Clinical record
A 20‐year‐old man presented to hospital with 3 days of pleuritic chest pain, fevers and diaphoresis. Past medical history was significant for asthma and depression. Medications included fluticasone–salmeterol and mirtazapine. He had no personal or family history of myocarditis or inflammatory disorders. He was a non‐smoker with social alcohol intake, and did not report having used illicit drugs.
Symptom onset occurred 12 hours after administration of his second dose of the Pfizer (Comirnaty) coronavirus disease 2019 (COVID‐19) vaccine, with headaches, night sweats and chills. He developed chest pain at 48 hours. The first dose of the vaccine was administered 23 days before the second dose and was uncomplicated.
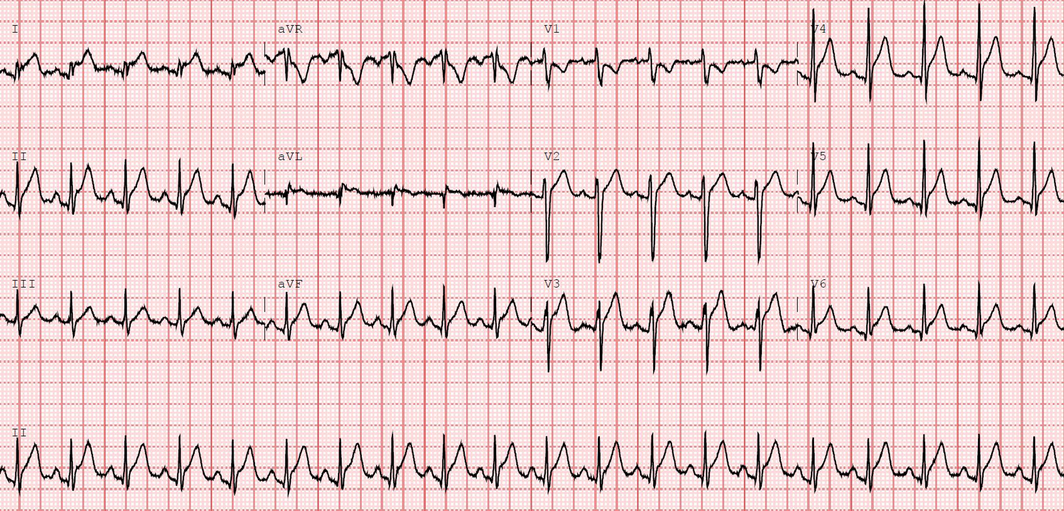
On presentation, he was febrile (38.1°C) and had tachycardia (130 beats/min). An electrocardiogram (ECG) revealed sinus tachycardia with global ST elevation (Box 1). His initial troponin level was 3557 ng/L (reference interval [RI], < 26 ng/L), peaking at 9853 ng/L 3 hours later. His C‐reactive protein concentration was elevated at 50.9 mg/L (RI, < 5.0 mg/L) and erythrocyte sedimentation rate was 45 mm/h (RI, 1–19 mm/h). Full blood examination, biochemistry and liver function results were all normal. Septic screening (chest x‐ray and urine and blood cultures) was negative. A SARS‐CoV‐2 nucleic acid test result was also negative, and a limited vasculitic screen was unremarkable.
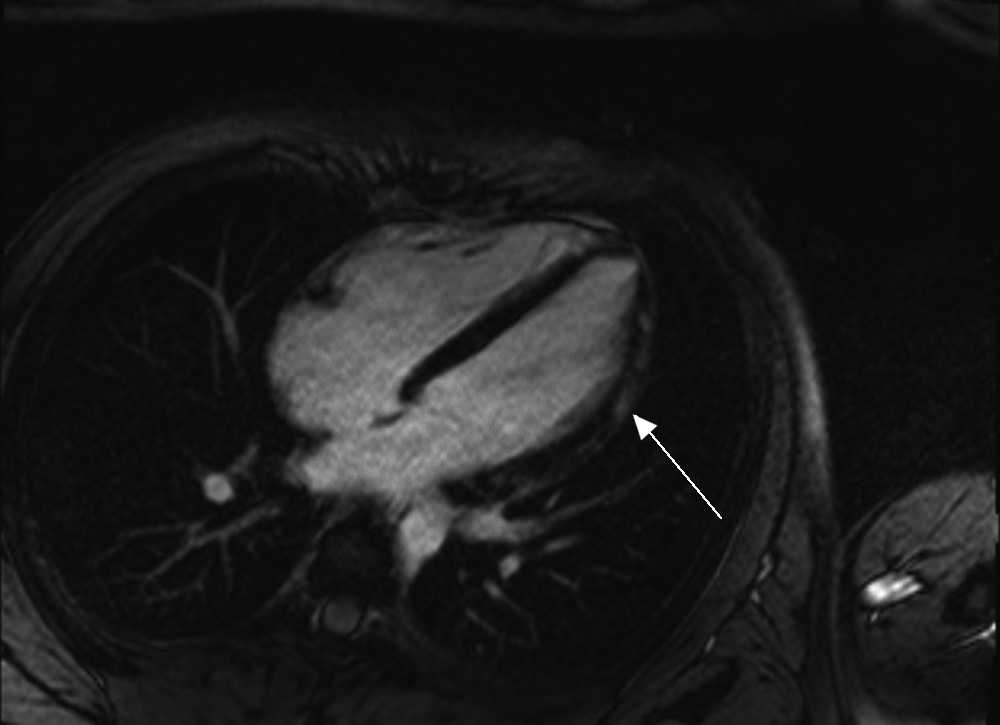
Transthoracic echocardiography showed normal biventricular function with no pericardial effusion. Cardiac magnetic resonance imaging showed normal left ventricular size and mildly impaired systolic function with hypokinesis of the mid and apical lateral segments associated with epicardial late gadolinium enhancement (Box 2), consistent with myocarditis.
He was commenced on ibuprofen 400 mg three times a day, pantoprazole 40 mg daily (to reduce risk of gastritis), and bisoprolol 2.5 mg daily, with complete resolution of symptoms within 24 hours. No rhythm disturbances were detected on telemetry and he was discharged after 48 hours. He was advised to avoid strenuous activity and scheduled for repeat transthoracic echocardiography in 3 months.
Discussion
Myocarditis is a known complication of SARS‐CoV‐2 infection occurring in 0.3–2.3% of patients.1 However, there has been emerging evidence of myocarditis following COVID‐19 messenger RNA (mRNA)‐based vaccinations (Pfizer [Comirnaty] and Moderna [Spikevax] vaccines). By 2 January 2022, 415 cases of myocarditis temporally related to Pfizer (Cominarty) COVID‐19 mRNA vaccination had been reported to the Therapeutic Goods Administration,2 although not all may be attributable to the vaccine.
COVID‐19 mRNA vaccine‐related myocarditis is more common in males, younger patients and following the second or subsequent dose. A US Centers for Disease Control and Prevention summary described the highest incidence of myocarditis in males aged 12–29 years, with 40.6 per million males affected following the second dose, compared with only 4.2 per million females aged 12–29 years. Rates decreased to 2.4 and 1.0 per million in males and females aged over 30 years, respectively.3 Notably, the background incidence of myocarditis in the general population aged 18–34 years is estimated at 37 per 100 000 patient years in males and 16 per 100 000 patient years in females.2 Among 1226 cases of mRNA vaccination‐related myocarditis, no deaths were reported.3
In a recent case series, all 61 patients diagnosed with COVID‐19 vaccination‐related myocarditis had mild presentations with chest pain and troponinaemia mostly within 3 days of vaccination.4 The vast majority (87%) had ECG abnormalities, most commonly ST‐segment elevation, and all had cardiac magnetic resonance imaging findings, most commonly late gadolinium enhancement. Left ventricular dysfunction occurred in around one‐third of patients. Treatments included non‐steroidal anti‐inflammatories, colchicine and, less commonly, steroids or intravenous immunoglobulin. Long term outcomes of COVID‐19 vaccine‐related myocarditis have not yet been described, but in this series nearly 90% of patients had symptom resolution within a median hospitalisation duration of 4.6 days.4 A case series reporting incomplete symptom resolution found seven of 23 otherwise healthy military members with vaccine‐related myocarditis had ongoing chest discomfort but were otherwise well enough for discharge.5
COVID‐19 mRNA vaccine‐related myocarditis is an extremely rare and mild complication, and is much less frequent than myocarditis secondary to SARS‐CoV‐2 infection. In the setting of potential myocarditis or pericarditis, current Australian guidelines suggest management in the primary care or outpatient setting is appropriate if presentation is mild and if ECG, troponin and inflammatory markers can be reviewed within 12 hours. If the patient is unwell, has chest pain and is aged over 30, or if ECG findings are abnormal, emergency department referral is recommended. Confirmed myocarditis or pericarditis may require brief hospitalisation for cardiac monitoring, or cardiology referral for additional investigations.2
Given the scarcity, mild presentation and clinical course of mRNA vaccine‐related myocarditis, and the efficacy of vaccines in reducing SARS‐CoV‐2 infection, the benefit–risk assessment for COVID‐19 mRNA vaccination is still overwhelmingly favourable for all age and sex groups, including young people.4
- • COVID‐19 mRNA vaccine‐related myocarditis is an extremely rare side effect that is most common in males aged 12–29 years, after the second vaccine dose.
- • Symptoms are usually mild, requiring only brief hospitalisation, and treatments are readily available.
- • Initial review and work‐up may occur in the outpatient setting if patients are young (< 30 years), clinically stable and investigation results can be obtained in a timely manner. Confirmed myocarditis or pericarditis may involve emergency department review, hospitalisation and/or cardiology referral.
- • COVID‐19 vaccinations (including boosters) are crucial in reducing the spread and severity of infection. The incidence and severity of COVID‐19‐related myocarditis is higher than vaccine‐related myocarditis. Thus, vaccine‐related myocarditis should not impede vaccination efforts.
Provenance: Not commissioned; externally peer reviewed.
- 1. Daniels CJ, Rajpal S, Greenshields JT, et al. Prevalence of clinical and subclinical myocarditis in competitive athletes with recent SARS‐CoV‐2 infection: results from the Big Ten COVID‐19 Cardiac Registry. JAMA Cardiol 2021; 6: 1078–1087.
- 2. Australian Government. Guidance on myocarditis and pericarditis after mRNA COVID‐19 vaccines. Canberra: Australian Government, 2021. https://www.health.gov.au/sites/default/files/documents/2021/11/covid‐19‐vaccination‐guidance‐on‐myocarditis‐and‐pericarditis‐after‐mrna‐covid‐19‐vaccines_1.pdf (viewed Jan 2022).
- 3. Gargano JW, Wallace M, Hadler SC, et al. Use of mRNA COVID‐19 vaccine after reports of myocarditis among vaccine recipients: update from the Advisory Committee on Immunization Practices — United States, June 2021. MMWR Morb Mortal Wkly Rep 2021; 70: 977–982.
- 4. Bozkurt B, Kamat I, Hotez PJ. Myocarditis with COVID‐19 mRNA vaccines. Circulation 2021; 144: 471–484.
- 5. Montgomery J, Ryan M, Engler R, et al. Myocarditis following immunization with mRNA COVID‐19 vaccines in members of the US Military. JAMA Cardiol 2021; 6: 1202–1206.





No relevant disclosures.