Environmental iodine deficiency causes a wide spectrum of devastating mental and physical disorders, collectively described as iodine deficiency disorders.1 While endemic goitre is the most visible consequence of iodine deficiency, the most significant and profound consequences are on the developing brain.2 Impaired intellectual development of people living in iodine-deficient regions is of particular concern, especially when all of the adverse effects of iodine deficiency can be prevented by long-term, sustainable iodine prophylaxis.
For over two decades the Australian Centre for Control of Iodine Deficiency Disorders (ACCIDD) has performed sporadic surveys of urinary iodine excretion (UIE) levels in Australia. In 1992, we reported that the median UIE levels in the Australian population were above 200 μg/L, consistent with iodine sufficiency.3 However, this situation has changed dramatically in recent years, with several studies from Victoria, New South Wales4-8 and Tasmania,9,10 showing median UIE levels of < 100 μg/L. In pregnant women, median UIE levels have been found to be well below 100 μg/L, indicating that around half of these women in NSW, Victoria and Tasmania are iodine deficient.4,5
The decline in iodine intake in Australia appears to be due to changes within the dairy industry, with chlorine-containing sanitisers now replacing iodine-containing sanitisers. Iodine released from these chemicals into milk has been the major source of dietary iodine in Australia for at least four decades, but is now declining. Another contributory factor to the re-emergence of iodine deficiency in Australia has been the decreasing consumption of iodised salt.3 To our knowledge, few if any food manufacturers use iodised salt in the preparation and manufacture of foods.
Our study was conducted with the aim of documenting the population iodine nutritional status of schoolchildren in Australia.
The sampling scheme was a one-stage random cluster sample, the cluster sampling frame comprising all Year 4 school classes in government and non-government schools. Children in Year 4 are typically aged 8–10 years. Cluster sampling is often used when a complete list of individuals in the population of interest is unavailable, but a complete list of groups or “clusters” of these individuals is available.11
Class sizes were obtained and entered into an MS Excel (Microsoft Corporation, Redmond, Calif, USA) database. If a school had more than one class in the target cluster population, each class was entered separately into the database. If the exact class size was unknown, the average class size of 25 was used. For composite classes, it was assumed that the children were distributed evenly between the two classes in the composite.
The Survey Toolbox software program Random Village component was used to generate a randomly sorted list of classes,11 which were then selected in order until the target sample size (400 students from each state) was achieved.
If selected schools declined the invitation to participate in the study, more schools were selected in the same fashion. Tasmania was excluded from the study because a voluntary iodine fortification program, using iodised salt in bread, is ongoing in that state; and the Northern Territory was excluded for logistical reasons.
Our study, which aimed to provide a snapshot of iodine nutrition in Australian schoolchildren, aged 8–10 years, occurred between July 2003 and December 2004. We used a purpose-built vehicle known as a “ThyroMobil” (Merck, Darmstadt, Germany), which has been used for similar studies of iodine nutrition in other countries.12-14 At each school, students who brought their first morning urine sample to school with a pre-labelled specific identification number were registered, weighed and their height was measured (to calculate body surface area [BSA]) and they underwent a thyroid ultrasound scan.
Urine samples were collected and stored frozen in the ThyroMobil before being couriered back to Sydney where they were stored at − 20°C until assayed. Urinary iodine measurements were performed by ammonium persulfate digestion15 before a Sandell–Kolthoff reaction in a microtitre plate format.16
Eighty-eight classes (in 88 schools), from the 135 classes selected across five states, participated in the study, with a class participation rate of 65%. In total, 1709 students (comprising 881 boys and 828 girls) participated, representing 85% of the estimated target number of students (Box 1).
Box 1 shows the median UIE levels and interquartile ranges for the five states, and in Box 2 the relative frequency distributions of UIE level by state are grouped according to World Health Organization criteria for severity of iodine deficiency.17 Box 2 also shows that, in NSW and Victoria, 58.8% and 72.6% of children, respectively, had UIE levels < 100 μg/L. Similarly, close to half the South Australian children (47.5%) had UIE concentrations < 100 μg/L, indicating inadequate iodine nutrition.
The log UIE levels varied markedly by state (P < 0.001). After adjusting for sex, BSA and age, Victorian children had significantly lower adjusted levels than children in the other four states (P < 0.001). There was no statistically significant difference between the adjusted log UIE levels for children from NSW and South Australia (P = 0.06), which were significantly less (P < 0.001) than those for children from Western Australia and Queensland, which were comparable (P = 0.8).
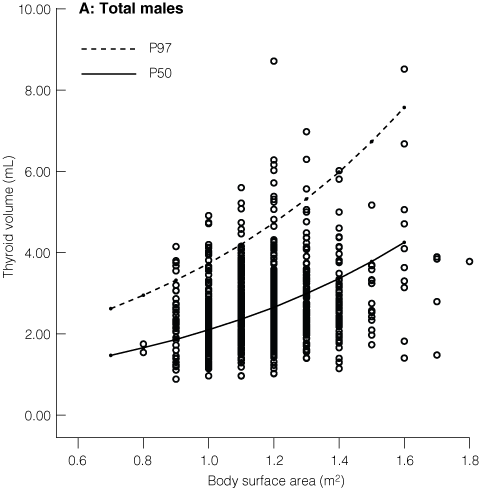
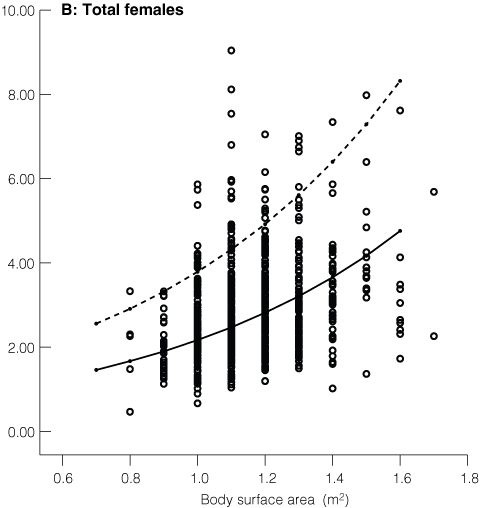
Box 3 shows the distribution of ultrasound measured thyroid volume (mL) by BSA and sex for children from all five states, and gives the international normative 50th and 97th percentile values (P50 and P97).18 Box 4 shows the percentage of study children with a thyroid volume exceeding the P50 or P97 by sex, state and BSA or age. With the exception of Victoria, Australian children had relatively larger thyroid glands compared with new international reference values for thyroid gland size in iodine-sufficient schoolchildren when adjusted by BSA or by age.
The National Iodine Nutrition Study is the largest study of its kind ever carried out in Australia, involving over 1700 students from 88 schools across five states. Random cluster sampling of classes generated a sample quickly and easily and gave each class an equal chance of being chosen, thereby providing an unbiased estimate of the overall population value.
Mainland Australian children as a group are borderline iodine deficient. However, there are significant and unexpected variations across the continent, with, overall, Western Australian and Queensland children being iodine replete and Victorian and NSW children being mildly iodine deficient. The results from NSW and Victoria are very similar to those previously reported.5-7 Tasmania was not included, but a study in 2001, before a bread fortification program was commenced, showed that the median UIE in Tasmanian schoolchildren was 84 μg/L, with 20% having levels under 50 μg/L.10 These results for Tasmania are similar to our results for NSW and Victoria.
About half (46.3%) of all the students tested in mainland Australia had UIE levels in the range of mild (36.7%) to moderate (9.6%) iodine deficiency. Most of these children came from south-eastern Australia. Victoria (18.8%) and NSW (13.6%) had the highest percentage in the range of 20–49 μg/L.
Children living in Western Australia and Queensland are clearly ingesting more iodine than their counterparts living elsewhere in Australia. We are not sure of the reasons for these regional variations, but the most likely explanations include:
possible differences in the proportion of the population using iodised salt;
variations in regional milk iodine content; and
drinking water iodine levels.
These possibilities are currently being investigated. For example, the limited number of drinking water samples collected during the survey showed relatively high iodine levels in water and milk in northern and central Queensland, which could explain why the UIE levels indicated iodine sufficiency in this state.
Thyroid ultrasound, along with measurement of UIE levels, has been recommended by the WHO and the International Council for the Control of Iodine Deficiency Disorders (ICCIDD) for monitoring the sustained impact of iodine deficiency control programs through universal salt iodisation. Since their introduction in 1997, the recommended normative values for thyroid volume in children have been revised on two occasions. The validity of the 1997 WHO/ICCIDD recommended values were challenged by a number of studies, including surveys using a ThyroMobil.14 It was later agreed that these reference values had significantly overestimated thyroid volumes in children.18 Indeed, in the study in Tasmania in 2000, 5.3% (based on age) and 5.9% (based on BSA) of Tasmanian boys had increased thyroid volumes when compared with the 1997 international normative values. When compared with the 2001 corrected WHO/ICCIDD reference values,18 the prevalence of raised thyroid volumes increased to 24.6% (based on age) and 22% (based on BSA).10 A new set of international reference values for thyroid volume assessed by ultrasound examination, based on studies of children living in areas of long-term iodine sufficiency, were released in 2003.19 The new reference values once again have lowered the reference volumes compared with the 2001 corrected values. The thyroid volume results in our study were compared with these new international reference values.
The thyroid volume results showed varying degrees of increased thyroid size across Australian states when compared with the international reference standards. Western Australian children had the largest glands, despite having the highest median UIE level of the five states. The explanation for this phenomenon is not clear. It may relate to factors other than iodine, such as dietary or environmental goitrogens. No association between spot urinary iodine level and thyroid volume assessed by ultrasound examination among schoolchildren has been reported.20 With the exception of Victorian schoolchildren, we can conclude that thyroid volumes in Australian schoolchildren are marginally increased when compared with international normative data obtained from children in iodine sufficient countries, but this increase does not allow us to conclude that iodine deficiency is causing endemic goitre in Australia.
The Victorian thyroid volume data were not in keeping with the general data trends in the other four states. Most of the thyroid volumes measured in Victorian children were below the 50th percentile and none were above the 97th percentile of the international standards. We believe that this is a consequence of methodological error underestimating thyroid volume, and highlights the importance of training and standardisation of methods among observers to minimise the measurement error.20 Victoria was the only state in which thyroid ultrasound measurements were performed by two independent investigators. While these two investigators received some instruction from the reference sonographer, the in-field calibration exercise performed in other states was not logistically possible at the time the data were collected.
It is alarming that mild iodine deficiency is extensive in children living in NSW, Victoria, Tasmania and South Australia and no action has been taken by public health authorities to increase iodine intake in our population. It is reasonable to assume that pregnant women and breastfeeding mothers are also iodine deficient, putting the next generation of children born in this country at risk of the neuropsychological consequences of iodine deficiency.21 The implementation of mandatory fortification of all edible salt for human consumption is long overdue in this country.
1 Summary data on participating schoolchildren by state
State |
Students participated/ students targeted |
M : F ratio |
Mean (SD) age (years) |
Mean (SD) weight (kg) |
Mean (SD) height (cm) |
Mean (SD) body surface area (m2) |
Median urinary iodine excretion (μg/L) (interquartile range) |
||||||||
NSW |
427/400 (106%) |
1 : 1 |
9.3 ± 0.6 |
34.9 ± 8.5 |
138.5 ± 6.7 |
1.16 ± 0.15 |
89.0 (65.0–123.5) |
||||||||
VIC |
348/400 (87%) |
1 : 0.8 |
9.7 ± 0.5 |
38.2 ± 8.9 |
141.0 ± 6.8 |
1.22 ± 0.15 |
73.5 (53.0–104.3) |
||||||||
SA |
317/400 (79%) |
1 : 0.9 |
9.0 ± 0.5 |
35.3 ± 7.9 |
137.3 ± 7.3 |
1.16 ± 0.15 |
101.0 (74.0–130.0) |
||||||||
WA |
323/400 (80%) |
1 : 0.8 |
8.9 ± 0.6 |
32.8 ± 7.6 |
136.9 ± 6.4 |
1.11 ± 0.14 |
142.5 (103.5–214.0) |
||||||||
QLD |
294/400 (73%) |
1 : 1.3 |
9.1 ± 0.4 |
32.9 ± 7.2 |
137.3 ± 6.3 |
1.12 ± 0.13 |
136.5 (104.3–183.8) |
||||||||
Total |
1709/2000 (85%) |
1 : 0.9 |
9.2 ± 0.6 |
34.9 ± 8.3 |
138.3 ± 6.9 |
1.20 ± 0.10 |
104.0 (71.0–147.0) |
||||||||
2 Relative frequency distribution of urinary iodine excretion (UIE) levels in schoolchildren by state
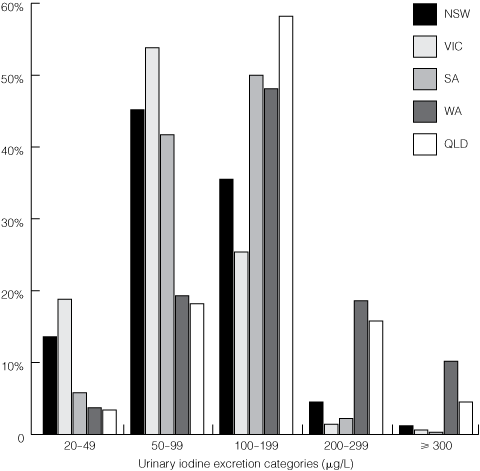
Iodine replete, UIE ≥ 100 μg/L; mild iodine deficiency, UIE 50–99 μg/L; moderate iodine deficiency, UIE 20–49 μg/L.
3 Distribution of thyroid volume in schoolchildren by body surface area and sex. International normative 50th and 97th percentiles (P50 and P97) are shown
4 Percentage of children with a thyroid volume (mL) greater than the new international standard 50th and 97th percentile values (P50 and P97)
|
Based on body surface area (95% CI) |
Based on age (95% CI) |
|||||||||||||
State |
Boys |
Girls |
Boys |
Girls |
|||||||||||
Percentage > international standard P50 |
|||||||||||||||
NSW |
58.1 (51.3–64.9) |
57.6 (50.9–64.3) |
61.3 (54.6–68.0) |
63.8 (57.3–70.3) |
|||||||||||
VIC |
18.0 (12.6–23.5) |
18.3 (12.2–24.4) |
24.9 (18.8–31.0) |
26.1 (19.2–33.1) |
|||||||||||
SA |
41.0 (33.5–48.4) |
46.3 (38.3–54.3) |
50.9 (43.3–58.5) |
54.4 (46.4–62.4) |
|||||||||||
WA |
70.0 (63.1–76.9) |
71.6 (64.4–78.9) |
75.3 (68.8–81.8) |
81.1 (74.8–87.4) |
|||||||||||
QLD |
55.0 (46.5–63.6) |
50.0 (42.3–57.5) |
61.5 (53.2–69.9) |
60.6 (53.1–68.2) |
|||||||||||
Total |
47.7 (44.3–51.0) |
49.3 (45.8–52.7) |
53.9 (50.6–57.3) |
57.6 (54.2–60.9) |
|||||||||||
Percentage > international standard P97 |
|||||||||||||||
NSW |
3.9 (1.3–6.6) |
7.1 (3.7–10.6) |
6.4 (3.0–9.7) |
10.0 (5.9–14.1) |
|||||||||||
VIC |
0 |
0 |
0 |
0.7 (0–1.9) |
|||||||||||
SA |
4.8 (1.6–8.1) |
10.1 (5.2–14.9) |
6.6 (2.8–10.3) |
10.7 (5.8–15.7) |
|||||||||||
WA |
11.2 (6.4–15.9) |
14.9 (9.1–20.6) |
11.8 (6.9–16.6) |
19.6 (13.2–26.0) |
|||||||||||
QLD |
2.3 (0–4.9) |
3.1 (0.4–5.8) |
3.1 (0.1–6.0) |
5.6 (2.1–9.2) |
|||||||||||
Total |
4.4 (3.0–5.8) |
7.0 (5.2–8.7) |
5.6 (4.0–7.1) |
9.3 (7.3–11.3) |
|||||||||||
- Mu Li1
- Creswell J Eastman2
- Kay V Waite3
- Gary Ma4
- Karen Byth5
- Margaret R Zacharin6
- Duncan J Topliss7
- Philip E Harding8
- John P Walsh9
- Lynley C Ward10
- Robin H Mortimer11
- Emily J Mackenzie12
- Zelda Doyle13
- 1 School of Public Health, University of Sydney, Sydney, NSW.
- 2 Institute of Clinical Pathology and Medical Research (ICPMR), Westmead Hospital, Sydney, NSW.
- 3 Royal Children's Hospital, Melbourne, VIC.
- 4 Department of Endocrinology and Diabetes, The Alfred Hospital, Melbourne, VIC.
- 5 Adelaide, SA.
- 6 Department of Endocrinology and Diabetes, Sir Charles Gairdner Hospital, Perth, WA.
- 7 Royal Brisbane and Women’s Hospital, Brisbane, QLD.
- 8 Broad Street Consultants, Hobart, TAS.
We thank all the school principals, staff, students and their parents/guardians who participated in this study. We are grateful to Cerebos Australia Limited for financial support for the study, and to the Merck Company (Germany) for providing the ThyroMobil. We thank all volunteers who assisted with the field studies and with driving the ThyroMobil.
Australian National Iodine Nutrition Study —National Steering Committee Members: C J Eastman (Project Chairman), M Li (Project Coordinator), G Ma, K Waite, S Boyages, K Guttikonda, A McElduff, M Zacharin, D Topliss, J Stockigt, G Wilcox, B Hetzel, P Harding, R Mortimer, J Walsh, B Stuckey, P van Buynder, J Burgess, T Greenaway, K Hynes, J Seal, J Lewis, B Garside, F Heazlewood.
None identified.
- 1. Hetzel BS. Iodine deficiency disorders and their eradication. Lancet 1983; 2: 1126-1129.
- 2. Boyages S. Clinical review 49: iodine deficiency disorders. J Clin Endocrinol Metab 1993; 77: 587-591.
- 3. Eastman CJ. Where has all our iodine gone? [editorial] Med J Aust 1999; 171: 455-456. <MJA full text>
- 4. Gunton JE, Hams G, Fiegert M, McElduff A. Iodine deficiency in ambulatory patients attending a Sydney teaching hospital: is Australia truly iodine replete? Med J Aust 1999; 171: 467-470. <MJA full text>
- 5. Li M, Ma G, Guttikonda K, et al. Re-emergence of iodine deficiency in Australia. Asia Pac J Clin Nutr 2001; 10: 200-203.
- 6. McElduff A, McElduff P, Gunton JE, et al. Neonatal thyroid-stimulating hormone concentrations in northern Sydney: further indications of mild iodine deficiency? Med J Aust 2002, 176: 317-320. <MJA full text>
- 7. McDonnell CM, Harris M, Zacharin MR. Iodine deficiency and goitre in schoolchildren in Melbourne, 2001. Med J Aust 2003; 178: 159-162. <MJA full text>
- 8. Guttikonda K, Travers CA, Lewis PR, Boyages S. Iodine deficiency in urban primary school children: a cross-sectional analysis. Med J Aust 2003; 179: 346-348. <MJA full text>
- 9. Hynes K. Urinary iodine status of Tasmanian primary schoolchildren. Final project report. Hobart: Menzies Centre for Population and Health Research, University of Tasmania, December 2001.
- 10. Guttikonda K, Burgess JR, Hynes K, et al. Recurrent iodine deficiency in Tasmania, Australia: a salutary lesson in sustainable iodine prophylaxis and its monitoring. J Clin Endocrinol Metab 2002; 87: 2809-2815.
- 11. Cameron AR. Survey Toolbox for livestock diseases — a practical manual and software package for active surveillance in developing countries. Canberra: Australian Centre for International Agricultural Research, 1999: 330 pp. (ACIAR Monograph No. 54.)
- 12. Delange F, Benker G, Caron P, et al, Thyroid volume and urinary iodine in European schoolchildren: standardisation of values for measurement of iodine deficiency. Eur J Endocrinol 1997; 136: 180-187.
- 13. Pretell E, Delange F, Hostalek U, Members of the Latin American Thyromobil Study Group. Advantages of the Thyromobil in rapidly assessing iodine nutrition in eleven Latin American countries. Endocrine J 2000; 47: 162. (Abstract No. P-228.)
- 14. Djokomoeljanto R, Setyawan H, Dramaix M, Hadisaputro S, et al. The ThyroMobil model for standardised evaluation of iodine deficiency disorders control in Indonesia. Thyroid 2001; 11: 365-372.
- 15. Pino S, Fang, S, Braverman LE. Ammonium persulphate: a safe alternative oxidising reagent for measuring urinary iodine. Clin Chem 1996; 42: 239-243.
- 16. Ohashi T, Yamaki M, Pandav C, et al. Simple microplate method for determination of urinary iodine. Clin Chem 2000; 46: 529-536.
- 17. World Health Organization. WHO, UNICEF, ICCIDD. Indicators for assessing iodine deficiency disorders and their control through salt iodisation. WHO/NUT/94.6. Geneva: WHO, 1994.
- 18. Zimmermann MB, Molinari L, Spehl M, et al. Toward a consensus on reference values for thyroid volume in iodine-replete schoolchildren: results of a workshop on inter-observer and inter-equipment variation in sonographic measurement of thyroid volume. Eur J Endocrinol 2001; 144: 213-220.
- 19. Zimmermann MB, Hess SY, Molinari L, et al. New reference values for thyroid volume by ultrasound in iodine sufficient schoolchildren: a WHO/NHD Iodine Deficiency Study Group Report. Am J Clin Nutr 2004; 79: 231-237.
- 20. Fuse Y, Igari T, Yamada C, et al. Epidemiological survey of thyroid volume and iodine intake in schoolchildren, postpartum women and neonates living in Ulaan Baatar. Clin Endocrinol (Oxf) 2003; 59: 298-306.
- 21. Eastman CJ. Iodine supplementation: the benefits for pregnant and lactating women in Australia and New Zealand. O & G (RANZCOG) 2005; 7(1 Autumn): 65-66.





Abstract
Objective: To document the population iodine nutritional status in Australian schoolchildren.
Design and setting: Cross-sectional survey of schoolchildren aged 8–10 years, based on a one-stage random cluster sample drawn from all Year 4 school classes in government and non-government schools in the five mainland Australian states of New South Wales, Victoria, South Australia, Western Australia and Queensland. The study was conducted between July 2003 and December 2004.
Participants: 1709 students from 88 schools (881 boys and 828 girls), representing 85% of the estimated target number of students. The class participation rate was 65%.
Main outcome measures: (i) Urinary iodine excretion (UIE) levels (compared with the criteria for the severity of iodine deficiency of the World Health Organization/International Council for the Control of Iodine Deficiency Disorders: iodine replete, UIE ≥ 100 μg/L; mild iodine deficiency, UIE 50–99 μg/L; moderate iodine deficiency, UIE 20–49 μg/L; severe iodine deficiency, UIE < 20 μg/L); (ii) Thyroid volumes measured by ultrasound (compared with new international reference values).
Results: Overall, children in mainland Australia are borderline iodine deficient, with a national median UIE of 104 μg/L. On a state basis, NSW and Victorian children are mildly iodine deficient, with median UIE levels of 89 μg/L and 73.5 μg/L, respectively. South Australian children are borderline iodine deficient, with a median UIE of 101 μg/L. Both Queensland and Western Australian children are iodine sufficient, with median UIE levels of 136.5 μg/L and 142.5 μg/L, respectively. Thyroid volumes in Australian schoolchildren are marginally increased compared with international normative data obtained from children living in iodine sufficient countries. There was no significant association between UIE and thyroid volume.
Conclusion: Our results confirm the existence of inadequate iodine intake in the Australian population, and we call for the urgent implementation of mandatory iodisation of all edible salt in Australia.