|
Research
Quantitative heel ultrasound as a predictor for osteoporosis
Vasi Naganathan, Lyn March, David Hunter, Nick A Pocock,
Joanna
Markovey and Philip N Sambrook
MJA 1999; 171: 297-300
For related articles, see Prince, Maguire & Lobb et al
Abstract -
Introduction -
Methods -
Results -
Discussion -
References -
Authors' details
-
-
More articles on Rheumatology
|
Abstract |
Objective: To determine the diagnostic value of
quantitative ultrasound (QUS) to predict bone mineral density (BMD)
categories as defined by dual-energy x-ray absorptiometry.
Design: Cross-sectional survey.
Setting: Rheumatology department of a tertiary care
hospital (Royal North Shore Hospital, Sydney, NSW), 1997-1998.
Subjects: 326 healthy women aged 45-80 years who had
volunteered for a twin study. Our study included both members of
non-identical twin pairs but only one randomly selected member of
identical twin pairs.
Main outcome measures: BMD categories as defined by
dual-energy x-ray absorptiometry of lumbar spine and left hip, and
QUS of calcaneus; sensitivity, specificity and likelihood ratios
(LRs) of QUS parameters to diagnose osteoporosis as defined by
BMD.
Results: The sensitivity of QUS to diagnose BMD
osteoporosis varied between 9% and 47%, depending on the QUS
parameter. The specificity of QUS was high (88%-100%). If all QUS
parameters were normal, osteoporosis was unlikely (LR, 0-0.2). One
QUS parameter, broadband ultrasound attenuation (BUA), was highly
predictive of osteoporosis by BMD when in the osteoporotic range
(LR, Infinity), but had low sensitivity (9%). QUS results in the
osteoporotic range for other parameters and all QUS results in the
osteopenic range were less predictive (LR, 1.0-5.2) of osteoporotic
BMD.
Conclusion: These results suggest that, for most of those
tested for osteoporosis by QUS in the community, uncertainty remains
about expected BMD.
|
| | Introduction |
Quantitative heel ultrasound has recently been introduced in
Australian pharmacies as a "screening" tool for osteoporosis. The
technology is relatively cheap, radiation-free and portable, but
its accuracy in diagnosing osteoporosis is unclear.
Bone mineral density (BMD), measured by dual energy x-ray
absorptiometry (DEXA), is the best predictor of fracture risk and is
currently considered the "gold standard" for diagnosing
osteoporosis. Although prospective studies have shown that
quantitative ultrasound (QUS) predicts future fracture risk
independently of BMD,1,2 most women with abnormal
results will proceed to formal BMD measurement to assess the need for
therapeutic intervention; women with normal QUS results may be
reassured they do not need BMD measurement. It is unclear how many
women have unnecessary further investigations or are falsely
reassured. When used in this way, QUS has diagnostic value only if it
can accurately predict BMD categories as determined by DEXA.
Our aim was to examine the role of QUS in predicting BMD diagnostic
categories. We determined conventional sensitivity and
specificity, as well as likelihood ratios (LRs). These have the
advantage of allowing test results to be assessed for several
diagnostic categories rather than only at a single cut-off between
"normal" and "abnormal".3 |
| |
Methods |
| |
Subjects and setting | |
The study was a cross-sectional survey of healthy women aged 45-80
years who had volunteered to take part in a twin study. They were
recruited from the Australian Twin Registry and media advertising.
Our ultrasound study included both members of each non-identical
twin pair and one randomly selected member of each identical twin
pair.
The study was conducted in the Rheumatology Department of the Royal
North Shore Hospital, Sydney, NSW (a tertiary care hospital), in 1997
and 1998. It was approved by the hospital's Human Research Ethics
Committee.
| |
Assessment |
Subjects had BMD measurements of their lumbar spine (L1-L4) and left
hip (neck of femur and total hip) by DEXA using a Hologic QDR450
instrument (Hologic Inc, Waltham, Mass, USA). The same
machine was used on all patients.
QUS of the left calcaneus was performed on the same day using a CUBA Mark
II ultrasound instrument (McCue Ultrasonics, London, UK).
The two most commonly used QUS parameters were measured:
broadband ultrasound attenuation (BUA), which is thought to reflect
bone mass and architecture, and velocity of sound (VOS), which
reflects mass and elasticity of bone.4 | |
Analyses |
Each BMD and QUS value was converted to a T score (number of standard
deviations from the population mean for young, healthy, sex-matched
adults). This population mean was estimated from measurements in 50
women aged 20-30 years who also took part in the twin study. T scores
were used to categorise BMD values as normal (T > -1) or indicating
osteopenia (T, -2.5 to -1) or osteoporosis (T < -2.5), as proposed
by a working party of the World Health Organization.5 QUS values were
classified in the same way. Although no consensus has been reached on
what T-score cut-offs and diagnostic categories to use with QUS, the
instrument used commonly in Australian pharmacies uses the WHO
criteria and cut-off values.
Subjects were classified as having osteoporosis if at least one of the
three BMD measurements (lumbar spine, neck of left femur or total left
hip) indicated osteoporosis, and as having osteopenia if at least one
measurement indicated osteopenia but none indicated osteoporosis.
BUA and VOS results were combined as a cQUS category: this was defined
as normal if both results were normal, as osteopenic if either
indicated osteopenia but neither indicated osteoporosis, and as
osteoporotic if either indicated osteoporosis. Some QUS
scanners calculate a stiffness parameter (unrelated to mechanical
stiffness) from a linear combination of normalised BUA and VOS
values. We calculated stiffness in an analogous manner,6 and categorised
it as normal, osteopenic or osteoporotic based on T scores in the same
way as other QUS values.
We calculated the sensitivity and specificity of QUS parameters in
predicting BMD-defined osteoporosis and osteopenia. We also
calculated the likelihood ratio (LR) for each QUS result
(sensitivity/1 - specificity), defined as the ratio of
the probability of the particular QUS result (normal, osteopenic or
osteoporotic) in women with BMD-defined osteoporosis or
osteopenia to the probability of the same result in women with normal
BMD.7
As there is no consensus on what QUS cut-off values should be used to
diagnose osteoporosis, the statistical analyses were repeated
using a range of QUS T-score cut-off values for osteoporosis between
-2.5 and -1.0.
|
| |
Results |
| |
Subjects and osteoporosis | |
There were 326 subjects, with mean age 58.5 years; 255 (78%) were
postmenopausal. Of the 326, 47 (14%) had a BMD measurement indicating
osteoporosis at one or more of the three sites where BMD was measured,
and a further 160 (49%) had a measurement indicating osteopenia.
QUS results are compared with BMD results in Box 1. The percentage of women with values in the osteoporotic range varied between QUS parameters (1% for BUA, 17% for VOS and the combined BUA-VOS category, and 14% for stiffness).
| |
Sensitivity and specificity of QUS | |
Sensitivity and specificity of QUS for predicting BMD diagnostic
categories are shown in Box 2. Sensitivity and specificity varied
between QUS parameters. A BUA result in the osteoporotic range (T <
-2.5) had very low sensitivity for predicting BMD-defined
osteoporosis (9%), but high specificity (100%). In contrast, VOS,
cQUS and stiffness results in the osteoporotic range had
sensitivities of almost 50%, and specificities that were again high.
For predicting either osteoporosis or osteopenia, stiffness had the
best combination of sensitivity (77%) and specificity (81%).
Positive and negative predictive values are also shown in Box 2.
Negative predictive values were high (87%-91%) for QUS as a predictor
of BMD-defined osteoporosis versus osteopenia/normal BMD. This
indicated that a woman with BMD-defined osteoporosis was unlikely to
have a QUS result in the normal-osteopenic range.
| |
Likelihood ratios of QUS | |
LRs for different QUS results to predict BMD-defined osteoporosis
are summarised in Box 2 and interpreted in Box 3.
Normal QUS result: A BUA, VOS, cQUS or stiffness result
in the normal range had a low LR (0-0.2) (ie, a normal result
significantly lowered the odds or probability of the woman's having
BMD-defined osteoporosis).
Osteoporotic QUS result: A BUA result in the osteoporotic range
had an LR approaching infinity and so was highly predictive of
BMD-defined osteoporosis. In contrast, a VOS, cQUS or
stiffness result in the osteoporotic range had a much lower LR
(4.0-5.2), increasing the odds of BMD-defined osteoporosis, but to a
lesser extent than a BUA result in the osteoporotic range.
Osteopenic QUS result: QUS results in the osteopenic range were
less predictive, as LR ranged from 1.0 to 2.4. Between 37% and 50% of
subjects (depending on the QUS parameter) had results in this range
(Box 1).
LRs for predicting low BMD (ie, osteoporosis or osteopenia; BMD T
score < -1) are also shown in Box 2, and followed a similar pattern to
LRs for predicting BMD-defined osteoporosis.
When QUS T-score cut-off values for osteoporosis were increased from
-2.5 to -1.0, sensitivity increased, but at the expense of decreasing
specificity and LR (data not shown). For example, a BUA cut-off of -1.0
gave 83% sensitivity, 69% specificity and LR, 2.6. Corresponding
values for a VOS cut-off of -1.0 were 96%, 41% and 1.6.
|
| |
Discussion |
We found that QUS had variable usefulness in predicting BMD
categories. Specificity for predicting BMD-defined osteoporosis
was high for all QUS parameters (88%-100%), but sensitivity was low
and variable (9%-47%). A BUA result in the osteoporotic range was
highly predictive of BMD-defined osteoporosis (LR, Infinity), but
had low sensitivity (9%). Results in the osteoporotic range for other
QUS parameters and in the osteopenic range for all QUS parameters were
less predictive of BMD-defined osteoporosis (LRs, 1.0-5.2).
In the light of our study, how can we interpret QUS results?
- If a BUA result is in the osteoporotic range (LR, Infinity), then BMD-defined
osteoporosis is almost certain (predictive value, 100%). However,
the low sensitivity of BUA (9%) means that many women with
osteoporosis would be missed if BUA alone were used.
- If results are
normal for both BUA and VOS (cQUS normal; LR, 0), we can
confidently rule out BMD-defined osteoporosis. If LR is 0, then, no
matter what the pre-test probability of osteoporosis, the post-test
probability will be < 5% (Box 3).
- All results in the osteopenic range, and VOS and stiffness results in
the osteoporotic range, are less predictive of BMD category.
Therefore, if QUS were performed on a population similar to ours (14%
prevalence of osteoporosis), then (from Box 1) 1% would have a BUA
result in the osteoporotic range (likely to have osteoporosis) and
33% would have both BUA and VOS results in the normal range
(osteoporosis could fairly confidently be ruled out, with a
post-test probability < 5%). However, there would be a degree of
uncertainty about the remaining 66%, who would then need a DEXA scan to
identify those with osteoporosis.
Previous studies of QUS as a predictor of BMD have generally used
conventional sensitivity and specificity analyses only, not LRs,
and have not used the WHO BMD definitions. For example, two
community-based cross-sectional studies on 700
postmenopausal10 and 1000 perimenopausal
women,11 respectively, found that
there was a 40%-50% overlap in the number of women in the lowest
quartile of both DEXA and QUS measurements. Two other studies found
QUS parameters to have a sensitivity of 65%-70% for BMD in the lowest
quartile.6,12 Only one study other than
ours has evaluated QUS in terms of WHO BMD definitions. It found BUA and
VOS to have higher sensitivities, of 77% and 69%, respectively, for
diagnosing osteoporosis in 100 women aged 60-69 years.13These
higher sensitivities may have been due to use of higher BUA and VOS
cut-off values. As expected, specificities were lower than in our
study.
The ultrasound instrument used in this study, the McCue Cuba Mark
II, is not identical to the Achilles ultrasound instrument (Lunar,
Madison, Wis, USA) used in Australian pharmacies. Nevertheless, a
comparison of the two machines found that BUA measurements on a Cuba
Mark II instrument were highly correlated with "stiffness"
measurements on a Lunar Achilles instrument (r =
0.906; 95% CI, 0.873-0.931).14 Another study compared
measurements of 30 women between the Cuba Mark II used in our study and
an Achilles, finding an r value of 0.8.15 Therefore, it is unlikely
that the Achilles instrument would be a significantly better
predictor of BMD than our Cuba Mark II.
There is no consensus on what cut-off values to use with QUS to diagnose
osteoporosis. We found that changing the cut-off could achieve
higher sensitivity, but only by accepting higher rates of false
positives (lower specificity) and less discriminating LRs.
Although there is enough evidence to support the use of QUS as an
independent predictor of fracture risk,1,2 our study shows that QUS
should not been seen as a substitute for BMD measurement. The results
of our study suggest that, when women in the community are "screened"
for osteoporosis using QUS, a few women will be confidently
identified with BMD-defined osteoporosis. Another small group will
be able to be reassured that they are unlikely to have osteoporosis.
However, for the great majority, the presence or absence of
osteoporosis will remain uncertain.
|
| |
References | |
- Hans D, Dargent MP, Schott AM, et al. Ultrasonographic heel
measurements to predict hip fracture in elderly women: the EPIDOS
prospective study. Lancet 1996; 348 (9026): 511-514.
-
Bauer DC, Gluer CC, Cauley JA, et al. Broadband ultrasound
attenuation predicts fractures strongly and independently of
densitometry in older women. A prospective study. Study of
Osteoporotic Fractures Research Group. Arch Intern Med
1997; 157: 629-634.
-
Sackett DL, Haynes BR, Guyatt GH, et al. Clinical epidemiology. A
basic science for clinical medicine. 2nd ed. Boston: Little, Brown
and Company, 1991.
-
Gluer CC, Wu CY, Jergas M, et al. Three quantitative ultrasound
parameters reflect bone structure. Calcif Tissue Int 1994;
55: 46-52.
-
World Health Organization Study Group. Assessment of fracture
risk and its application to screening for prostmenopausal
osteoporosis. World Health Organ Tech Rep Ser 1994; 843:
1-129.
-
Herd RJ, Blake GM, Miller CG, et al. The ultrasonic assessment of
osteopenia as defined by dual X-ray absorptiometry. Br J Radiol
1994; 67: 631-635.
-
Fletcher RH, Fletcher SW, Wagner EH. Clinical epidemiology: the
essentials. 3rd ed. Baltimore: Williams & Wilkins, 1988: 65.
-
Fagan TJ. Normogram for Bayes theorem [letter]. N Engl J Med
1975: 293; 257.
-
Australian National Consensus Conference 1996. The prevention
and management of osteoporosis. Consensus statement. Med J
Aust 1997; 167 Suppl: S1-S15.
-
van Daele Burger H, Algra D, et al. Age-associated changes in
ultrasound measurements of the calcaneus in men and women: the
Rotterdam Study. J Bone Miner Res 1994; 9: 1751-1757.
-
Massie A, Reid DM, Porter RW. Screening for osteoporosis:
comparison between dual energy X-ray absorptiometry and broadband
ultrasound attenuation in 1000 perimenopausal women.
Osteoporos Int 1993; 3: 107-110.
-
Young H, Howey S, Purdie DW. Broadband ultrasound attenuation
compared with dual-energy X-ray absorptiometry in screening for
postmenopausal low bone density. Osteoporos Int 1993; 3:
160-164.
-
Langton CM, Ballard PA, Bennett DK, Purdie DW. A comparison of the
sensitivity and specificity of calcaneal ultrasound measurements
with clinical criteria for bone densitometry (DEXA) referral.
Clin Rheumatol 1997; 16: 117-118.
-
Greenspan SL, Bouxsein ML, Melton ME, et al. Precision and
discriminatory ability of calcaneal bone assessment technologies.
J Bone Miner Res 1997; 12: 1303-1313.
-
Harris ND, Griffiths MR, Nguyen TV, et al. Quantitative
ultrasound of the heel: a comparison of Lunar and McCue instruments
[abstract]. Proceedings of the Australian and New Zealand Bone and
Mineral Society 7th Annual Scientific Meeting. 1997. 29 Sept-1 Oct;
Canberra, ACT: 62.
(Received 24 Dec 1998, accepted 9 Jul 1999)
|
| | Authors' details |
Department of Rheumatology, Royal North Shore Hospital, Sydney,
NSW.
Vasi Naganathan, FRACP, Research Scholar;
Lyn March,
FRACP, FAFPHM, Staff Specialist;
David Hunter, MB BS,
Advanced Physician Trainee in Rheumatology;
Joanna Markovey,
MSc, Research Bone Densitometry Technician;
Philip N
Sambrook, FRACP, MD, Head of Department.
Department of Nuclear Medicine, St Vincent's Hospital, Sydney, NSW.
Nick A Pocock, FRACP, MD, Senior Staff Specialist.
Reprints will not be available from the authors.
Correspondence: Dr V
Naganathan, Department of Rheumatology, Royal North Shore
Hospital, St Leonards, NSW 2065.
Email: vasinATmed.usyd.edu.au
|
| |
1: Association between quantitative heel ultrasound (QUS) results and bone mineral density* in 326 women aged 45-80 years | | Bone mineral density
| | | QUS result | Normal (n = 119) | Osteopenia (n = 160) | Osteoporosis (n = 47) | Total (n = 326) |
| | Broadband ultrasound (BUA) | | Normal | 103 | 91 | 8 | 202 (62%) | | Osteopenia | 16 | 69 | 35 | 120(37%) | | Osteoporosis | 0 | 0 | 4 | 4 (1%) | | Velocity of sound (VOS) | | Normal | 81 | 33 | 2 | 116 (36%) | | Osteopenia | 36 | 96 | 23 | 155 (47%) | | Osteoporosis | 2 | 31 | 22 | 55 (17%) | | Combined category (cQUS) | | Normal | 79 | 28 | 0 | 107 (33%) | | Osteopenia | 38 | 101 | 25 | 164 (50%) | | Osteoporosis | 2 | 31 | 22 | 55 (17%) | | Stiffness | | Normal | 96 | 44 | 4 | 144 (44%) | | Osteopenia | 20 | 94 | 21 | 135 (41%) | | Osteoporosis | 3 | 22 | 22 | 47 (14%) |
| |
* Measured by dual energy x-ray absorptiometry. Combined result for broadband ultrasound and velocity of sound: normal if both normal; osteopenic if either osteopenic and neither osteoporotic; and osteoporotic if either osteoporotic.
|
| | Back to text | | |
2: Use of ultrasound parameters to predict osteoporosis or osteopenia defined by dual energy x-ray absorptiometry (DEXA) measurement of bone mineral density | | | | Predictive values | Likelihood ratio for ultrasound measurement (95% confidence interval) | | Sensitivity | Specificity | Positive | Negative | Normal | Osteopenia | Osteoporosis |
| | To predict osteoporosis | | Broadband ultrasound (BUA) | 9% | 100% | 100% | 87% | 0.2 (0.11-0.38) | 2.4 (1.9-3.1) | Infinity | | Velocity of sound (VOS) | 46% | 88% | 40% | 91% | 0.1 (0.03-0.4) | 1.0 (0.7-1.4) | 4.0 (2.6-6.2) | | Combined category* (cQUS) | 47% | 88% | 40% | 91% | 0 | (1.1 (0.8-1.5) | 4.0 (2.6-6.2) | | Stiffness | 47% | 91% | 46% | 91% | 0.2 (0.08-0.5) | 1.1 (0.8-1.6) | 5.2 (3.2-8.4) | | To predict osteoporosis or osteopenia | | Broadband ultrasound (BUA) | 52% | 87% | 87% | 51% | 0.6 (0.5-0.7) | 3.7 (2.3-6.0) | Infinity | | Velocity of sound (VOS) | 83% | 68% | 82% | 70% | 0.25 (0.18-0.35) | 1.9 (1.4-2.6) | 15.0 (3.7-60.5) | | Combined category* (cQUS) | 86% | 66% | 82% | 74% | 0.20 (0.14-0.29) | 1.9 (1.4-2.5) | 15.2 (3.8-61.3) | | Stiffness | 77% | 81% | 87% | 67% | 0.3 (0.2-0.4) | 3.3 (2.2-5.0) | 8.4 (2.7-26.5) |
| |
DEXA = dual energy x-ray absorptiometry. * Combined broadband ultrasound attenuation and velocity of sound category.
|
| | Back to text | | |
3: Interpretation of likelihood ratios
The likelihood ratio (LR) indicates how much a test result raises or lowers the probability of an individual's having "disease". It can be used to determine post-test probability of a disease from the estimated pre-test probability using
a normogram (Figure). Thus, if an early postmenopausal woman (aged 60-64 years) had a pre-test probability of osteoporosis of 15%,9 then, from the normogram, a cQUS result in the osteoporot ic range (LR, 4.0) would increase her probability to 40%. If she had a very high pre-test probability of osteoporosis, for example 50%, because of multiple risk factors (eg, including family history of osteoporosis and recent wrist fracture after a fall), then a
cQUS result in the osteoporotic range would increase her probability to 80%.
A BUA result in the osteoporotic range (LR, Infinity) would make BMD-defined osteoporosis highly likely, no matter what the pre-test risk. However, a cQUS result in the normal range (LR, 0) would make BMD-defined osteoporosis unlikely. QUS results in the osteopenic range, with LRs closer to unity (LR, 1.0-2.4), would make BMD category far less certain.
| 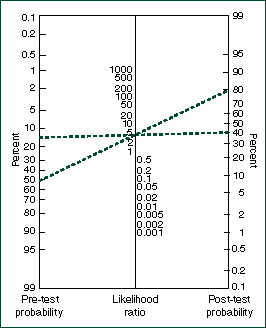
Normogram for applying likelihood ratios, adapted from Fagan.8 Lines show post-test probabilities when LR = 4.0 and pre-test probabilities are 15% and 50%, respectively.
|
| | Back to text | |
|





