Urinary diagnosis of gonorrhoea and chlamydia in men in remote Aboriginal communities
Steven J Skov, Penny Miller, Wayne Hateley, Ivan B Bastian, Jenny Davis and Peter W Tait
MJA 1997; 166: 468
For editorial comment see Waddell
Readers may print a single copy for personal use. No further reproduction or distribution of the articles should proceed without the permission of the publisher. For permission, contact the Australasian Medical Publishing Company
Journalists are welcome to write news stories based on what they read here, but should acknowledge their source as "an article published on the Internet by The Medical Journal of Australia <http://www.mja.com.au/>".
Abstract - Introduction - Methods - Results - Discussion - Acknowledgements - References - Authors' details
- ©MJA1997
Abstract |
Aims: (1) To evaluate the acceptability and validity
of an intervention based on urine tests for diagnosis and treatment of
gonorrhoea and chlamydia in men in remote Aboriginal communities.
(2) To provide a prevalence estimate of these infections in the male
population in the surveyed communities. Methods: First-void urine samples from 460 men in remote communities and 33 men in the Alice Springs Gaol were tested for gonorrhoea and chlamydia with at least one of polymerase chain reaction (PCR), enzyme immunoassay (EIA) and culture (gonorrhoea only). Results: One hundred and three men (20.9%) were infected with gonorrhoea or chlamydia. The prevalence of infection for gonorrhoea only was 11.7%, for chlamydia only 4.1% and for dual infection 5.1%. Eighty-eight infected men and 45 of their sexual partners were recorded as having been treated within two months of testing. PCR tests detected the largest number of infections and were the easiest to use. Conclusions: The prevalence of these infections was higher than anticipated. Urine PCR tests were acceptable to men and are well suited to the remote-community setting. As an effective alternative to urethral swabs, they permit a range of community-based strategies to address high rates of infection with gonorrhoea and chlamydia. |
Introduction |
The rates of sexually transmitted diseases (STDs) in central
Australia, particularly gonorrhoea and chlamydia, are among the
highest in Australia and indeed the world,1,2 despite a modest decline in
gonorrhoea over the past 15 years (according to notifiable diseases
data from the health departments of South Australia, Western
Australia and the Northern Territory). Although these infections
are more common in this region than in the rest of Australia among both
Aboriginal and non-Aboriginal people, most of the excess morbidity
occurs among Aboriginal people (health departments' data). Many
factors contribute to this situation. Dispossession, unemployment
and their sequelae are the fundamental determinants of the poor
health of Aboriginal people and also underlie the observed high
levels of STDs. The serious under-resourcing of central Australian
health services, the difficulty in maintaining confidentiality in
small communities, the stigma attached to STDs, the unpleasant
nature of urethral and endocervical swabs and, in particular, the
frequent lack of male health care workers -- all militate against
effective STD programs.3-5 Urine tests for the detection of gonorrhoea and chlamydia are now commercially available. Enzyme immunoassay (EIA) tests have sensitivities above 80% and specificities above 93%,6-9 while polymerase chain reaction (PCR) tests have reported sensitivities of 90%-100% and specificities of 98%-100%.10-14 These technologies offered exciting possibilities to address some of the difficulties in delivering STD services, so, after receiving approval from the Alice Springs Institutional Ethics Committee, the Tri-State STD/HIV Project (TSP), in collaboration with Nganampa Health Council (NHC) and the Central Australian Aboriginal Congress (CAAC), both organisations controlled by Aboriginal communities, undertook the programs described in this article. |
Methods |
The study was undertaken in four parts.
Phase I: NHC provides health services to several remote Aboriginal communities and conducts annual syphilis serology screening programs in these communities. NHC maintains a population register, and all persons between the ages of 12 and 40 years who are in the communities during the screening programs are sought out and asked to participate. In April 1995, 189 men participating in this program in three communities serviced by NHC were asked to give a first-void urine sample in addition to the blood sample for the syphilis test. Phase II: In June 1995, another 218 men in the remaining three NHC communities had urine tests only; all males over the age of 12 who were present in the communities at the time were asked to participate. Phase III: In October 1995, a further 53 men were tested who had not been previously tested. Phase IV: CAAC, as part of its many functions, provides the health service to the Alice Springs Gaol. In June 1995, 33 new inmates were asked to give a first-void urine sample as part of their comprehensive health check on admission. Diagnostic tests used for gonorrhoea were culture, EIA (Abbott Gonozyme, Chicago, IL) and PCR (Roche Amplicor CT/NG, Branchburg, NJ) and for chlamydia were EIA (SYVA MicroTrak II, Palo Alto, CA) and PCR (Roche Amplicor, Branchburg, NJ) (Box 1, below). If there was insufficient urine for all tests, the order of priority was to do PCR first, then culture, chlamydia EIA and finally gonococcal EIA. To check for cross-contamination between PCR specimens, all Phase III specimens that provided sufficient urine were divided before any PCR procedures were performed. When a positive result was obtained the other half of the original sample was retested.
Chocolate and/or Thayer-Martin agar plates incubated in candle jars or CO2 gaspacks were used for gonococcal culture. BioCult GC tubes (Orion Diagnostica, Espoo, Finland), which contain a modified Thayer-Martin dipslide and a CO2-generating tablet, were also trialled. Culture media were inoculated with sediment from 10 mL of centrifuged urine and incubated for up to 72 hours at 35oC on-site and/or at Western Pathology in Alice Springs. Neisseria gonorrhoeae was identified by standard methods: typical morphology of colonies, oxidase paper test, microscopy with Gram stain, and latex agglutination (Phadebact GC, Boule Diagnostics AB, Huddinge, Sweden). Urine samples for EIA and PCR were prepared on-site according to the test manufacturer's specifications and then sent to Western Pathology in Alice Springs for processing. Male Aboriginal health workers and registered nurses in each clinic were responsible for liaison with the community and collection of specimens. Each man was asked to give 25-40 mL of first-void urine in a sterile collection jar. No urethral swabs were sought. Initial preparation of urine specimens for transport to the laboratory was completed within three hours of collection. A diagnosis of infection was made and treatment was initiated if any test was positive. Resident clinic staff were responsible for offering treatment, further investigation and safe-sex education to infected men and their sexual partners. |
Results | |
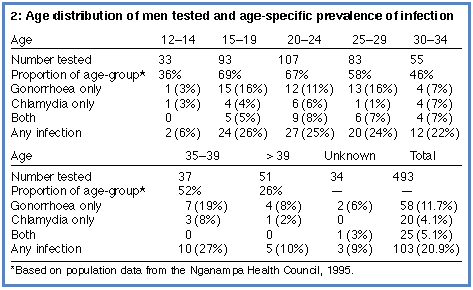
Study population | We tested urine samples from 493 men, of whom 460 lived in six remote communities and 33 were gaol inmates. Acceptability to clients was high, with fewer than 10 men (exact count not possible) declining testing. The 460 men from remote communities represented about 60% of the male population over the age of 12 in the area serviced by the participating clinics (Box 2, below). The proportion of men tested in different communities varied from 28% to 85%. |
The rate of infection | Based on all methods of diagnosis combined, 103 of the men tested (20.9%) were found to be infected with either gonorrhoea or chlamydia. The rate of infection in different communities varied between 14.5% and 26%. Among the gaol inmates, who originated from all parts of the southern NT, seven (21.2%) were infected. Most infections (90.3%) occurred in men aged 15-39 years and the five-year age-specific prevalence of infection in this group varied from 22% to 27% (Box 2, above). |
Treatment and follow-up | Eighty-eight men were treated within two months of testing. The average delay between testing and treatment was 18 days. Forty-five sexual partners of infected men were also recorded as having been treated. |
Performance of the tests used |
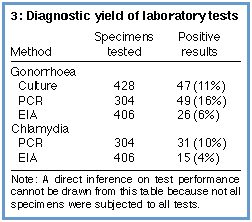
Different combinations of tests were used in different phases. The
volume of urine received from each man varied, so it was not always
possible to do all the intended tests. A summary of the numbers of
specimens subjected to the various tests and their relative
performance is shown in Box 3 (below).
During Phase II, culture for gonorrhoea using both BioCult GC tubes and standard Thayer-Martin plates was performed on 193 urine specimens. Thirteen diagnoses were made with the BioCult GC tubes and only seven were made with standard Thayer- Martin plates: all cases positive on Thayer-Martin plates were also positive on the BioCult GC tubes. During Phase III, 19 of the 20 initially positive PCR results were retested (in one case there was insufficient urine). In 18 of these cases there was complete agreement between the first and second test. One chlamydia-positive specimen was negative on the second testing. However, because of delays in transport, the tests on this specimen were done at six and eight days after collection, well in excess of the four days maximum recommended by the test manufacturers. |
Discussion |
These results highlight the need for improvements in current STD
control, including surveillance programs. Based on routine clinic
activity and notifications, NHC reported only 38 men with either
gonorrhoea or chlamydia during the whole of 1994 (health
departments' notifiable diseases data), compared with the 96 cases
detected in this study. Other regions of Australia that have
comparable notification rates of these infections1 may have similar problems in
underdetection.
The variation in participation rates in different communities and age-groups was largely attributable to the numbers of people who happened to be present at the time of the study (people in these remote communities being highly mobile), the working relationship between community members and the health staff, and the assiduousness of the health staff in conducting the program. In central Australia it is often considered, without specific evidence, that people who do not participate in such programs may be at higher risk of infection. Recent work by NHC showed no difference in rates of syphilis infection between those tested during the main body of a screening program and those tested later as part of an effort to test non-participants (Dr Penny Miller, unpublished data). The screening programs in our study were intended to identify the most effective and practical tests for everyday use in remote clinics. Collection of urethral swabs was avoided because it would not have been acceptable to the community. Hence, the study lacked a diagnostic "gold standard" and could not formally evaluate sensitivities and specificities. The published specificities of all the tests were high -- above 98% for urine PCR tests.10-14 The procedures used in Phase III demonstrated no problems with cross-contamination in PCR testing. Several studies have suggested that urine PCR tests for chlamydia are more sensitive than urethral-swab culture and highly specific.10-13 Less work has been done on urine PCR for diagnosing gonococcal infection, but two studies indicate sensitivities above 90% and specificities of 100%.13,14 In ideal conditions, the sensitivities of urethral-swab microscopy and culture are 90%-98%,15,16 but are likely to be much less in remote communities because of high staff turnover, the use of non-nutritive transport media, and frequently prolonged transport times. In such circumstances, urine PCR may be as good as or better than urethral-swab microscopy and culture for diagnosis of gonorrhoea. In any event, according to local management protocols,17 all men presenting to clinics with urethritis are offered immediate treatment for both gonorrhoea and chlamydia. In terms of practical application, PCR was superior to culture for gonorrhoea and EIA for both infections. PCR tests detected more infections with either organism than did the other tests (Box 3). Assuming that the high published specificities for the PCR tests held under field conditions, this increased rate of detection would be attributable to a superior sensitivity. The PCR test was also the easiest to use: urine is simply stored and transported at 4¡-8¡C in the same jar used to collect it. The urine must then be processed in the laboratory within four days (test manufacturer's instructions), which is usually feasible in most remote situations. In contrast, both EIA tests used require centrifuging before transport, and the SYVA MicroTrak II EIA required addition of a transport buffer. However, PCR tests do not provide information about antibiotic susceptibility. If PCR technology is to be used as the principal diagnostic tool for gonorrhoea, there would need to be accompanying sentinel systems for gonococcal culture and antibiotic susceptibility. We found that culture of first-void urine was positive in 72% of diagnoses of gonorrhoea. Sensitivities of 70%-100% for urine culture of gonorrhoea have been reported.18,19 On this basis, urine culture in addition to urine PCR for diagnosis of gonorrhoea could be performed routinely at sentinel sites and during similar programs to those reported here in order to maintain antibiotic-sensitivity surveillance. In the field situation, the BioCult GC tubes detected more gonorrhoea than standard Thayer-Martin plates and were easier and more convenient to use. The accuracy, ease of use and acceptability to men of urine PCR tests suggest several strategies to make clinical services more accessible to people, reduce the amount of disease in the community and identify individuals who are in need of safe-sex education. The use of urine tests for routine diagnosis may encourage more men to present with an STD, even if there is no male practitioner present. Health services could adopt active case-finding strategies such as opportunistic testing when people present for other reasons, including annual comprehensive health checks or community surveys. Such strategies are under consideration by health services in central Australia. For example, CAAC is seeking funding to establish an outreach program via a mobile clinic to make comprehensive well-men's check-ups, including STD checks, accessible to Aboriginal men in Alice Springs. We also examined screening as an STD control strategy. With fewer than 10 men refusing to participate, these programs resulted in 88 men who had either gonorrhoea or chlamydia and at least 45 of their sexual partners being treated. We did not have the resources to determine whether the men were symptomatic at the time of the test. However, none of them had presented to the clinic for treatment. When these findings are considered in the light of routine notifications and our own local experience, it is likely that most of these people would not have been diagnosed and treated outside these programs. Health services catering to remote Aboriginal communities in other parts of Australia may wish to consider this new technology and its usefulness in comprehensive STD education and control programs. |
Acknowledgements | The Tri-State STD/HIV Project (TSP) is jointly funded and managed by the Commonwealth Department of Health and Family Services, Territory Health Services, the South Australian Health Commission and the Western Australian Health Department. The work would not have been possible without the support of the clinical staff of Nganampa Health Council (NHC) and the Central Australian Aboriginal Congress (CAAC). Technical advice and material resources were contributed by the TSP, NHC, CAAC, Western Pathology, Alice Springs Hospital laboratory, the Australian Army, the Northern Territory AIDS/STD Unit, Roche Diagnostics, and the Institute of Medical and Veterinary Science. John Kaldor, Russell Waddell, Frank Bowden and John Boffa all commented on earlier drafts of this paper. |
References |
(Received 24 June, accepted 17 Oct, 1996) |
Authors' details
Tri-State STD/HIV Project, Alice Springs, NT.Steven J Skov, MPH, FAFPHM, Medical Officer, Tri-State STD/HIV Project.
Nganampa Health Council, Alice Springs, NT.
Penny Miller, MB BS, STD/HIV Program Coordinator.
Wayne Hateley, STD/HIV Aboriginal Health Worker.
Royal Darwin Hospital, NT.
Ivan B Bastian, MB BS, MSc, Microbiology Registrar.
Australian Army, Darwin, NT.
Jenny Davis, Medical Technician.
Central Australian Aboriginal Congress, Alice Springs, NT.
Peter W Tait, MB BS, DipRACOG, FRACGP, Acting Senior Doctor.
Reprints will not be available. Correspondence: Dr Penny Miller, Nganampa Health Council, PO Box 2232, Alice Springs, NT 0871.
- To top of article - ©MJA 1997
<URL: http://www.mja.com.au/> © 1997 Medical Journal of Australia.
Received 25 May 2026, accepted 25 May 2026
- Steven J Skov
- Penny Miller
- Wayne Hateley
- Ivan B Bastian
- Jenny Davis
- Peter W Tait