH group and none in the SHIPEM group (Box 5).
Our 6-year registry with 12-month follow-up of PCI in both onsite and transferred patients demonstrates that transfer for PCI is safe and feasible for selected patients, and produces outcomes comparable with those of patients who receive onsite PCI, despite longer door-to-balloon times. These SHIPEM study results may reflect a selection bias towards better outcomes, which may offset the negative effect of longer door-to-balloon times. Our findings are comparable with those of other large randomised controlled trials.1
Timely access to PCI for patients presenting to hospitals without PCI facilities remains a challenge in real hospital settings. The DANAMI-2 study demonstrated that, in a coordinated trial with a 30-day combined end point of death, re-infarction and stroke for patients transferred for PCI, with a median door-to-balloon time of 118 minutes, PCI was superior to onsite fibrinolysis.8 Our SHIPEM patient treatment times were comparable to those in the DANAMI-2 trial, with a median first-door-to-balloon time of 128 minutes, despite not having the same dedicated transfer process. A meta-analysis of five randomised controlled trials also reported that transfer for PCI achieved a 42% reduction in death, re-infarction and stroke when compared with onsite fibrinolysis.12
Major trials have shown a correlation between door-to-balloon times and 30-day outcomes following PCI.1 The American and European guidelines specify a maximum door-to-balloon time of 90 minutes for onsite PCI,3,4 but the CSANZ/NHFA guidelines stratify their recommendations on symptom-to-door times.5 Our results demonstrate that the proportion of patients presenting early is low, and that in a real hospital situation, the CSANZ/NHFA guidelines may not be attainable. The guidelines also only include recommendations for door-to-balloon times for transferred patients with symptom-to-door times of 3–12 hours.5
The American College of Cardiology Door-to-Balloon Alliance suggests that centres aim for ≥ 75% of patients achieving a door-to-balloon time of ≤ 90 minutes, but does not address the issue of transfer.13 In the BHH group in our study, 52.1% of patients achieved a door-to-balloon time of ≤ 90 minutes. This compares with US registry data demonstrating that 40% of nontransferred and 5% of transferred STEMI patients achieve a door-to-balloon time of ≤ 90 minutes.14-16 The median transfer time for SHIPEM patients was 36 minutes, and the DANAMI-2 trial demonstrated that PCI was the preferred reperfusion strategy, provided transfer times were ≤ 120 minutes.8 Only 13.9% of the SHIPEM group achieved a first-door-to-balloon time of ≤ 90 minutes. European guidelines recommend that transferred patients receive PCI in ≤ 120 minutes,4 and 43.3% of the SHIPEM patients achieved a first-door-to-balloon time of ≤ 120 minutes.
Reasons for delay in transfer of SHIPEM patients included assessment and referral for transfer from the non-PCI-capable hospital, waiting for the ambulance to arrive and travel time. These delays are somewhat offset by the shorter door-to-balloon times at the PCI centre for SHIPEM patients, who bypass the emergency department. Delays are also offset by activating the catheterisation laboratory team during transfer. Possible strategies to improve door-to-balloon time include a government policy and funding for the ambulance service, ambulance field triage, and “live-in” 24-hour catheterisation laboratory teams.13 Reducing symptom-to-door time also requires increased public awareness and education.
These factors may explain why there was no improvement over time in door-to-balloon times for either the BHH or the SHIPEM group, despite publication of the CSANZ/NHFA guidelines.5 Ambulance field triage (with prehospital ECG) and by-passing non-PCI-capable centres are strategies currently being implemented in metropolitan Melbourne. These strategies are based on two pilot studies in New South Wales17 and Victoria.18 It would not be feasible to establish PCI facilities in all hospitals, because of resource, financial and geographical constraints. There will always remain a need for fibrinolysis in rural and remote areas in Australia. A recent trial has shown the benefits of a strategy of fibrinolysis followed by early (≤ 6 hours) transfer for PCI.19
Our study represents a local experience, illustrating the feasibility and safety of transferring patients from non-PCI-capable hospitals to a larger centre with PCI facilities. Although our clinical outcomes were acceptable, there remains significant room for improvement in door-to-balloon time and symptom-to-door time. Strategies to improve these require ongoing research, government-driven policy, public health awareness and a geographically tailored approach.20
- Michael J Moore1
- Louise Roberts1
- Houng-Bang Liew2
- Esther M Briganti3
- Gishel New1
- 1 Department of Cardiology, Box Hill Hospital, Melbourne, VIC.
- 2 Queen Elizabeth Hospital, Sabah, Malaysia.
- 3 Epworth Hospital, Melbourne, VIC.
We would like to thank Yew Mun Cheong, Dharsh Fernando, Chris Goods, George Proimos, Michael Rowe, Wayne Childs, Sue Ling Ching, Jennifer Coller, Bon-Chun Chou, Pallav Garg, Chris Lim, Kang-Teng Lim, Edward Mah, Swati Mukherjee, Vicki Pandeli, James Sapontis, Matthew Swale and Andrew Teh for their involvement in procedures and data collection.
Esther Briganti has received speaker fees from Novo Nordisk and Merck Sharp & Dohme, and an educational grant from Novo Nordisk. These are not related to issues covered in this article.
- 1. Keeley EC, Boura JA, Grines CL. Primary angioplasty versus intravenous thrombolytic therapy for acute myocardial infarction: a quantitative review of 23 randomised trials. Lancet 2003; 361: 13-20.
- 2. Masoudi FA, Bonow RO, Brindis RG, et al. ACC/AHA 2008 statement on performance measurement and reperfusion therapy: a report of the ACC/AHA Task Force on Performance Measures (work group to address the challenges of performance measurement and reperfusion therapy). Circulation 2008; 118: 2649-2661.
- 3. Antman EM, Hand M, Armstrong PW, et al. 2007 focused update of the ACC/AHA 2004 guidelines for the management of patients with ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines: developed in collaboration with the Canadian Cardiovascular Society endorsed by the American Academy of Family Physicians: 2007 writing group to review new evidence and update the ACC/AHA 2004 guidelines for the management of patients with ST-elevation myocardial infarction, writing on behalf of the 2004 writing committee. Circulation 2008; 117: 296-329.
- 4. Van de Werf F, Bax J, Betriu A, et al. Management of acute myocardial infarction in patients presenting with persistent ST-segment elevation: the task force on the management of ST-segment elevation acute myocardial infarction of the European Society of Cardiology. Eur Heart J 2008; 29: 2909-2945.
- 5. Acute Coronary Syndrome Guidelines Working Group. Guidelines for the management of acute coronary syndromes 2006. Med J Aust 2006; 184 (8 Suppl): S1-S32. <MJA full text>
- 6. Bonnefoy E, Lapostolle F, Leizorovicz A, et al. Primary angioplasty versus prehospital fibrinolysis in acute myocardial infarction: a randomised study. Lancet 2002; 360: 825-829.
- 7. Widimsk P, Groch L, Zelizko M, et al. Multi-centre randomized trial comparing transport to primary angioplasty vs immediate thrombolysis vs combined strategy for patients with acute myocardial infarction presenting to a community hospital without a catheterization laboratory. The PRAGUE study. Eur Heart J 2000; 21: 823-831.
- 8. Andersen HR, Nielsen TT, Rasmussen K, et al. A comparison of coronary angioplasty with fibrinolytic therapy in acute myocardial infarction. N Engl J Med 2003; 349: 733-742.
- 9. Smith SC Jr, Feldman TE, Hirshfeld JW Jr, et al. American College of Cardiology/American Heart Association/Society for Cardiovascular Angiography and Interventions 2005 guideline update for percutaneous coronary intervention: a report of the ACC/AHA Task Force on Practice Guidelines (ACC/AHA/SCAI Writing Committee to update 2001 guidelines for percutaneous coronary intervention). Circulation 2006; 113: e166-e286.
- 10. Gutierrez MJ, Aggarwal A, Gilbert K, et al. Bleeding complications after contemporary pharmacoinvasive therapy for ST elevation myocardial infarction. J Thromb Thrombolysis 2004; 18: 187-192.
- 11. Yan BP, Ajani AE, Duffy SJ, et al. Use of drug-eluting stents in Victorian public hospitals. Med J Aust 2006; 185: 363-367. <MJA full text>
- 12. Dalby M, Bouzamondo A, Lechat P, et al. Transfer for primary angioplasty versus immediate thrombolysis in acute myocardial infarction: a meta-analysis. Circulation 2003; 108: 1809-1814.
- 13. Krumholz HM, Bradley EH, Nallamothu BK, et al. A campaign to improve the timeliness of primary percutaneous coronary intervention: door-to-balloon: an alliance for quality. JACC Cardiovasc Interv 2008; 1: 97-104.
- 14. Cannon CP, Gibson CM, Lambrew CT, et al. Relationship of symptom-onset-to-balloon time and door-to-balloon time with mortality in patients undergoing angioplasty for acute myocardial infarction. JAMA 2000; 283: 2941-2947.
- 15. McNamara RL, Herrin J, Bradley EH, et al. Hospital improvement in time to reperfusion in patients with acute myocardial infarction, 1999 to 2002. J Am Coll Cardiol 2006; 47: 45-51.
- 16. Nallamothu BK, Bates ER, Herrin J, et al. Times to treatment in transfer patients undergoing primary percutaneous coronary intervention in the United States: National Registry of Myocardial Infarction (NRMI) — 3/4 analysis. Circulation 2005; 111: 761-767.
- 17. Carstensen S, Nelson GC, Hansen PS, et al. Field triage to primary angioplasty combined with emergency department bypass reduces treatment delays and is associated with improved outcome. Eur Heart J 2007; 28: 2313-2319.
- 18. Hutchison A, Barger B, Jarvie I, et al. Field 12 lead ECG to triage STEMI significantly improves door to balloon times: the Metropolitan Ambulance Service and MonashHEART acute myocardial infarction 12 lead pilot (MonAMI–MAS) project. Heart Lung Circ 2008; 17 Suppl 3: S187.
- 19. Cantor WJ, Fitchett D, Borgundvaag B, et al. Routine early angioplasty after fibrinolysis for acute myocardial infarction. N Engl J Med 2009; 360: 2705-2718.
- 20. Boersma E. Primary coronary angioplasty vs. thrombolysis group. Does time matter? A pooled analysis of randomized clinical trials comparing primary percutaneous coronary intervention and in-hospital fibrinolysis in acute myocardial infarction patients. Eur Heart J 2006; 27: 779-788.





Abstract
Objectives: To compare clinical outcomes between patients with ST-elevation myocardial infarction (STEMI) presenting to a hospital with facilities for primary percutaneous coronary intervention (PCI) and patients transferred from a non-PCI-capable unit, and to determine the success rate of meeting clinical guidelines for management of STEMI.
Design, setting and participants: Prospective study of patients with STEMI who underwent PCI at Box Hill Hospital (BHH), Melbourne, between 1 July 2002 and 30 June 2008. We compared two patient groups: “BHH patients”, who were admitted directly to BHH (a hospital with PCI capability), and “SHIPEM (Shipping Infarcts for Primary Angioplasty in Eastern Melbourne Registry) patients”, who were transferred from other hospitals without PCI capability.
Main outcome measures: Clinical outcomes; symptom-to-first-door time (time between symptom onset and arrival at first hospital); first-door-to-balloon time (time between arrival at the first hospital and inflation of the angioplasty balloon); compliance with Cardiac Society of Australia and New Zealand/National Heart Foundation of Australia (CSANZ/NHFA) guidelines for management of patients with STEMI.
Results: There were 598 patients in the BHH group and 189 in the SHIPEM group. The median first-door-to-balloon time was 89 minutes (interquartile range [IQR], 69–107 minutes) for BHH patients and 128 minutes (IQR, 104–157 minutes) for SHIPEM patients. These figures did not vary significantly over the 6 years of the registry. In the BHH group, 180 patients (30.1%) had a symptom-to-first-door time of ≤ 60 minutes, with 32 (17.8%) receiving PCI in ≤ 60 minutes. The corresponding figure for the SHIPEM group was 48 patients (25.4%), with 1 (2.1%) receiving PCI within 60 minutes. In the BHH group, 304 patients (50.8%) had a symptom-to-first-door time of 61–180 minutes, with 166 (54.6%) receiving PCI in ≤ 90 minutes. In the SHIPEM group, 50 patients (26.5%) had a symptom-to-first-door time of > 180 minutes, with 21 (42.0%) receiving PCI in ≤ 120 minutes.
Conclusion: Our study demonstrates that transfer for PCI is feasible and safe in selected patients, with outcomes comparable to those of patients presenting to a PCI-capable unit. However, the CSANZ/NHFA targets, predicated by symptom-to-first-door time, are not being met and have not improved over time, which suggests that strategies to improve symptom-to-first-door, first-door-to-balloon and transfer times need to be addressed.