The Australian response to the first wave of pandemic (H1N1) 2009 influenza (nH1N1) has included the identification of vulnerable populations, to whom early treatment and careful follow-up has been targeted.1 One of these populations, Indigenous Australians, comprises 2.5% of the Australian population but accounted for 16% of patients hospitalised with nH1N1 and 9.7% of those admitted to an intensive care unit (ICU) with nH1N1.1,2 However, these nationwide figures may not reveal the true extent of the impact of nH1N1 on jurisdictions with sizeable Indigenous populations.
The Top End of the Northern Territory of Australia is unique for its small, widely dispersed population of 172 000 in an area of 477 000 square kilometres and its high proportion (26.5%) of Indigenous Australians.3,4 There are high rates of chronic disease in the Indigenous population,5 whose levels of mobility between communities,6 inadequate health hardware (eg, taps, hot water, toilets, drains),7 and overcrowded living conditions are likely to facilitate spread of the nH1N1 virus.8 Here, we describe the impact of nH1N1 on this vulnerable population.
The Top End of the NT encompasses the administrative regions of Darwin, Katherine and East Arnhem and is served by the Royal Darwin Hospital (RDH), a 350-bed tertiary referral centre containing the region’s only ICU, and two small regional hospitals. During the study period — 1 June 2009 to 31 August 2009 — all of these hospitals implemented a consistent policy of influenza screening for all patients presenting with an influenza-like illness (ILI).
We collected and analysed data on: all Top End nH1N1 notifications (notification analysis); all patients admitted through Top End emergency departments with an ILI (ILI analysis); all patients admitted to the RDH with nH1N1 (RDH admission analysis); and all patients admitted to the RDH ICU with nH1N1 (ICU analysis).
An ILI was defined as a fever (temperature ≥38ºC or a documented fever history) with a cough and/or sore throat.9 Influenza A was diagnosed by polymerase chain reaction (PCR) analysis of combined nose and throat swabs, and nH1N1 was diagnosed by PCR analysis using primers specific for the nH1N1 haemagglutinin gene.10
Remote dwelling was defined using the Australian Bureau of Statistics remoteness area (RA) classification,11 with urban defined as RA code 2 and remote as RA code 3 or 4.
For the notification analysis, population-level notification data were provided by the NT Centre for Disease Control. Before 18 June 2009, testing of anyone presenting to a health care facility with an ILI was encouraged; after this time, only those with severe disease or underlying risk factors were tested — in line with national policy.9
For the ILI analysis, hospital admission records were linked with influenza testing data obtained from the RDH microbiology laboratory. We included all patients who were admitted through an emergency department or maternity ward of a hospital in the region during the study period and had a specimen taken for influenza A PCR analysis within 48 hours of admission (before or after). In cases where a person had multiple admissions fulfilling these criteria, the first influenza A-positive admission was included; if none were influenza A positive, the first admission was included. Data including patient demographics, ICU admission and in-hospital mortality were obtained from hospital databases.
For the ICU analysis additional ICU-specific data were collected prospectively by RDH ICU staff for the Australian and New Zealand Intensive Care Society registry.2 Daily ICU bed occupancy was defined as the proportion of the eight beds staffed for ventilated patients occupied each day, and could exceed 100%.
Continuous variables were compared using the Mann–Whitney U test. Categorical variables were compared using the χ2, Fisher exact or Wilcoxon rank sum tests as appropriate. Non-annualised incidence rates were calculated for the region using 2008 NT population data provided by the Health Gains Unit of the NT DHF. These were standardised to the 2001 Australian standard population using the direct method.12 Adjusted incidence rate ratios (IRRs) were derived using a Poisson regression model. A univariate analysis was performed for the ILI analysis. In the RDH admission analysis, patients requiring ICU care were compared with those not requiring ICU care. Clinically important variables and those with P < 0.25 in the univariate analysis were included in an initial logistic regression model with backwards stepwise elimination of variables. Further univariate analyses were performed comparing Indigenous patients with non-Indigenous patients. Statistical analyses were performed using STATA version 11 (StataCorp, College Station, Tex, USA). Two-sided P values of < 0.05 were considered significant.
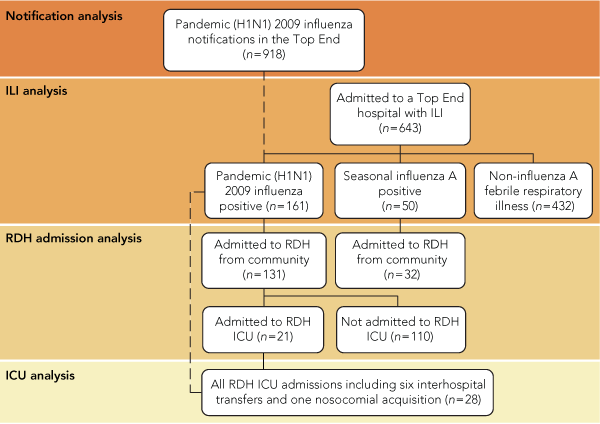
The numbers and categories of notifications and patients included in the four parts of this study are summarised in Box 1.
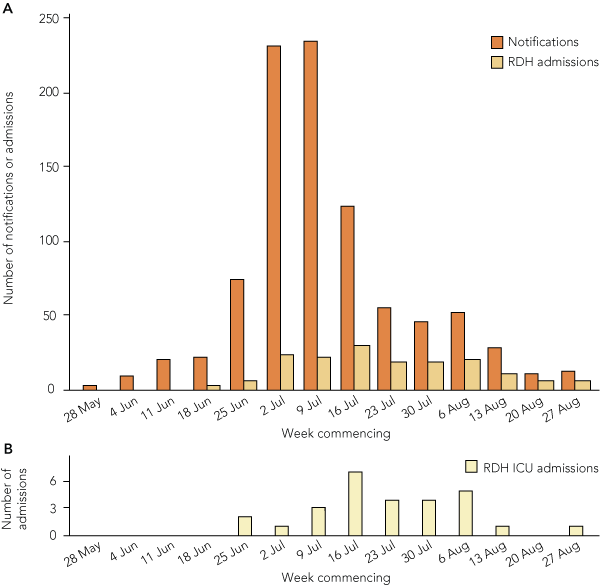
A total of 918 cases of nH1N1 in the Top End were notified during the study period. This corresponds to an age-adjusted notification rate of 497 (95% CI, 494–500) per 100 000 estimated resident population (ERP). There were 473 female (51.5%) and 445 male (48.5%) cases. The number of notifications increased sharply in Week 5 (25 June – 1 July) and peaked in Week 7 (9–15 July) (Box 2).
During the study period, 643 patients were admitted through Top End emergency departments with an ILI. Of these patients, 161 (25%) tested positive for nH1N1, a further 50 (8%) tested positive for seasonal influenza A, and 432 (67%) had a non-influenza A febrile respiratory illness (Box 3). Indigenous patients were over-represented in all three of these patient groups, and accounted for 72% of those who tested positive for nH1N1. There was no significant difference in either the need for ICU admission or in-hospital mortality between the three groups.
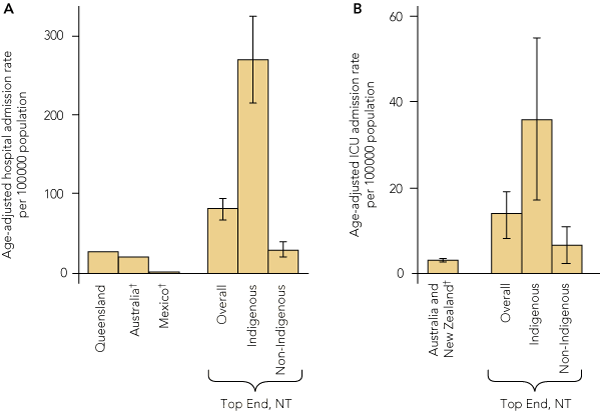
The overall age-adjusted incidence of admission with an ILI was 361 per 100 000 ERP over the study period. The corresponding incidence rate for nH1N1 was 82 per 100 000 ERP overall, with stark differences between Indigenous and non-Indigenous populations (269 [95% CI, 214–324]) v 29 [95% CI, 20–39] per 100 000 ERP). The adjusted IRR for nH1N1 admission was 12 (95% CI, 7.8–18; adjusted for age and remoteness) and the IRR for ICU admission was 5.2 (95% CI, 2.3–12) — both calculated for Indigenous versus non-Indigenous patients (Box 4). For the Indigenous population alone, the IRR of hospital admission for remote compared with urban dwelling was 0.63 (95% CI, 0.43–0.92).
There were significant differences between Indigenous and non-Indigenous patients admitted with nH1N1 (Box 5). Indigenous patients were more likely to live remotely and were younger, with lower haemoglobin and serum albumin levels, and higher white cell counts and C-reactive protein (CRP) levels than non-Indigenous patients. The proportions of patients who reported being a current smoker and patients who reported hazardous alcohol use were also higher in Indigenous patients.
Twenty-eight of the 161 patients in the Top End who were positive for nH1N1 (17%) were admitted to the RDH ICU; this included six interhospital transfers and one nosocomial acquisition. The median (IQR) APACHE (Acute Physiology and Chronic Health Evaluation) II score for these patients was 16 (14–23), and four patients died. The average daily ICU bed occupancy was 110% and nH1N1 admissions contributed 23% of this load. The median (IQR) length of stay was 4.5 (1.0–6.7) days. Admission of patients positive for nH1N1 to the RDH ICU peaked in Week 8 (16–22 July) (Box 2).
In the Top End of northern Australia, nH1N1 had a disproportionate impact on the Indigenous population at community, hospital and ICU levels. Rates of notification, hospital admission and ICU admission were 3.5 times, 12 times and 5 times higher, respectively, than for the non-Indigenous population. In contrast, hospital and ICU admission rates for the non-Indigenous population in this region were equivalent to those for Australia as a whole.2,13 The hospital admission rates in the Top End are significantly higher than those reported for Australia overall and overseas.13,14 This is true for the overall hospital admission rate that includes both Indigenous and non-Indigenous populations, but is particularly so for the Indigenous population alone.
It is possible that the actual nH1N1 admission rates were higher than estimated here. Although PCR analysis of optimal samples has a sensitivity of nearly 100%,15 the similar demographics, outcomes and epidemic curves for non-influenza ILI and nH1N1 raise the possibility that a proportion of the non-influenza ILI cases were false negatives. Alternatively, other circulating respiratory viruses may have caused similar clinical presentations.
Several factors are likely to underpin high admission rates for the Indigenous population. First, we infer an excess incidence of nH1N1 cases within the Indigenous population from the high notification rate for this group. Although this may reflect differential testing driven by additional comorbidities in the Indigenous population, it is consistent with the distribution of early notifications in the NT when testing was universal.16 Likely explanations for this include a highly mobile Indigenous population, crowded living circumstances and inadequate health hardware.6-8 Such conditions contribute to the transmission of skin pathogens, and a similar effect may occur with pathogens transmitted by aerosol and droplet spread.17,18 The lack of adequate housing and subsequent overcrowding in Indigenous communities is particularly striking — recent studies have found that dwellings in Indigenous communities house a mean of 3.4 persons per bedroom, and up to 7.5 persons per bedroom.19,20
Second, Indigenous populations worldwide have a high prevalence of risk factors for severe influenza.21,22 Among Indigenous people living in the Top End, the population-based prevalence of chronic lung disease has been estimated at eight per cent, four times higher than the comparable age-adjusted Australian rate, and a similar excess has been shown for the other major comorbidities.5 Given this substantial underlying disparity, it is noteworthy that there were few differences in risk factors between Indigenous and non-Indigenous hospitalised patients in our study. This suggests that excess comorbidities contributed to the high hospitalisation rates among Indigenous people (otherwise the differences in risk factors would have been preserved).
Reports to date have consistently found that Indigenous people with nH1N1 are over-represented in ICUs.2,23 Our data suggest that this over-representation flows from an excess of community and hospital cases, rather than an environmentally or genetically determined susceptibility to severe disease as has been hypothesised.23 We observed a similar pattern with pregnant women, who were over-represented in the hospital admissions (46% of women of child-bearing age) but not the ICU admissions. A previous study showed that 25% of all women admitted with nH1N1 in Melbourne were pregnant.24 However, by restricting the denominator to women of child-bearing age this figure becomes a more comparable 38% (Justin Denholm, Infectious Diseases Physician, Victorian Infectious Diseases Service, personal communication).
Risk factors that were associated with ICU admission on multivariate analysis were hypoxia and chest x-ray infiltrates. Although both obesity and hypoalbuminaemia were more than twice as common in ICU patients on univariate analysis, neither were significant on multivariate analysis. A high prevalence of obesity in ICU patients with nH1N1 has been reported elsewhere,2,23 and hypoalbuminaemia has been shown to be associated with severe nH1N1 infection25 and ICU admission in Top End patients with community-acquired pneumonia.26 The absence of associations between severe nH1N1 disease and obesity and hypoalbuminaemia on multivariate analysis may be due to a small sample size.
The consistently high ICU bed occupancy observed during the study period was due to the substantial burden of nH1N1 on critical care resources. The nH1N1-related bed occupancy of 23% was higher than the 5.2% reported across Australasia,2 and directly affected the capacity of the RDH as both a remote regional ICU and as the National Critical Care and Trauma Response Centre. The mortality rate of 14% for patients who were admitted to the RDH ICU (four deaths among 28 patients) was comparable to national figures.2 We noted two cases of severe hyperthermia and rhabdomyolysis with nH1N1; to date, this brings the total number of rhabdomyolysis cases reported worldwide to four.27 Lack of access to ECMO did not appear to adversely affect outcomes, which is reassuring for other jurisdictions without access to ECMO.
In conclusion, pandemic (H1N1) 2009 influenza has disproportionately affected the Indigenous population of the NT. This is likely to be due to a combination of increased attack rates and a high prevalence of comorbidities in this population. These findings have significant implications for planning hospital and ICU capacity during an influenza pandemic in regions with large Indigenous populations. They also confirm the need to improve health and living circumstances and prioritise vaccination in this population.
1 Summary of influenza notifications and patient groups from the Top End of the Northern Territory (1 June to 31 August 2009) included in this study
 |
|
ILI = influenza-like illness. RDH = Royal Darwin Hospital. ICU = intensive care unit. |
2 Notifications and RDH admissions (A) and RDH ICU admissions (B) for pandemic (H1N1) 2009 in the Top End of the Northern Territory by week (1 June to 31 August 2009)
 |
3 Characteristics of patients admitted for acute care to Top End hospitals with an influenza-like illness (ILI), by result of influenza testing (1 June to 31 August 2009)*
4 Rates of admission to hospital (A) and ICU (B) with pandemic (H1N1) 2009 influenza in previous studies2,13,14 and this study (Top End, Northern Territory)*
 |
- Shaun M Flint1
- Joshua S Davis2,3,4
- Jiunn-Yih Su1
- Erin P Oliver-Landry5
- Benjamin A Rogers2
- Aaron Goldstein4
- Jane H Thomas6
- Uma Parameswaran2
- Colin Bigham6
- Kevin Freeman7
- Paul Goldrick6
- Steven Y C Tong3,4,2
- 1 Northern Territory Centre for Disease Control, Darwin, NT.
- 2 Department of Infectious Diseases, Royal Darwin Hospital, Darwin, NT.
- 3 Menzies School of Health Research, Darwin, NT.
- 4 Charles Darwin University, Darwin, NT.
- 5 Northern Territory Clinical School, Royal Darwin Hospital, Darwin, NT.
- 6 Department of Intensive Care, Royal Darwin Hospital, Darwin, NT.
- 7 Microbiology Laboratory, Northern Territory Government Pathology Service, Darwin, NT.
We thank all the Microbiology Laboratory staff, the Infection Control team, and medical staff of the Royal Darwin Hospital, and Lesley Scott and the surveillance staff of the Northern Territory Centre for Disease Control, for their diligent and unstinting work during the period of this study. We also thank Ross Andrews, Naor Bar-Zeev and Thomas Snelling for critical review of the manuscript. Joshua Davis and Steven Tong received PhD scholarships for salary support from the NHMRC.
None identified.
- 1. Bishop JF, Murnane MP, Owen R. Australia’s winter with the 2009 pandemic influenza A (H1N1) virus. N Engl J Med 2009; 361: 2591-2594.
- 2. Webb SA, Pettila V, Seppelt I, et al. Critical care services and 2009 H1N1 influenza in Australia and New Zealand. N Engl J Med 2009; 361: 1925-1934.
- 3. Australian Bureau of Statistics. Regional population growth, Australia, 2008–09. Canberra: ABS, 2010. (ABS Cat. No. 3218.0.) http://abs.gov.au/AUSSTATS/abs@.nsf/Lookup/3218.0Main+Features12008-09?OpenDocument (accessed Apr 2010).
- 4. Australian Bureau of Statistics. Population distribution, Aboriginal and Torres Strait Islander Australians. Canberra: ABS, 2006. (ABS Cat. No. 4705.0.) http://www.ausstats.abs.gov.au/Ausstats/subscriber.nsf/0/377284127F903297 CA25733700241AC0/$File/47050_2006.pdf (accessed Apr 2010).
- 5. Zhao Y, Connors C, Wright J, et al. Estimating chronic disease prevalence among the remote Aboriginal population of the Northern Territory using multiple data sources. Aust N Z J Public Health 2008; 32: 307-313.
- 6. Warchivker I, Tjapangati T, Wakerman J. The turmoil of aboriginal enumeration: mobility and service population analysis in a central Australian community. Aust N Z J Public Health 2000; 24: 444-449.
- 7. Torzillo PJ, Pholeros P, Rainow S, et al. The state of health hardware in Aboriginal communities in rural and remote Australia. Aust N Z J Public Health 2008; 32: 7-11.
- 8. Australian Bureau of Statistics, Australian Institute of Health and Welfare. The Health and Welfare of Australia’s Aboriginal and Torres Strait Islander peoples, 2008. Canberra: ABS, 2008. (ABS Cat. No. 4704.0.)
- 9. Communicable Diseases Network Australia. H1N1 influenza 09 infection: ‘Protect Phase’ guidelines for Australian public health units. Version 4.1, 8 July 2009. http://www.health emergency.gov.au/internet/healthemergency/publishing.nsf/Content/clinical-vulnerable/$File/CDNA-H1N1-Protect-SoNG.pdf (accessed Mar 2010).
- 10. Whiley DM, Bialasiewicz S, Bletchly C, et al. Detection of novel influenza A(H1N1) virus by real-time RT-PCR. J Clin Virol 2009; 45: 203-204.
- 11. Australian Bureau of Statistics. Australian standard geographical classification (ASGC), July 2009. Canberra: ABS, 2009. (ABS Cat. No. 1216.0.)
- 12. Australian Institute of Health and Welfare. Australia’s health 2008. Canberra: AIHW, 2008. (AIHW Cat. No. AUS 99.)
- 13. Australian Government Department of Health and Ageing. Australian influenza surveillance summary. Report No. 18, 2009, reporting period: 5 September 2009 – 11 September 2009. http://www.health.gov.au/internet/main/publishing.nsf/Content/cda-surveil-ozflu-flucurr.htm/$FILE/ozflu-no18-2009.pdf (accessed Mar 2010).
- 14. Echevarría-Zuno S, Mejía-Aranguré JM, Mar-Obeso AJ, et al. Infection and death from influenza A H1N1 virus in Mexico: a retrospective analysis. Lancet 2009; 374: 2072-2079.
- 15. Lam WY, Leung TF, Lee N, et al. Development and comparison of molecular assays for the rapid detection of the pandemic influenza A (H1N1) 2009 virus. J Med Virol 2010; 82: 675-683.
- 16. Flint S, Su J-Y, Scott L, Krause V. The early experience of pandemic (H1N1) 2009 in the Northern Territory, Australia. NT Dis Control Bull 2009; 16 (2): 1-8.
- 17. Tong SY, Bishop EJ, Lilliebridge RA, et al. Community-associated strains of methicillin-resistant Staphylococcus aureus and methicillin-susceptible S aureus in indigenous Northern Australia: epidemiology and outcomes. J Infect Dis 2009; 199: 1461-1470.
- 18. Tong SY, McDonald MI, Holt DC, Currie BJ. Global implications of the emergence of community-associated methicillin-resistant Staphylococcus aureus in Indigenous populations. Clin Infect Dis 2008; 46: 1871-1878.
- 19. Bailie RS, Stevens MR, McDonald E, et al. Skin infection, housing and social circumstances in children living in remote Indigenous communities: testing conceptual and methodological approaches. BMC Public Health 2005; 5: 128.
- 20. McDonald MI, Towers RJ, Andrews RM, et al. Low rates of streptococcal pharyngitis and high rates of pyoderma in Australian aboriginal communities where acute rheumatic fever is hyperendemic. Clin Infect Dis 2006; 43: 683-689.
- 21. Health status of American Indians compared with other racial/ethnic minority populations — selected states, 2001–2002. MMWR Morb Mortal Wkly Rep 2003; 52: 1148-1152.
- 22. Schanzer DL, Langley JM, Tam TW. Co-morbidities associated with influenza-attributed mortality, 1994–2000, Canada. Vaccine 2008; 26: 4697-4703.
- 23. Kumar A, Zarychanski R, Pinto R, et al. Critically ill patients with 2009 influenza A(H1N1) infection in Canada. JAMA 2009; 302: 1872-1879.
- 24. Denholm JT, Gordon CL, Johnson PD, et al. Hospitalised adult patients with pandemic (H1N1) 2009 influenza in Melbourne, Australia. Med J Aust 2010; 192: 84-86. <MJA full text>
- 25. Gordon CL, Johnson PD, Permezel M, et al. Association between severe pandemic 2009 influenza A (H1N1) virus infection and immunoglobulin G(2) subclass deficiency. Clin Infect Dis 2010; 50: 672-678.
- 26. Davis JS, Cross GB, Charles PG, et al. Pneumonia risk stratification in tropical Australia: does the SMART-COP score apply? Med J Aust 2010; 192: 133-136. <MJA full text>
- 27. D’Silva D, Hewagama S, Doherty R, et al. Melting muscles: novel H1N1 influenza A associated rhabdomyolysis. Pediatr Infect Dis J 2009; 28: 1138-1139.





Abstract
Objective: To describe the impact of pandemic (H1N1) 2009 influenza (nH1N1) on Indigenous people in the Top End of the Northern Territory at community, hospital and intensive care unit (ICU) levels.
Design, setting and participants: We analysed influenza notifications for the Top End from 1 June to 31 August 2009, as well as data on patients admitted through Top End emergency departments with an influenza-like illness. In addition, data on patients with nH1N1 who were admitted to Royal Darwin Hospital (RDH) and the RDH ICU were prospectively collected and analysed.
Main outcome measures: Age-adjusted notification rates for nH1N1 cases, Top End hospital admission rates for patients with nH1N1 and RDH ICU admission rates for patients with nH1N1, stratified by Indigenous status.
Results: There were 918 nH1N1 notifications during the study period. The age-adjusted hospital admission rate for nH1N1 was 82 per 100 000 (95% CI, 68–95) estimated resident population (ERP) overall, with a markedly higher rate in the Indigenous population compared with the non-Indigenous population (269 per 100 000 versus 29 per 100 000 ERP; adjusted incidence rate ratio, 12 [95% CI, 7.8–18]). Independent predictors of ICU admission compared with hospitalisation were hypoxia (adjusted odds ratio [aOR], 4.5; CI, 1.5–13.1) and chest x-ray infiltrates (aOR, 4.3; CI, 1.5–12.6) on hospital admission.
Conclusions: Pandemic (H1N1) 2009 influenza had a disproportionate impact on Indigenous Australians in the Top End, with hospitalisation rates higher than those reported elsewhere in Australia and overseas. These findings have implications for planning hospital and ICU capacity during an influenza pandemic in regions with large Indigenous populations. They also confirm the need to improve health and living circumstances and to prioritise vaccination in this population.