In pregnant women, most infections are no more serious than in non-pregnant women of similar age. However, some infections can be transmitted to the fetus in utero or to the infant during or immediately after delivery, with potentially serious sequelae (Box 1). Uncommonly, serious infectious illness in the mother can have non-specific fetal or obstetric effects and lead to miscarriage, premature labour or fetal death; these infections must be treated as any other serious illness. Much more common and a source of anxiety is mild illness or suggestive laboratory findings in the absence of symptoms. Investigation and management are often difficult and associated with potential ethical and medicolegal pitfalls.
Ideally, a woman and her partner should consult their general practitioner when planning pregnancy. Pre-pregnancy testing should include routine antenatal screening tests (Box 2), along with assessment of varicella immunity. Women negative for rubella or varicella IgG should be offered measles–mumps–rubella (MMR) or varicella vaccine, respectively. Women who receive MMR should be retested for rubella IgG seroconversion after two months and revaccinated if necessary. Rarely, seroconversion does not occur, even after two doses. As further doses are unlikely to be effective, these women should be advised that they are not immune; the risk of contact with rubella is small, but contact should be avoided if possible. Varicella vaccine is given in two doses two months apart, and pregnancy should be postponed for at least two months after the second dose.
Women in close contact with toddlers (eg, childcare workers) may be at increased risk of cytomegalovirus (CMV) infection during pregnancy. They should be tested for CMV IgG before conception. If seronegative, they should be counselled to reduce risk by handwashing after changing babies' nappies and avoiding contact with babies' saliva, and retested at monthly intervals during the first half of pregnancy.
A pre-pregnancy visit is also an opportunity to give dietary and other advice to reduce the risk of listeriosis4 and toxoplasmosis:
Avoid raw or undercooked fresh meat and refrigerated ready-to-eat food that is not freshly prepared (eg, cold meats, salads, soft cheeses and pâté).
Peel or wash raw fruit and vegetables thoroughly to remove contaminating soil.
Wash hands after disposing of cat litter or gardening (to remove soil).
This visit also allows counselling of both partners to avoid casual sexual contact and intravenous drug use and consequent risk of infection.
Screening should generally be applied to all pregnant women.5 Selective screening based on risk factors is unreliable — eliciting risk factors for all relevant infections is time-consuming and unlikely to identify all those at risk — and women are generally more willing to accept routinely offered tests than to acknowledge, if they are aware of it, being at high risk.6 Informed consent should be obtained for routine antenatal screening, which implies that women have the option of refusing.
Current recommendations for routine antenatal screening are shown in Box 2. Most recommendations are not controversial, but may vary between populations and over time because of changes in the epidemiology of infections and the availability and cost of screening tests and interventions. Tests not recommended for routine antenatal screening in Australia are shown in Box 3.
If a pregnant woman is exposed to an infection known to be transmissible to the fetus or infant, she should be investigated as soon as possible to determine her susceptibility. If she is susceptible, then determining the basis for diagnosis in the contact (eg, whether laboratory-confirmed) and type of contact (casual and transient, or, for example, with her own child) may help in assessing risk of infection. Serological testing should be repeated up to three weeks after contact to detect seroconversion. Immunoprophylaxis is available for susceptible pregnant women after contact with varicella (see below). Pregnant women who have sexual contact with a man with a sexually transmissible infection should be tested for other sexually transmissible infections, even if routine antenatal screening has already been done.
Symptoms of infection in pregnant women should be investigated unless the cause is obvious. Investigations depend on presentation (Box 4). Diagnosis may be based on clinical symptoms and confirmatory laboratory results (IgG seroconversion, or significant increase in level, IgM or detection of the pathogen), a history of compatible clinical symptoms only (if too late for serological confirmation), or serological evidence only.
The best serological evidence of recent systemic infection is IgG seroconversion (change from negative to positive result for specific IgG) or a significant increase in IgG level when measured quantitatively. The first serum specimen should be collected as soon as possible after contact or symptom onset. If routine antenatal screening has been done, the laboratory should have stored serum that can be tested in parallel (see case report, Box 5). A negative specific IgG result early in the illness does not exclude recent infection. Testing should be repeated up to three weeks after contact.
Specific IgM (or sometimes IgA) may be present without IgG early in the infection but this result must be interpreted with caution. If IgG seroconversion does not occur, the IgM result is likely to be a false positive. If both IgG and IgM are present and their levels remain unchanged in sequential specimens, IgM testing should be repeated using a different method. For rubella IgM, the result should be confirmed by testing of IgM fractions after sucrose density centrifugation of the serum. If CMV or toxoplasma IgM remains positive, an IgG avidity assay will help confirm or exclude recent infection. IgG avidity measures how tightly antibody binds to antigen and increases with time after initial infection; high avidity indicates that infection occurred at least several months previously, although avidity may remain low for many months. Interpretation depends on individual test methods and should be discussed with a clinical microbiologist, as should any other doubts about diagnosis.
In general, IgM tests should not be performed in the absence of a suggestive clinical illness or contact, as the positive predictive value can be relatively low. Infection should not be diagnosed on the basis of a single positive IgM result (case report, Box 6).
If a vertically transmissible infection is confirmed or cannot be excluded in a pregnant woman, the risk to the fetus depends on the stage of pregnancy and the type of infection. For some infections, it may be appropriate to determine whether the fetus has been infected (eg, CMV infection, toxoplasmosis and, in some circumstances, varicella). This requires specialised tests, such as culture or nucleic acid testing of amniotic fluid. Expert advice and appropriate investigations are essential before interventions such as termination of pregnancy or administration of potentially toxic drugs are considered.
Management of selected vertically transmissible infections is summarised in Box 4.
Although rubella is generally preventable by vaccination, congenital rubella still occurs in Australia.11 Up to 10% of women of child-bearing age are susceptible, as they have not been vaccinated, vaccination has failed, or vaccine-induced immunity has waned. Although the Australian Measles Control Campaign raised the proportion of school children immune to rubella to over 95% in 1998, 15%–20% of young men remain susceptible and a potential source of infection for pregnant women with inadequate immunity.
If infection occurs in the first trimester of pregnancy, the risk of fetal infection and damage is high (about 90% in the first two months of pregnancy, and 50% in the third), and termination of pregnancy is usually recommended. Risk of fetal damage falls steeply after the first trimester and is negligible after 16 weeks; between 12 and 16 weeks, deafness has been reported.
If a pregnant woman has close contact with or develops rubella-like illness, her IgG and IgM titres should be measured, even if she was previously positive for rubella IgG — rarely, women with apparently adequate immunity can be reinfected (although the risk of fetal abnormality is probably less than 5%, even in the first trimester12). If contact is in the second or third trimester and rubella IgG was detected in the first trimester, further investigation is not necessary. Women who remain susceptible to rubella should receive MMR vaccine post partum, unless two previous attempts at immunisation have failed.
Cytomegalovirus is the most common cause of congenital infection and non-hereditary deafness. Currently, there is no vaccine or treatment that can be given during pregnancy (ganciclovir is used to treat serious CMV infection associated with conditions such as organ transplantation or HIV/AIDS). CMV infection is transmitted by contact with saliva, urine or genital secretions and often causes mild hepatitis, atypical lymphocytosis and non-specific symptoms during the self-limiting primary infection. The virus then becomes latent, but is reactivated periodically during episodes of mild immunosuppression caused by intercurrent infection, pregnancy or stress. Reactivation is asymptomatic, except in severely immunocompromised individuals.
Primary maternal infection occurs in about 1 per 300 pregnancies in Australia and results in fetal infection in about 40% of cases (affecting 1 per 1000 infants); fetal damage is most likely early in pregnancy. Most congenitally infected infants are apparently normal at birth, but long-term sequelae, most commonly deafness and mild intellectual impairment, occur in up to 40% (10%–15% of all infants of women with primary infection during pregnancy). Symptomatic multisystem disease, characterised by growth retardation, microcephaly, intracranial calcification, thrombocytopenia and hepatitis, is uncommon.13
Reactivation of maternal infection during pregnancy can also cause fetal or perinatal infection (about 1% of infants), but sequelae are uncommon and usually mild.14 It is usually difficult to distinguish asymptomatic primary maternal infection from reactivation or to determine precisely when infection occurred.
If primary maternal CMV infection is suspected because of close contact or a compatible illness, serological and liver function tests and a blood film will usually confirm the diagnosis (case report, Box 5). More often, primary CMV infection is suspected because of a positive CMV IgM result in routine antenatal screening. False positive CMV IgM results are common, because of cross-reactions, viral reactivation or persistent low-level IgM after past primary infection. IgG avidity testing will help distinguish recent from long-past infection.15 Ideally, women who believe they are at risk of CMV infection, such as childcare workers, should be tested for IgG before conceiving.
If recent CMV infection is likely or cannot be excluded, especially in the first trimester, amniocentesis should be considered to determine whether the fetus is infected. It should be done at about 19 weeks' gestation, or at least six weeks after the likely time of infection. CMV isolation or positive nucleic acid test results from amniotic fluid indicate fetal infection, but not necessarily morbidity, while negative results indicate that severe fetal morbidity is extremely unlikely. If fetal infection is confirmed, the stage of pregnancy at which it occurred, viral load in the amniotic fluid and evidence of fetal abnormality or growth retardation on ultrasound examination may aid in considering termination of pregnancy.15
Like CMV infection, toxoplasmosis is usually asymptomatic or has mild, non-specific symptoms. Primary infection during pregnancy can cause serious fetal effects. However, unlike CMV infection, toxoplasmosis during pregnancy can be treated, potentially reducing the fetal effects.
Although asymptomatic women with perceived risk (eg, contact with cats) are often tested for toxoplasma IgG, pre-pregnancy or antenatal screening is not recommended. There is no clearly defined group of women at increased risk, about 75% of women are susceptible,16 and seroconversion during pregnancy is uncommon. False positive toxoplasma IgM results are not uncommon (case report, Box 6), and low levels of IgM may persist for many months or years after primary infection. Like CMV, toxoplasma infection remains latent for life, but clinical reactivation is confined to severely immunosuppressed individuals. Infants of women who are seropositive before conception are not at risk.
Toxoplasmosis is acquired through eating raw or undercooked meat or ingesting soil contaminated with toxoplasma oocysts, which are excreted in the faeces of infected cats. Pregnant women should be specifically advised to avoid these exposures (see Pre-pregnancy testing and counselling). Direct contact with cats is rarely a source of infection (they are usually infected as kittens and excrete oocysts for a relatively short time).
Investigation of suspected acute toxoplasmosis is similar to that of suspected CMV infection. If maternal infection is confirmed or cannot be excluded, antibiotic treatment appears to reduce the risk of fetal infection and sequelae,8 although this has not been confirmed by randomised controlled trials.17 For maternal infection in the first trimester, it is particularly important to determine whether the fetus is infected,18 as likelihood of fetal infection is low (about 15%), but, if it occurs, fetal damage is likely to be severe. Later in pregnancy, infection is more likely, but fetal damage is less likely and, if it occurs, less severe.19
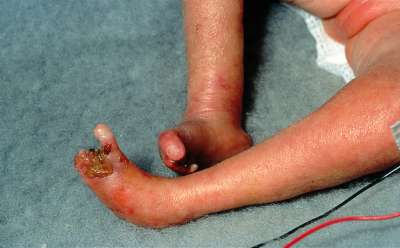
Fetal infection occurs in 10%–15% of cases of varicella (chickenpox) in pregnant women but is usually transient and asymptomatic. The most common clinical manifestation, if any occurs, is shingles in the first year of life. However, 2%–3% of infants of women who have chickenpox in the first half of pregnancy develop fetal varicella syndrome, with potentially severe defects, including skin scarring in a dermatomal distribution, ipsilateral limb hypoplasia, visceral, neurological and eye lesions20 (Box 7). Maternal varicella within a few days before or after delivery can result in potentially severe varicella in the infant,21 who should be given zoster immune globulin (ZIG) as soon as possible after birth.
More than 90% of women of child-bearing age in Australia are immune to varicella virus,9,16 with a history of infection providing reliable evidence of immunity. If in doubt when contact occurs, pregnant women should be tested for varicella IgG as soon as possible (including those who have been vaccinated, if seroconversion has not been confirmed). If seronegative, they should be offered ZIG, preferably within 48 hours of contact (maximum, 72 hours). ZIG may not prevent infection but reduces illness severity (case report, Box 8). It is not effective after rash onset.9
The diagnosis of chickenpox is usually obvious. The disease is more likely to be severe in adults than in children and may be complicated by pneumonia, especially in smokers and in the latter half of pregnancy, and is occasionally fatal. Use of aciclovir is not recommended during pregnancy, but evidence is accumulating that it has no adverse fetal effects.22 Given during the incubation period or within 24 hours of rash onset, it can reduce risk of infection or illness duration and severity. Its use should be considered during the incubation period for women who have not received ZIG, or soon after rash onset, especially in women with risk factors for severe disease, such as chronic lung disease, smoking or impaired immunity, or in the latter half of pregnancy.9 If disease progresses, admission to hospital and intravenous aciclovir are indicated.
The recommended use of varicella vaccine in susceptible women of child-bearing age will reduce the incidence of congenital and neonatal varicella in Australia.2
Parvovirus B19 causes erythema infectiosum, or fifth disease, which occurs in epidemic waves lasting two to three years, mainly among primary-school-aged children. Infection is often asymptomatic, but, like rubella, can cause rash and arthralgia or arthritis, particularly in adults. Complications of parvovirus infection during pregnancy are excess fetal loss in the first 20 weeks (up to 10% excess) and hydrops (about 3%) if maternal infection occurs between nine and 20 weeks' gestation23 (Box 9). Hydrops is caused by severe fetal anaemia and presents about five weeks after maternal infection. About a third of cases resolve spontaneously, and outcome is significantly improved in the remainder by intrauterine transfusion.24
About 60% of Australian women of child-bearing age are susceptible to parvovirus B19,25 and the annual seroconversion rate varies from about 1%–2% during non-epidemic years to 10%–15% during epidemics.26 Household and occupational contact (eg, in a schoolteacher) lead to seroconversion in about 50% and 20%–30%, respectively, of those susceptible.7
Routine antenatal screening for parvovirus antibody is not indicated, nor is it generally recommended that susceptible pregnant women with occupational exposure to the virus stay away from work. Epidemics are protracted, infection is prevalent in the community, and infectivity precedes rash onset. If contact occurs during pregnancy, IgG tests should be done to determine susceptibility and, if seronegative, repeated two to three weeks later.
If infection is confirmed during pregnancy, the fetus should be monitored for signs of hydrops by ultrasound examination over the next six to 12 weeks, with appropriate specialist referral if it occurs. PCR examination of amniotic fluid is not recommended after proven maternal parvovirus infection, but it can be helpful during investigation of non-immune hydrops of unknown cause.
Many different enteroviruses circulate in the community, especially during summer. They cause a variety of syndromes, mainly in children, including non-specific febrile illness; maculopapular, petechial or vesicular rash (hand, foot and mouth disease); upper respiratory tract infection; and aseptic meningitis. Infection is usually benign, but can be transmitted to the infant if it occurs late in pregnancy and, rarely, can cause life-threatening meningoencephalitis, cardiomyositis or hepatitis.27 No specific intervention is available for pregnant women, but a history of relevant symptoms may aid diagnosis of severe neonatal infection.
Many other maternal infections can be transmitted to the fetus or infant.
Listeriosis is an uncommon foodborne illness caused by Listeria monocytogenes. Pregnant women are particularly susceptible to the disease, which can result in fetal death or chronic intrauterine and congenital or perinatal infection.4,28 Investigation of symptoms such as headache, myalgia and fever associated with relatively inconspicuous gastrointestinal symptoms in a pregnant woman should include a recent dietary history and blood culture (Box 4). It is treatable with penicillin.
Gonorrhoea and chlamydial infection can be transmitted to the infant during delivery and initially cause superficial infection (conjunctivitis) or upper respiratory tract colonisation. Gonococcal infection may become disseminated, while chlamydial infection may cause pneumonia at four to six weeks of age.
Herpes simplex virus (HSV) causes neonatal HSV sepsis syndrome and encephalitis, which are often fatal or produce long-term sequelae, but are rare in Australia.29 The risk of vertical transmission of HSV (types 1 and 2) is greatest during primary maternal infection, which is often associated with viraemia. Primary genital herpes (usually type 2) may be clinically severe, with heavy and prolonged viral excretion, and higher risk of transmission to the infant during vaginal delivery than recurrent disease. Treatment with aciclovir and, if lesions are present at term, caesarean section should be considered in primary genital HSV infection. Caesarean section is not routinely recommended in women with recurrent genital herpes, as the risk of vertical transmission is small. It should be considered only if active genital lesions that cannot be covered are present at the onset of labour.30
Some vertically transmissible infections can be prevented or treated if detected by routine antenatal screening. Appropriate investigation and management of other suspected infections can reduce unnecessary intervention, prevent or moderate adverse outcomes and relieve anxiety. However, antenatal screening that is not based on accepted criteria or a well-defined plan of action can cause unnecessary anxiety and potentially dangerous intervention.
Evidence-based recommendations
Women planning pregnancy or already pregnant should be tested routinely for chronic HBV infection5 (E1), syphilis5 (E3), susceptibility to rubella5 and asymptomatic bacteriuria31 (E1, E2) and managed according to established protocols (E1–E3).
HIV antibody testing should be offered to all pregnant women, and any who are seropositive should be offered antiretroviral therapy1 (E2).
Non-pregnant women of childbearing age should be tested for immunity to varicella and offered vaccine if non-immune9 (E2).
Suspected infection or contact with infections known to be vertically transmissible in early pregnancy should be investigated. Intervention should be based on laboratory-confirmed maternal and, if appropriate, fetal infection and consideration of known risks of fetal damage (E3–E4, depending on infection).15,18
Aciclovir treatment of varicella and herpes simplex virus during pregnancy can significantly reduce morbidity in the mother and potentially in the infant9 (E2). Aciclovir is not recommended in pregnancy, although there is no evidence of adverse fetal effects22 (E3). Its use should be considered when it is judged that benefits outweigh risks.
1: Vertically transmissible* pathogens and usual route of transmission
Usual route of transmission |
|||||||||||||||
Pathogen (disease) |
Intrauterine† |
Perinatal‡ |
Postnatal§ |
||||||||||||
Rubella virus |
++ |
– |
+ |
||||||||||||
Treponema pallidum (syphilis) |
++ |
– |
– |
||||||||||||
Toxoplasma gondii |
++ |
– |
– |
||||||||||||
Cytomegalovirus |
++ |
++ (G,H) |
+ |
||||||||||||
Parvovirus |
++ |
– |
– |
||||||||||||
Varicella zoster virus (chickenpox) |
+ |
++ (H) |
– |
||||||||||||
Human immunodeficiency virus |
± |
++ (H) |
+ |
||||||||||||
Hepatitis B virus |
± |
++ (H) |
+ |
||||||||||||
Hepatitis C virus |
± |
++ (H) |
– |
||||||||||||
Herpes simplex virus |
± |
++ (G,H) |
– |
||||||||||||
Chlamydia trachomatis |
– |
++ (G) |
– |
||||||||||||
Neisseria gonorrhoeae |
– |
++ (G) |
– |
||||||||||||
Listeria monocytogenes |
++ |
+ (G,H) |
– |
||||||||||||
Group B streptococci |
+/– |
++ (G,H) |
– |
||||||||||||
++ = main route of transmission. |
|||||||||||||||
2: Recommended routine antenatal screening tests in Australia
Test |
Comment |
Intervention |
|||||||||||||
Rubella IgG |
Continue screening at each pregnancy |
If negative, give measles–mumps–rubella vaccine before conception or post partum. |
|||||||||||||
Hepatitis B surface antigen |
To detect chronic carriers |
If positive, administer hepatitis B immune globulin and vaccine to infant at birth (prevents carriage in 95%).* |
|||||||||||||
Syphilis |
To detect active infection |
If reactive, indicating active syphilis, treat with penicillin and consult a specialist in treatment of sexually transmissible diseases. |
|||||||||||||
HIV antibody |
Should be offered to all pregnant women1† |
If positive, antiretroviral therapy for mother and infant significantly reduces vertical transmission; consult a specialist in treatment of HIV infection. |
|||||||||||||
Varicella IgG |
Not routine; should be offered to women of child-bearing age |
If susceptible, offer vaccine post partum.2 |
|||||||||||||
Urine culture |
Treatment will reduce adverse pregnancy outcome |
If culture shows bacteria > 108/L and a single species (asymptomatic bacteriuria), treat with antibiotics, repeat culture, and treat again if still culture-positive. |
|||||||||||||
Vaginal/rectal swabs for group B streptococcal carriage |
Controversial (other criteria for intrapartum antibiotic prophylaxis include clinical risk factors during labour)3‡ |
Intrapartum antibiotic prophylaxis for carriers. |
|||||||||||||
* Hepatitis B vaccination starting at birth is now standard for all infants in Australia. |
|||||||||||||||
3: Tests not recommended for antenatal screening in Australia
Cytomegalovirus IgG and IgM
No treatment or vaccine available for seronegative women
IgM is unreliable marker of recent infection
Toxoplasma IgG and IgM
Primary infection during pregnancy is rare in Australia
IgM is unreliable marker of recent infection
Routine screening is recommended in some countries
Parvovirus IgG
Risk of maternal and fetal infection and damage is low
No treatment or vaccine available7
Hepatitis C virus antibody
Infection is rare, except in well-defined high-risk groups (mainly intravenous drug users), and transmission to fetus is uncommon
No vaccine or treatment is available to prevent transmission
Urine test for chlamydial nucleic acid
Incidence is generally low in Australia
Routine antenatal screening may be cost-effective in some population groups with relatively high incidence
4: Investigation and management of symptomatic infective illness during pregnancy
Presentation |
Possible diagnoses |
Tests |
Treatment |
||||||||||||
Influenza- or glandular fever-like illness (lethargy, fever, malaise, myalgia, ± headache, ± lymphadenopathy) |
Primary CMV infection (often also causes mild hepatitis, lymphocytosis) |
IgG and IgM (paired sera) |
Nil, termination of pregnancy if high risk of fetal morbidity |
||||||||||||
Primary toxoplasmosis (lymphadenopathy often prominent) |
IgG and IgM (paired sera) |
Spiramycin and/or pyrimethamine–sulfadoxine8 |
|||||||||||||
Listeriosis (foodborne, may be associated with diarrhoea) |
History, culture of faeces ± blood or urine |
Penicillin |
|||||||||||||
Many other viral infections |
Culture, serology if appropriate |
||||||||||||||
Maculopapular rash ± Fever ± Arthritis/arthralgia |
Rubella |
IgG and IgM (paired sera) |
Nil, termination of pregnancy if high risk of fetal morbidity |
||||||||||||
Parvovirus infection |
IgG and IgM (paired sera) |
Nil, monitor by ultrasound for hydrops7 |
|||||||||||||
Enterovirus infection |
Throat swab or faecal culture |
Nil |
|||||||||||||
Vesicular rash |
Varicella |
Rash is usually typical, allowing clinical diagnosis; if in doubt, lesion swab and serology |
Oral or intravenous aciclovir9 |
||||||||||||
Hand, foot and mouth disease (enterovirus infection) |
Throat swab or faecal culture |
Nil |
|||||||||||||
Genitourinary symptoms (frequency, dysuria) ± Loin pain ± Fever ± Genital ulcer ± Vaginal discharge |
Urinary tract infection (cystitis; pyelonephritis) |
Urine microscopy and culture |
|||||||||||||
Chlamydia, gonorrhoea |
Cervical swab, Gram stain and culture; urine PCR (if positive, check syphilis, HBV and HIV serology) |
According to Antibiotic guidelines10 |
|||||||||||||
Genital herpes |
First episode: lesion swab, HSV1 and HSV2 IgG and IgM (paired with stored serum if available) |
Aciclovir |
|||||||||||||
Maternal fever near term ± Preterm labour ± Premature rupture of membranes |
Urinary tract infection |
Urine microscopy, culture |
According to Antibiotic guidelines10 |
||||||||||||
Chorioamnionitis (eg, due to group B streptococci or abnormal vaginal flora associated with bacterial vaginosis) |
Vaginal Gram stain and culture (for group B streptococci) |
Amoxycillin, metronidazole, gentamicin |
|||||||||||||
CMV = cytomegalovirus. PCR = polymerase chain reaction. HSV = herpes simplex virus. HBV = hepatitis B virus. |
|||||||||||||||
5: Case report — cytomegalovirus infection in pregnancy
Presentation: A 35-year-old woman presented to her general practitioner at 12 weeks' gestation. About three weeks earlier, her husband had an illness characterised by fever, headache and diarrhoea, which was diagnosed serologically by his local doctor as cytomegalovirus (CMV) infection. The woman was well but concerned about her baby. Ultrasound examination at 11 weeks' gestation had found no abnormalities.
Investigations: Serological testing of the woman's serum revealed both IgG and IgM to CMV. She was seen two weeks later (14 weeks' gestation) by an infectious diseases specialist, and the test was repeated, with parallel testing of serum stored by the testing laboratory after routine antenatal testing at eight weeks' gestation. Parallel testing showed CMV IgG seroconversion, confirming the diagnosis of recent asymptomatic CMV infection, probably at 11–12 weeks' gestation. Ultrasound examination was repeated at 14 weeks' gestation and showed no abnormalities and normal fetal growth since the first ultrasound examination.
Management: After discussing the risks of fetal infection (about 40%) and damage (about 40% if the fetus was infected, or 15%–20% overall), amniocentesis was offered. This was declined, as the patient and her husband decided not to consider terminating the pregnancy even if culture and polymerase chain reaction of amniotic fluid were positive for CMV.
Outcome: A healthy boy was born at term. Culture of his urine collected soon after birth was negative for CMV, confirming that he had not been infected in utero.
Febrile illness in a pregnant woman or her close contacts should be investigated.
Laboratories usually store serum samples from pregnant women for at least 12 months. These can be useful in the investigation of other infections later in pregnancy.
Proven CMV infection in the first trimester of pregnancy should not automatically be an indication for termination. Detailed discussion of risks and options and possible further investigation is essential.
6: Case report — investigation of possible toxoplasmosis in pregnancy
Presentation: A 27-year-old primigravida presented to her general practitioner at about 12 weeks' gestation. She had recently returned to Australia after living in the Philippines, where routine antenatal screening had shown toxoplasma IgM but not IgG. She had been well, apart from mild morning sickness early in her pregnancy, and, in particular, had had no fever or lymphadenopathy. She was vegetarian and opposed to termination of pregnancy on religious grounds.
Investigations: Toxoplasma antibody testing was repeated and was positive for toxoplasma IgG and IgM, indicating that she had seroconverted.
Management: She was referred to an infectious diseases specialist, who ordered another test to confirm the IgM result and measure IgG avidity. As the serum tested in Manila was not available, it was not possible to confirm seroconversion by testing sera in parallel. After discussion, the couple opted for immediate treatment with spiramycin (which requires authorisation to import, at considerable expense). Amniocentesis was planned at about 20 weeks' gestation to determine whether the fetus was infected. Although the patient had ruled out termination whatever the result of amniocentesis, antibiotic treatment could stop if it showed the fetus was not infected.
Results: Serology results were available the next day and showed neither toxoplasma IgG nor IgM. The first serum tested on the patient's return to Australia was retrieved from another laboratory and was also negative for both toxoplasma IgG and IgM when tested by a different method.
Outcome: The pregnancy continued uneventfully.
Seroconversion should be confirmed as far as possible by testing sera in parallel.
False-positive toxoplasma IgM results are not uncommon, and testing should always be repeated using an alternative method.
False-positive toxoplasma IgG results are much less common, but significant intervention should never be recommended on the basis of a single serological test result, no matter how reliable the laboratory.
8: Case report — varicella in pregnancy
Presentation: A 23-year-old woman who was 15 weeks' pregnant presented with her two-year-old daughter, who had developed a vesicular rash the previous day. The rash appeared typical of early chickenpox. The woman had no past history of chickenpox, and her mother could not recall her having been infected.
Investigation and management: Serological testing the next day was negative for varicella IgG, and zoster immune globulin (ZIG) was given, although it was at least 72 hours since likely initial contact.
Clinical course: Two weeks later (at 17 weeks' gestation), the woman developed a sparse varicelliform rash and minimal systemic symptoms, which resolved rapidly. Routine ultrasound examinations at this time and four weeks later found no fetal abnormalities. The possibility of amniocentesis and polymerase chain reaction (PCR) testing for varicella virus was discussed. A negative PCR result has a high negative predictive value, while a positive test indicates fetal infection but not necessarily morbidity. The woman and her husband decided that in the absence of any abnormality on ultrasound examination they would not consider terminating the pregnancy, even if the PCR result indicated fetal infection.
Outcome: A normal infant was delivered at term. He progressed normally, but at nine months of age developed a vesicular rash in the distribution of dermatome T12. This resolved uneventfully and did not recur.
Women who have close contact with chickenpox during pregnancy and no previous history of the disease should be tested as soon as possible for varicella antibody (most are seropositive, even in the absence of a past history).
ZIG should be given as soon as possible after contact. Although its effectiveness diminishes if given more than 72 hours after contact, it may modify the severity of the rash, if it occurs. However, the risk of fetal infection is unrelated to the severity of maternal illness.
The most common clinical manifestation of fetal varicella infection is herpes zoster in the first year of life, which is usually benign.
The role of intrauterine diagnosis of fetal varicella infection by amniocentesis and PCR of amniotic fluid is not yet established.
9: Estimated outcomes of occupational exposure to parvovirus B19 during an epidemic*
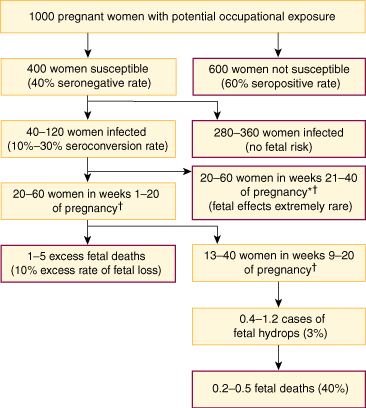
* Based on data from prospective studies.7
† On the assumption that, among women infected during a 40-week pregnancy, half will be infected during any 20-week period, and about a third during any 12-week period (eg, weeks 9–20).
- Gwendolyn L Gilbert1
- Series Editors:
- Institute of Clinical Pathology and Medical Research, Westmead Hospital, Sydney, NSW.
- 1. Ziegler JB. Antenatal screening for HIV in Australia: time to revise policies? Med J Aust 1999; 171: 201-203.
- 2. Smith WJ, Jackson LA, Watts DH, Koepsell TD. Prevention of chickenpox in reproductive-age women: cost-effectiveness of routine prenatal screening with postpartum vaccination of susceptibles. Obstet Gynecol 1998; 92: 535-545.
- 3. Gilbert GL, Isaacs D, Burgess MA, et al. Prevention of neonatal group B streptococcal sepsis: is routine antenatal screening appropriate. Aust N Z J Obstet Gynaecol 1995; 35: 120-126.
- 4. Australia New Zealand Food Authority. Listeria and pregnancy. <http://www.foodstandards.gov.au/mediareleasespublications/factsheets/factsheets1999/listeriaandpregnancy.cfm> Sighted August 2005.
- 5. Gilbert GL. Routine antenatal screening and prenatal diagnosis of vertically transmissible infection. Baillieres Clin Obstet Gynaecol 1993; 7: 1-23.
- 6. Carusi D, Learman LA, Posner SF. Human immunodeficiency virus test refusal in pregnancy: a challenge to voluntary testing. Obstet Gynecol 1998; 91: 540-545.
- 7. Gilbert GL. Parvovirus B19 and its significance in pregnancy. Commun Dis Intell 2000; 24: 69-71.
- 8. Foulon W, Villena I, Stray-Pedersen B, et al. Treatment of toxoplasmosis during pregnancy: a multicenter study of impact on fetal transmission and children's sequelae at age 1 year. Am J Obstet Gynecol 1999; 180: 410-415.
- 9. Heuchan AM, Isaacs D. The management of varicella zoster virus exposure and infection in pregnancy and the newborn period. Med J Aust 2001; 174: 288-292.
- 10. Therapeutic Guidelines Limited. Therapeutic guidelines: antibiotic. Version 11, 2000. Melbourne: Therapeutic Guidelines Limited, 2000.
- 11. Sullivan EV, Burgess MA, Forrest JM. The epidemiology of rubella and congenital rubella in Australia, 1992 to 1997. 23: 209-14. Commun Dis Intell 1999; 23: 209-214.
- 12. Best JM, Banatvala JE, Morgan-Capner P, Miller E. Fetal infection after maternal reinfection with rubella: criteria for defining reinfection. BMJ 1989; 299: 773-775.
- 13. Gilbert GL. Congenital (fetal) infections. Semin Neonatol 1996; 1: 91-119.
- 14. Rousseau T, Douvier S, Reynaud I, et al. Severe fetal cytomegalic inclusion disease after documented maternal reactivation of cytomegalovirus infection during pregnancy. Prenat Diagn 2000; 20: 333-336.
- 15. Lazzarotto T, Varani S, Guerra B, et al. Prenatal indicators of congenital cytomegalovirus infection. J Pediatrics 2000; 137: 90-95.
- 16. Karunajeewa H, Siebert D, Hammond R, et al. Seroprevlaence of varicella zoster virus, parvovirus B19 and Toxoplasma gondii in a Melbourne obstetric population: implications for management. Aust N Z J Obstet Gynaecol 2001; 41: 23-28.
- 17. Wallon M, Liou C, Garner P, Peyron F. Congenital toxoplasmosis: systematic review of evidence of efficacy of treatment in pregnancy. BMJ 1999; 318: 1511-1514.
- 18. Foulon W, Pinon JM, Stray-Pedersen B, et al. Prenatal diagnosis of congenital toxoplasmosis: a multicenter evaluation of different diagnostic parameters. Am J Obstet Gynecol 1999; 181: 843-847.
- 19. Dunn D, Wallon M, Peyron F, et al. Mother-to-child transmission of toxoplasmosis: risk estimates for clinical counselling. Lancet 1999; 353: 1829-1833.
- 20. Enders G, Miller E, Cradock-Watson J, et al. Consequences of varicella and herpes zoster in pregnancy: prospective study of 1739 cases. Lancet 1994; 343: 1548-1551.
- 21. Forrest JM, Mego S, Burgess MA. Congenital and neonatal varicella in Australia. J Paediatr Child Health 2000; 36: 108-113.
- 22. Reiff-Eldridge R, Heffner CR, Ephross SA, et al. Monitoring pregnancy outcomes after prenatal drug exposure through prospective pregnancy registries: a pharmaceutical company commitment. Am J Obstet Gynecol 2000; 182: 159-163.
- 23. Miller E, Fairley CK, Cohen BJ, Seng C. Immediate and long term outcome of human parvovirus B19 infection in pregnancy. Br J Obstet Gynaecol 1998; 105: 174-178.
- 24. Fairley CK, Smoleniec JS, Caul OE, Miller E. Observational study of effect of intrauterine transfusions on outcome of fetal hydrops after parvovirus B19 infection. Lancet 1995; 346: 1335-1337.
- 25. Kelly HA, Siebert D, Hammond R, et al. The age-specific prevalence of human parvovirus immunity in Victoria, Australia compared with other parts of the world. Epidemiol Infect 2000; 124: 449-457.
- 26. Valeur-Jensen AK, Pedersen CB, Westergaard T, et al. Risk factors for parvovirus B19 infection in pregnancy. JAMA 1999; 281: 1099-1105.
- 27. Daley AJ, Isaacs D, Dwyer DE, Gilbert GL. A cluster of cases of neonatal coxsackievirus B meningitis and myocarditis. J Paediatr Child Health 1998; 34: 196-198.
- 28. Silver H. Listeriosis during pregnancy. Obstet Gynecol Survey 1998; 53: 737-740.
- 29. Mindel A, Taylor J, Tideman RL, et al. Neonatal herpes prevention: a minor public health problem in some communities. Sex Transm Dis 2000; 76: 287-291.
- 30. M arks C, Mindel A, Fethers K. Management of women with recurrent genital herpes in Australia. Sex Transm Dis 1999; 75: 55-57.
- 31. Smaill F. Antibiotics for asymptomatic bacteriuria in pregnancy. Cochrane Database of Systematic Reviews 2000; CD000490.





Abstract
Some infections are more serious in pregnant than non-pregnant women because of the potential for vertical transmission to the fetus or infant (eg, varicella, rubella, cytomegalovirus infection, toxoplasmosis and listeriosis).
Pre-pregnancy or routine antenatal screening for presence of, or susceptibility to, some of these infections and appropriate management can prevent adverse fetal or perinatal outcomes; screening should include rubella IgG, hepatitis B surface antigen, serological tests for syphilis and HIV antibody.
If certain other vertically transmissible infections are suspected because of a positive antenatal test result, confirmatory tests for maternal and, if indicated, fetal infection are essential before intervention is considered (eg, cytomegalovirus infection).
For some vertically transmissible infections that are not readily preventable, appropriate management of maternal infection can reduce fetal damage (eg, toxoplasmosis).