Soon after reaching 40 years of age, men experience a fall in serum total testosterone (TT) levels of about 0.8%–1.6% per year.1-3 The magnitude of this decline is influenced by factors including chronic illness and obesity; obese men have TT levels up to 25% lower than their non-obese peers.4 Obesity may also predict a greater rate of decline in serum TT with ageing.5,6
In Australia, 25.5% of men aged 55–64 years and 19.9% of men aged 65–74 years are obese, as defined by body mass index (BMI: ratio of body weight in kilograms to height in metres squared) ≥ 30 kg/m2; this represents a 2.5-fold increase over the past 20 years.7 More than one in three men aged 55 years or older has a waist circumference (WC) in the obese range (≥ 102 cm) — a 50% increase since 1989.8 This rise in obesity is likely to be an important factor contributing to the reported increase in prevalence of androgen deficiency (AD) in ageing men.9
Testosterone supplementation is increasingly used by ageing men,10-12 with candidate selection usually based on a combination of symptoms suggestive of AD and the determination of serum TT,12-14 as neither is consistently reliable in isolation. However, there is no consensus as to the level of TT below which treatment is warranted.10,12-14 International recommendations support the notion that TT levels less than 8 nmol/L require substitution and that levels above 12 nmol/L do not.15 The Endocrine Society of Australia (ESA) guidelines, adopted by the Pharmaceutical Benefits Scheme for subsidy of testosterone prescription in men aged 40 years and older, require, in the absence of demonstrable testicular or pituitary disease, documentation of a TT level less than 8 nmol/L or TT levels 8–15 nmol/L with luteinising hormone (LH) level greater than 1.5 times the upper limit of the eugonadal reference range for young men (as an indicator of primary testicular failure) on two separate morning samples.16
Men were excluded via telephone screening if they reported excessive alcohol intake, malignancy, serious chronic medical illness (including diabetes mellitus and sleep apnoea), prostate cancer or clinically significant benign prostate disease, use of testosterone therapy in the previous 12 months, or use of medications known to affect testosterone levels. Men were also excluded if they reported Class II or III obesity (BMI ≥ 35 kg/m2),17 because of the recognised association with hypogonadotrophic hypogonadism.18
TT was measured by immunoassay using a Chiron Diagnostic ACS: 180 automated chemiluminescence system (Chiron Corporation, East Walpole, Mass, USA) (interassay coefficient of variation [CV], 7.9% at 8 nmol/L; reference range [RR], 8–28 nmol/L). SHBG was determined on an IMMULITE Analyzer (CV, 7.9%) and oestradiol was assayed by radioimmunoassay (CV, 14.5%) (Diagnostics Product Corporation, Los Angeles, Calif, USA). The calculated free testosterone (calc FT) was determined using the Vermeulen equation.19 LH was measured on an AxSym (Abbott Diagnostics, Abbott Park, Ill, USA) (CV, 6.4%).
Height was determined by a stadiometer calibrated to 0.1 cm, and weight was measured to 0.1 kg to calculate BMI.17 WC was measured at the midpoint between the costal margin and iliac crest.17 For the purposes of this study, men were considered obese if they had either a BMI ≥ 30.0 kg/m2 or WC ≥ 102 cm.17,20
Ninety-nine men were obese (BMI ≥ 30.0 kg/m2 or WC ≥ 102 cm) and 124 men were non-obese (Box 1). Obese men had lower mean TT (12.7 ± 0.4 v 15.0 ± 0.4 nmol/L; P < 0.001) and mean SHBG (34.8 ± 1.4 v 40.2 ± 1.3 nmol/L; P = 0.005) levels than non-obese men. Mean calc FT levels were also lower in the obese men (275.7 ± 7.8 v 299.3 ± 7.4 pmol/L; P = 0.03). Median LH (4.4 [IQR, 3.4–6.7 IU/L] v 5.3 [IQR, 3.7–6.5 IU/L]; P = 0.21) and mean oestradiol (40.3 ± 1.8 v 38.9 ± 1.6 pmol/L; P = 0.46) levels were not different.
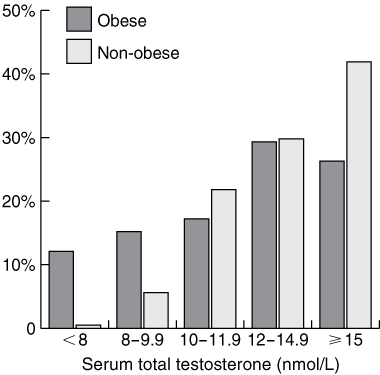
Of the obese men, 12% had a mean TT level < 8 nmol/L, compared with 1% of non-obese men (Box 2); the relative risk for mean TT < 8 nmol/L in the obese men was 2.23 (95% CI, 1.78–2.79; P = 0.0003). Mean TT levels < 12.0 nmol/L were recorded in 44% of obese men and 28% of non-obese men; the relative risk for TT < 12 nmol/L in obese men was 1.46 (95% CI, 1.10–1.94; P = 0.02).
When the ESA guidelines16 for the diagnosis of age-related AD were applied to the group, 15 obese men (15%) and four non-obese men (3%) were classified as being eligible for PBS-supported testosterone therapy (Box 3); the relative risk in obese men was 1.92 (95% CI, 1.44–2.55; P = 0.003).
This is the first description of the effect of obesity on serum testosterone levels in a group of ageing men with symptoms suggestive of AD, who are likely to reflect those who may seek advice about the role of testosterone therapy. These data confirm and extend previous observations from epidemiological studies.4,21 In this group of otherwise healthy but symptomatic men aged 54–86 years, mild obesity (defined by BMI or WC) was associated with a 15% reduction in TT levels.
The requirement for symptoms of AD was included to encourage involvement of men likely to present to health professionals to discuss male ageing or testosterone treatment. Although not directly comparable, the phenotype of our volunteers is likely to be similar to that referred to as “LOH: Late Onset Hypogonadism” — the “clinical and biochemical syndrome” described by others.15 We excluded men who reported moderate or severe obesity because of the known association with hypogonadotrophic hypogonadism.18,21
It has been suggested that lower TT levels in obese men reflect the effect of obesity per se or the effect of the accompanying insulin resistance, both of which suppress SHBG levels, and that indices of bioavailable or free testosterone levels remain normal in mild to moderate obesity.21,22 SHBG levels were predictably lower in the obese men, but, in contrast to previous reports,21,22 calc FT levels were also significantly lower. The failure to observe a reciprocal rise in serum LH levels suggests the possibility that mild obesity is associated with a degree of hypogonadotrophic hypogonadism; this has previously been described in massive obesity.18 Alternatively, the lower TT and calc FT levels in the obese men may not necessarily reflect deficient androgen action, including at the hypothalamo-pituitary level. This uncertainty regarding the true androgen state of mildly obese men underscores the importance of a comprehensive clinical assessment for features of AD before the measurement of biochemical androgen profiles, and highlights the potential pitfalls of screening based only on testosterone levels. Furthermore, it questions the reflex assumption that low to normal TT levels, and even calc FT levels, necessarily indicate testosterone therapy in the absence of strong and specific features of AD; resolution of the place of testosterone therapy will rely on clinical research targeting this group.
Applying the ESA guidelines,16 AD was determined to exist in one in seven men aged 55 years or older who were generally or abdominally obese (but with a BMI not exceeding 35 kg/m2) and with symptoms consistent with AD, in contrast to only one in 33 non-obese men of similar age, yet with the same symptom profile.
Our data also support the requirement for confirmation of low TT levels for the diagnosis of AD. Week-to-week variability in TT levels is well documented,23 and in our cohort, three out of 11 obese men and three out of four non-obese men with initial TT < 8 nmol/L would have been reclassified on retesting.
The threshold level of TT for confirmation of the clinical suspicion of AD is uncertain. Expert opinion suggests that a TT level below 6.9 nmol/L warrants treatment12,14 — this would apply to 5% of our obese men but only 1% of our non-obese men. Recent guidelines also advise that TT levels < 8 nmol/L or free testosterone levels < 180 pmol/L are supportive of androgen deficiency;15 12% (TT) and 7% (calc FT) of obese men and 1% (TT) and 5% (calc FT) of non-obese men met these criteria. The US Institute of Medicine recommendation for studies of testosterone therapy in ageing men suggests inclusion of subjects with baseline TT < 10.4 nmol/L;10 by this criterion, only 11% of non-obese men but 30% of obese men in our study would be eligible.
Underscoring consideration of any biochemical assessment is an understanding of the differing performance characteristics of commercial TT assay platforms — across seven common platforms, the lower reference value varies between 7.2 and 12.6 nmol/L.24 Although our samples were analysed with a single assay platform, it is important to recognise that the numerical values are applicable only to that assay.
Consideration was given to the possibility that we may have preferentially recruited obese men. The 1999–2000 Australian Diabetes, Obesity and Lifestyle Study (AusDiab),7 which was ongoing at the time of our study, determined that 26% of Australian men aged 55–64 years are obese.7 Our data were consistent with this, documenting a 22% incidence of obesity in men of comparable age.
Our study confirms that obesity is an important determinant of serum TT levels in symptomatic ageing men, with almost one in seven obese men eligible for PBS-supported testosterone therapy. As obesity prevalence rates increase,17 it is expected that the number of ageing men with TT values falling within a range consistent with AD will rise in parallel. Whether these men have a true hypoandrogenic state, or whether the lower TT levels are an artefact of obesity, remains unclear, and careful clinical assessment is needed. With the strong association between obesity and cardiovascular risk, and the emerging observational evidence linking low TT levels with an increase in cardiovascular disease,25 studies of the effects of testosterone therapy in these men are clearly warranted.
Received 20 June 2006, accepted 30 August 2006
- Carolyn A Allan1,3
- Boyd J Strauss2
- Henry G Burger1
- Elise A Forbes1
- Robert I McLachlan1,3
- 1 Prince Henry's Institute, Melbourne, VIC.
- 2 Clinical Nutrition and Metabolism Unit, Monash Medical Centre and Department of Medicine, Monash University, Melbourne, VIC.
- 3 Andrology Australia, Melbourne, VIC.
Carolyn Allan received a National Health and Medical Research Council (NHMRC) Medical Postgraduate Research Scholarship, #169015 2001–2004. Robert McLachlan is a Principal Research Fellow, NHMRC #169020 2002–2006. This work was supported by Mayne Pharma Australia Pty Ltd.
Mayne Pharma Australia Pty Ltd provided funding for this study. Study design and conduct, data collection, analysis and interpretation, and preparation of this manuscript were undertaken solely by the authors.
- 1. Feldman HA, Longcope C, Derby CA, et al. Age trends in the level of serum testosterone and other hormones in middle-aged men: longitudinal results from the Massachusetts male aging study. J Clin Endocrinol Metab 2002; 87: 589-598.
- 2. Harman SM, Metter EJ, Tobin JD, et al. Longitudinal effects of aging on serum total and free testosterone levels in healthy men. Baltimore Longitudinal Study of Aging. J Clin Endocrinol Metab 2001; 86: 724-731.
- 3. Leifke E, Gorenoi V, Wichers C, et al. Age-related changes of serum sex hormones, insulin-like growth factor-1 and sex-hormone binding globulin levels in men: cross-sectional data from a healthy male cohort. Clin Endocrinol (Oxf) 2000; 53: 689-695.
- 4. Gray A, Feldman HA, McKinlay JB, et al. Age, disease, and changing sex hormone levels in middle-aged men: results of the Massachusetts Male Aging Study. J Clin Endocrinol Metab 1991; 73: 1016-1025.
- 5. Mohr BA, Guay AT, O’Donnell AB, et al. Normal, bound and nonbound testosterone levels in normally ageing men: results from the Massachusetts Male Ageing Study. Clin Endocrinol (Oxf) 2005; 62: 64-73.
- 6. Derby CA, Zilber S, Brambilla D, et al. Body mass index, waist circumference and waist to hip ratio and change in sex steroid hormones: the Massachusetts Male Ageing Study. Clin Endocrinol (Oxf) 2006; 65: 125-131.
- 7. Cameron AJ, Welborn TA, Zimmet PZ, et al. Overweight and obesity in Australia: the 1999-2000 Australian Diabetes, Obesity and Lifestyle Study (AusDiab). Med J Aust 2003; 178: 427-432. Erratum in: Med J Aust 2004; 180: 418. (corrected online) <MJA full text>
- 8. Bennett S, Magnus P, Gibson D. Obesity trends in older Australians. AIHW Bulletin No. 12. Canberra: Australian Institute of Health and Welfare, 2004. (AIHW Catalogue No. AUS 42.)
- 9. McLachlan RI, Allan CA. Defining the prevalence and incidence of androgen deficiency in aging men: where are the goal posts? J Clin Endocrinol Metab 2004; 89: 5916-5919.
- 10. Liverman CT, Blazer DG, editors. Testosterone and aging: clinical research directions. Washington, DC: Institute of Medicine of the National Academies, 2003.
- 11. Handelsman DJ. Trends and regional differences in testosterone prescribing in Australia, 1991–2001. Med J Aust 2004; 181: 419-422. <MJA full text>
- 12. Cunningham GR, Swerdloff RS. Summary from the Second Annual Andropause Consensus Meeting. Chevy Chase, Md: Endocrine Society, 2001. http://www.endo-society.org/publicpolicy/legislative/upload/andro_cc_summary.pdf (accessed Sep 2006).
- 13. Lunenfeld B, Saad F, Hoesl CE. ISA, ISSAM and EAU recommendations for the investigation, treatment and monitoring of late-onset hypogonadism in males: scientific background and rationale. Aging Male 2005; 8: 59-74.
- 14. Bhasin S, Cunningham GR, Hayes FJ, et al. Testosterone therapy in adult men with androgen deficiency syndromes: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 2006; 91: 1995-2010. Erratum in: J Clin Endocrinol Metab 2006; 91: 2688.
- 15. Nieschlag E, Swerdloff R, Behre HM, et al. Investigation, treatment and monitoring of late-onset hypogonadism in males: ISA, ISSAM, and EAU recommendations. Int J Androl 2005; 28: 125-127.
- 16. Conway AJ, Handelsman DJ, Lording DW, et al. Use, misuse and abuse of androgens. The Endocrine Society of Australia consensus guidelines for androgen prescribing. Med J Aust 2000; 172: 220-224. Erratum in: Med J Aust 2000; 172: 334. <MJA full text>
- 17. World Health Organization. Obesity: preventing and managing the global epidemic. Report on a WHO consultation. WHO: Geneva, 1997.
- 18. Strain GW, Zumoff B, Kream J, et al. Mild hypogonadotropic hypogonadism in obese men. Metabolism 1982; 31: 871-875.
- 19. Vermeulen A, Verdonck L, Kaufman JM. A critical evaluation of simple methods for the estimation of free testosterone in serum. J Clin Endocrinol Metab 1999; 84: 3666-3672.
- 20. Lean ME, Han TS, Morrison CE. Waist circumference as a measure for indicating need for weight management. BMJ 1995; 311: 158-161.
- 21. Giagulli VA, Kaufman JM, Vermeulen A. Pathogenesis of the decreased androgen levels in obese men. J Clin Endocrinol Metab 1994; 79: 997-1000.
- 22. Kley HK, Solbach HG, McKinnan JC, et al. Testosterone decrease and oestrogen increase in male patients with obesity. Acta Endocrinol (Copenh) 1979; 91: 553-563.
- 23. Morley JE, Patrick P, Perry HM 3rd. Evaluation of assays available to measure free testosterone. Metabolism 2002; 51: 554-559.
- 24. Sikaris K, McLachlan RI, Kazlauskas R, et al. Reproductive hormone reference intervals for healthy fertile young men: evaluation of automated platform assays. J Clin Endocrinol Metab 2005; 90: 5928-5936.
- 25. Wu FC, von Eckardstein A. Androgens and coronary artery disease. Endocr Rev 2003; 24: 183-217.





Abstract
Objective: To determine the influence of obesity on the diagnosis of age-related androgen deficiency (AD) in symptomatic men according to current Australian guidelines.
Design, setting and participants: A community-based cohort of healthy ageing men with symptoms suggestive of AD was studied between May 2001 and February 2003. Men were classified as obese or non-obese according to body mass index (BMI) or waist circumference (WC).
Main outcome measure: Diagnosis of AD according to Endocrine Society of Australia (ESA) guidelines.
Results: 223 men aged 54–86 years with mean BMI 27.3 ± 0.2 kg/m2 (range 20.5–36.2 kg/m2) were recruited; 99 men were obese (BMI ≥ 30.0 kg/m2 or WC ≥ 102 cm) and 124 men were non-obese. Obese men had lower total testosterone (TT) (12.7 ± 0.4 v 15.0 ± 0.4 nmol/L); P < 0.001) and calculated free testosterone (275.7 ± 7.8 v 299.3 ± 7.4 pmol/L); P = 0.03) levels than non-obese men. TT levels < 8 nmol/L were recorded in 12% of obese men and 1% of non-obese men. Applying the ESA guidelines for the diagnosis of age-related AD, 15 obese men (15%) and 4 non-obese men (3%) were classified as being eligible for androgen therapy supported by the Pharmaceutical Benefits Scheme (PBS); the relative risk in obese men was 1.92 (95% CI, 1.44–2.55; P < 0.001).
Conclusion: Obesity is an important determinant of serum TT levels in ageing men. Almost one in seven obese men but only one in 30 non-obese men in our study were eligible for PBS-supported androgen therapy according to Australian guidelines. Although obese men are more likely to have biochemical hypoandrogenism, the clinical implications of this remain uncertain. Studies of testosterone therapy in this group of ageing men are needed to determine whether androgen replacement is beneficial.