On 2 November 2023, Cancer Australia unveiled the Australian Cancer Plan, emphasising a strategic commitment to “maximising cancer prevention and early detection”.1,2 This initiative holds particular significance for general practice, as most cancer diagnoses originate from symptomatic presentations to primary care, even when screening programs are available.3 For context, full‐time general practitioners on average diagnose up to 12 non‐cutaneous cancers annually, but they see patients consulting with symptoms associated with cancer almost daily.3,4
By its very nature, general practice is characterised by breadth and whole‐person patient‐centred care. Patients present with undifferentiated symptoms unconfined to a specific population group, organ or system in a setting where the individual likelihood of cancer is low. As a result, the pre‐test probability of a particular symptom being attributable to cancer is substantially lower than in secondary care settings, which are by default a selected subset of patients.5 The apparent strong associations between a single cancer type and specific symptoms seen in secondary care are diluted when applied to the low prevalence context of general practice. For example, although around half of patients diagnosed with pancreatic cancer initially exhibit abdominal pain,6 the risk of isolated abdominal pain being attributable to pancreatic cancer in the primary care population is well below 1%.7
Identifying patients with only red‐flag symptoms also fails to account for about half of patients diagnosed with cancer after presenting to general practice with non‐red‐flag symptoms.8 Significantly, non‐specific symptoms are associated with delays in diagnosis but also correlate with multiple types of cancer and various non‐cancerous conditions, making them challenging to address within tumour‐specific guidelines.9 For instance, symptoms such as change in bowel habit are associated with four different malignancies in women, while abdominal discomfort may be linked to as many as eight types of cancer in men and nine in women.10 Finally, contemporary general practice is becoming increasingly complex, where multimorbidity is the norm rather than the exception. Comorbid conditions complicate presentations and function as competing demands that can have an impact on the timeliness of cancer diagnosis.11
Efforts to improve early cancer diagnosis also occur alongside a push to reduce overtesting, overdiagnosis and unwarranted variation. Excessive use of diagnostic tests such as computed tomography scans or excessive referrals for gastrointestinal endoscopy poses direct harms to patients, risks overdiagnosis, affects health service waiting lists and adds costs to patients and/or the health system.12 Conversely, over‐relying on less invasive and lower cost investigations such as blood tests or x‐rays can provide false reassurance and may prolong diagnostic intervals.13 Importantly, paths of investigation may also be driven by medicolegal concerns, clinical uncertainty or by patient preferences to be investigated even when the risk of symptoms being attributable to cancer is low.14,15 Considering the costs of tests in relation to the human and health system savings derived from an early stage cancer diagnosis is also a crucial aspect of the discussion.9
The current landscape: cancer guidelines for GPs in Australia
Multiple cancer guidelines, including Cancer Australia's Optimal Care Pathways (OCPs),16 offer GPs guidance on investigating symptoms. OCPs cover various cancer types, from prevention to end‐of‐life care, are accessible online, and can be loaded into general practice software. The Implementing Pathways for Early Cancer Diagnosis (I‐PACED)17 within OCPs provide symptom risk assessment tools. Cancer Australia also offers specific guidelines for lung, breast, ovarian and endometrial cancers, and Cancer Council Australia provides a comprehensive guideline for symptomatic presentations of colorectal cancer.18
The advice provided in the OCPs relating to the initial investigation of symptoms relies primarily on expert specialist opinion rather than applying the extensive body of primary care cancer research generated over the past two decades on the predictive value of symptoms, signs and abnormal test results. Symptoms within these tumour‐specific guidelines can be related to multiple different tumour types. Symptoms attributable to multiple cancer types (as well as many non‐cancer conditions) cannot safely be investigated along a disease‐specific pathway. Prematurely steering patients into specific diagnostic pathways can result in diagnostic delays if the chosen pathway is incorrect, as negative test results within one pathway may erroneously reassure clinicians or patients, only for them to later receive a diagnosis for a different cancer type. For instance, in a patient with abdominal symptoms fitting the bowel cancer OCP, a negative colonoscopy might seem initially reassuring, but ongoing symptoms could ultimately uncover cancer in a different abdominal organ, such as the pancreas or ovaries.13 Another concern relates to the OCP I‐PACED resources and the emphasis on using symptom positive predictive values (PPVs). Assessing symptoms using quantitative PPVs is not a commonly accepted method to guide diagnostic approaches in Australian general practice.19 GPs commonly employ various clinical reasoning approaches, such as intuition and hypothetico‐deductive reasoning, along with a qualitative application of Bayes’ theorem, implicitly rather than explicitly accounting for the predictive values of different symptoms and signs.
The Cancer Australia GP guides18 offer structured guidance that works effectively for symptoms related to breast and lung cancers, where there is little symptom overlap. However, guides such as those for ovarian cancer encounter similar challenges with symptom overlap, where manifestations of various abdominopelvic cancers are depicted but investigations are focused only on ovarian cancer. A syndromic approach that addresses abdominopelvic symptoms shared among multiple cancer types could provide clearer guidance for clinicians. An example of this is the National Institute for Health and Care Excellence (NICE) in the United Kingdom which offers cancer referral guidelines based on presenting symptoms, abnormal test results as well as by cancer site, providing a more relevant option for primary care.20
Inconsistencies in recommendations across guidelines are also evident. For instance, Cancer Australia and OCP guidelines for lung cancer offer differing recommendations regarding imaging modalities for persistent respiratory symptoms after negative chest x‐rays. In addition, some recommendations deviate from evidence‐based practice. For instance, the OCP for pancreatic cancer suggests employing CA 19‐9 testing as an initial investigation for abdominal symptoms, contradicting other guidelines and best practice.20,21
Navigating symptomatic cancer diagnosis: risk assessment tools and decision support
In addition to guidelines, risk assessment and decision support tools are available to assist GPs in diagnosing cancer. Risk assessment tools estimate cancer likelihood using symptoms, clinical and/or demographic data, while decision support tools aid clinicians in making informed decisions by offering evidence‐based recommendations for diagnostic testing.
No cancer risk assessment tools are currently in mainstream use in Australian general practice. Two cancer risk assessment systems have been developed and implemented in the UK. The first system comprises a set of risk assessment tools derived from case–control studies using data from the UK Clinical Practice Research Datalink,22 as is used in the I‐PACED resources. These tools present a two‐way table of the PPVs of symptoms or abnormal blood test results for cancer among patients presenting in general practice (Box 1). The second is an online risk assessment calculator, QCancer (www.qcancer.org), which employs validated multivariable risk prediction algorithms encompassing patient symptoms and risk factors to calculate the likelihood of being diagnosed with 11 specific cancer types or an overall “any cancer” risk (Box 2).
The implementation of these tools has yielded mixed results. A study trialling lung and bowel risk assessment tools found that their use increased referrals for chest x‐rays, colonoscopies and urgent specialist clinics and resulted in more cancer diagnoses.23 However, the likely uptake of these tools and adherence to recommendations may be poor.24 One qualitative study examining the use of QCancer in Australia found it to be helpful for patients with complex medical histories, but experienced doctors were sceptical, especially when it disagreed with their clinical judgment.19 A randomised trial is currently assessing the integration of digital risk assessment tools into primary care electronic medical records, with the stage of cancer at diagnosis the primary outcome.25
The potential role of electronic clinical decision support (eCDS) tools in supporting the delivery of high quality, evidence‐based care is recognised by the Royal Australian College of General Practitioners.26 A recent systematic review of eCDS tools for cancer uncovered evidence of improved decision making but highlighted implementation barriers, including concerns about trust, role compatibility, and impact on clinical workflow.27 A limited number of eCDS tools is available in the Australian setting for symptomatic cancer diagnoses, such as the Cancer Australia lung cancer digital tool (https://islcguide.canceraustralia.gov.au). There is no evidence to date regarding the uptake or effectiveness of such tools. Future Health Today (FHT), a quality improvement tool, is being evaluated in Australian general practices through randomised trials.28 FHT is integrated into general practice electronic medical records to enhance management of a range of medical conditions, including cancer‐specific modules to identify patients potentially at risk of undiagnosed cancer by flagging abnormal test results. Other eCDS are being used in some primary health networks but are yet to be evaluated using randomised trials.29
Conclusion
The unveiling of the Australian Cancer Plan heralds a unique opportunity to elevate current resources and bolster cancer diagnosis within general practice. Recognising the inherent limitations of existing guidelines for GPs underscores the imperative for tailored resources and tools that are co‐designed and cater to the distinctive needs and responsibilities of GPs. This should duly acknowledge the expertise of GPs in investigating ambiguous symptoms that may signal a diverse spectrum of conditions, spanning both cancerous and non‐cancerous ailments. In a busy clinical environment, incorporating any electronic decision support tools to clinical systems must first be supported by evidence that demonstrates their effectiveness and acceptability. In the face of rapid technological advancements and the continuous stream of new evidence, we must also embrace guideline innovation. The use of dynamic, digitally based guidelines is becoming more prevalent through platforms such as the MAGIC authoring and publication platform (MAGICapp; https://magicevidence.org/magicapp/). These guidelines are easily accessible, relevant and updated and help prevent delays in integrating new evidence. By fostering collaborative initiatives that bring together GPs, researchers and cancer agencies, we can effectively bridge the gap between theoretical knowledge and practical application. Through such concerted efforts, we can collectively strive towards the shared goal of enhancing cancer diagnosis within general practice.
Box 1 – Example risk assessment tool for oesophagogastric cancer symptoms*
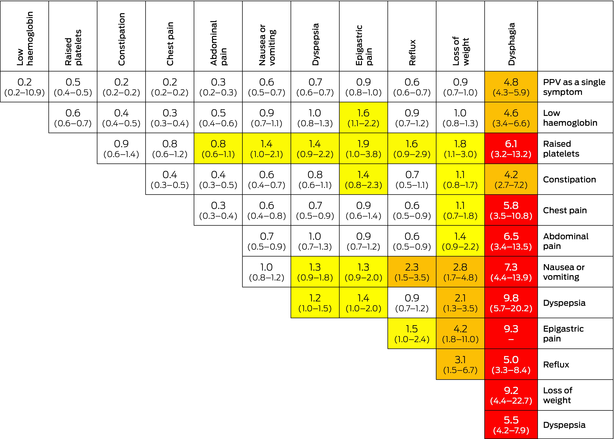
PPV = positive predictive value. * The tool uses colour coding to indicate different levels of PPV. Yellow shading represents a PPV > 1%; amber shading indicates a PPV > 2%; and red shading signifies a PPV > 5.0%. Source: Figure adapted from Stapley et al7
Provenance: Commissioned; externally peer reviewed.
- 1. Cancer Australia. Australian Cancer Plan [website]. Canberra: Commonwealth of Australia, 2021. https://www.canceraustralia.gov.au/australian‐cancer‐plan (viewed Nov 2023).
- 2. Chaji D, Boltong A, Der Vartanian C, et al. Setting the policy agenda for cancer control reform: Australia's first national cancer control plan. Med J Aust 2023; 219: 451‐454. https://www.mja.com.au/journal/2023/219/10/setting‐policy‐agenda‐cancer‐control‐reform‐australias‐first‐national‐cancer
- 3. Emery JD. The challenges of early diagnosis of cancer in general practice. Med J Aust 2015; 203: 391‐393. https://www.mja.com.au/journal/2015/203/10/challenges‐early‐diagnosis‐cancer‐general‐practice
- 4. Hamilton W. Cancer diagnosis in primary care. Br J Gen Pract 2010; 60: 121‐128.
- 5. Rubin G, Berendsen A, Crawford SM, et al. The expanding role of primary care in cancer control. Lancet Oncol 2015; 16: 1231‐1272.
- 6. Koo MM, Hamilton W, Walter FM, et al. Symptom signatures and diagnostic timeliness in cancer patients: a review of current evidence. Neoplasia 2018; 20: 165‐174.
- 7. Stapley S, Peters TJ, Neal RD, et al. The risk of pancreatic cancer in symptomatic patients in primary care: a large case‐control study using electronic records. Br J Cancer 2012; 106: 1940‐1944.
- 8. Jensen H, Tørring ML, Olesen F, et al. Cancer suspicion in general practice, urgent referral and time to diagnosis: a population‐based GP survey and registry study. BMC Cancer 2014; 14: 636.
- 9. McGarvey N, Gitlin M, Fadli E, Chung KC. Increased healthcare costs by later stage cancer diagnosis. BMC Health Serv Res 2022; 22: 1155.
- 10. Emery JD, Shaw K, Williams B, et al. The role of primary care in early detection and follow‐up of cancer. Nat Rev Clin Oncol 2014; 11: 38‐48.
- 11. Renzi C, Kaushal A, Emery J, et al. Comorbid chronic diseases and cancer diagnosis: disease‐specific effects and underlying mechanisms. Nat Rev Clin Oncol 2019; 16: 746‐761.
- 12. Brownlee S, Chalkidou K, Doust J, et al. Evidence for overuse of medical services around the world. Lancet 2017; 390: 156‐168.
- 13. Lyratzopoulos G, Vedsted P, Singh H. Understanding missed opportunities for more timely diagnosis of cancer in symptomatic patients after presentation. Br J Cancer 2015; 112 (Suppl): S84‐S91.
- 14. Banks J, Hollinghurst S, Bigwood L, et al. Preferences for cancer investigation: a vignette‐based study of primary‐care attendees. Lancet Oncol 2014; 15: 232‐240.
- 15. Venning B, Pearce A, Lourenco RDA, et al. Patient preferences for investigating cancer‐related symptoms in australian general practice: a discrete choice experiment. Br J Gen Pract 2024; https://doi.org/10.3399/BJGP.2023.0583 [Epub ahead of print].
- 16. Cancer Australia. Optimal Care Pathways. Canberra: Commonwealth of Australia, 2024. https://www.canceraustralia.gov.au/optimal‐cancer‐care‐pathways (viewed Apr 2024).
- 17. Cancer Australia. Optimal Care Pathways: for general practitioners and primary care staff — I‐PACED (Implementing Pathways for Cancer Early Diagnosis). Canberra: Commonwealth of Australia, 2024. https://www.cancervic.org.au/get‐support/for‐health‐professionals/optimal‐care‐pathways (viewed Apr 2024).
- 18. Cancer Australia. GP guides and resources. https://www.canceraustralia.gov.au/clinical‐best‐practice/gynaecological‐cancers/gp‐guides‐and‐resources. Canberra: Commonwealth of Australia, 2021. (viewed Apr 2024).
- 19. Chiang PPC, Glance D, Walker J, et al. Implementing a QCancer risk tool into general practice consultations: an exploratory study using simulated consultations with Australian general practitioners. Br J Cancer 2015; 112 (Suppl): S77‐S83.
- 20. National Institute for Health and Care Excellence. Suspected cancer: recognition and referral [NICE guideline NG12; updated Oct 2023]. Manchester (UK): NICE, 2015. https://www.nice.org.uk/guidance/ng12 (viewed Mar 2024).
- 21. Conroy T, Pfeiffer P, Vilgrain V, et al. Pancreatic cancer: ESMO clinical practice guideline for diagnosis, treatment and follow‐up. Ann Oncol 2023; 34: 987‐1002.
- 22. Hamilton W. The CAPER studies: five case‐control studies aimed at identifying and quantifying the risk of cancer in symptomatic primary care patients. Br J Cancer 2009; 101 (Suppl): S80‐S86.
- 23. Hamilton W, Green T, Martins T, et al. Evaluation of risk assessment tools for suspected cancer in general practice: a cohort study. Br J Gen Pract 2013; 63: e30‐e36.
- 24. Price S, Spencer A, Medina‐Lara A, Hamilton W. Availability and use of cancer decision‐support tools: a cross‐sectional survey of UK primary care. Br J Gen Pract 2019; 69: e437‐e443.
- 25. Hamilton W, Mounce L, Abel GA, et al. Protocol for a pragmatic cluster randomised controlled trial assessing the clinical effectiveness and cost‐effectiveness of Electronic RIsk‐assessment for CAncer for patients in general practice (ERICA). BMJ Open 2023; 13: e065232.
- 26. Royal Australian College of General Practitioners. Electronic clinical decision support in general practice [website]. Melbourne: RACGP, 2024. https://www.racgp.org.au/advocacy/position‐statements/view‐all‐position‐statements/clinical‐and‐practice‐management/electronic‐clinical‐decision‐support (viewed Mar 2024).
- 27. Chima S, Reece JC, Milley K, et al. Decision support tools to improve cancer diagnostic decision making in primary care: a systematic review. Br J Gen Pract 2019; 69: e809‐e818.
- 28. Hunter B, Biezen R, Alexander K, et al. Future Health Today: codesign of an electronic chronic disease quality improvement tool for use in general practice using a service design approach. BMJ Open 2020; 10: e040228.
- 29. Davies D, Morgan M, de Wet C. Supporting quality and safety in general practice: Response rates to computer decision support. Aust J Gen Pract 2022; 51: 884‐892.






Open access:
Open access publishing facilitated by The University of Melbourne, as part of the Wiley ‐ The University of Melbourne agreement via the Council of Australian University Librarians.
Brent Venning is supported by the Melbourne Academic Centre for Health (MACH) through the MACH‐Track program and by a University of Melbourne Research Training Program Scholarship. Jon Emery is supported by a National Health and Medical Research Council Investigator Grant (APP1195302) and is an Associate Director of the CanTest Collaborative (funded by Cancer Research UK, C8640/A23385).
No relevant disclosures.