The known: E‐cigarettes are increasingly popular in Australia, but little is known about the chemicals inhaled by users. Evidence is mounting that e‐cigarettes are not benign and can pose significant health risks.
The new: Our comprehensive chemical assessment of Australian e‐liquids identified a wide range of potentially harmful chemicals in these liquids, both in their purchased forms and after simulated vaping.
The implications: Australian e‐liquids include potentially toxic chemicals, for many of which no information on inhalation health effects are available. Despite the sale of nicotine‐containing e‐liquids without prescription being illegal, trace amounts were detected in some samples, with implications for their effects on health and addiction.
Electronic cigarettes (e‐cigarettes) are a relatively recent development, and are often marketed as alternative nicotine delivery systems for tobacco smokers.1 Research into e‐cigarettes has increased substantially in recent years, together with debate about their role in tobacco harm reduction and in normalising cigarette smoking. Central to the discussion are the potential health effects of e‐cigarettes, many of which are related to the chemical composition of the “e‐liquid” used.2
E‐liquids typically consist of a glycerol and propylene glycol base, flavourings, nicotine, and other chemicals that are heated, aerosolised, and inhaled. An emerging body of evidence suggests that e‐liquids often contain a range of potentially toxic chemicals.2,3,4,5,6,7,8,9,10 However, information about the composition of e‐liquids used in Australia is limited. This is important, as the Australian regulatory status of e‐cigarettes and e‐liquids is complex, differentiating the market from that of many other countries. E‐cigarettes and e‐liquids that do not contain nicotine are largely unregulated in most states, and no devices have been approved for therapeutic use.11 However, we have previously reported that e‐liquids labelled “nicotine‐free” often contain nicotine.6 Further, there is evidence that some common e‐liquid ingredients react with each other9 or with components of the e‐cigarette device,12 or may be converted into other chemical species when heated.
Analysis of e‐liquids that have been “aged” by repeated heating and cooling may therefore provide additional information about the potential effects on health of e‐cigarette use. We employed gas chromatography‒mass spectrometry to analyse 65 Australian e‐liquids for common excipients, flavourings, nicotine, polycyclic aromatic hydrocarbons, and other chemicals, both in their fresh state and after subjecting the e‐liquids to an accelerated ageing process that simulates the effect of “vaping”.
Methods
We purchased a range of e‐liquids from Australian online and brick‐and‐mortar stores. Online suppliers of e‐liquids were deemed eligible sources if they were operational, were based in Australia, sold e‐liquids (some stores sold only e‐cigarette devices), and sold their own brand of e‐liquids. Online suppliers were identified in a Google search (February 2020) for the term “Australian e‐liquid”. Of the first 25 search results, ten were for eligible online e‐liquid suppliers; we purchased the five best selling e‐liquids from each supplier, as reported on their websites. We also purchased 15 e‐liquids from brick‐and‐mortar stores in our home state, Western Australia, selected as a cross‐section of local brands and manufacturers at the time of purchase; three were manufactured in New South Wales, 12 in Western Australia. All 65 e‐liquids were described as being nicotine‐free, and they encompassed a wide range of flavours, including ice/menthol, fruit, dessert, and tobacco (Supporting Information 1).
Selection of chemicals for analysis
On the basis of similar studies,2,6,8 we selected chemicals for analysis that we expected to find in e‐liquids or were known toxins associated with e‐cigarettes, including common excipients and solvents (glycerol, propylene glycol, benzyl alcohol), flavouring chemicals (menthol, eugenol, thymol, benzaldehyde, trans‐cinnamaldehyde, ethyl vanillin, ethyl maltol, furfural, 4‐(4‐methoxyphenyl)‐2‐butanone), and other e‐liquid constituents (nicotine, nicotyrine, 2‐chlorophenol, phenol). We also measured the levels of ten polycyclic aromatic hydrocarbons (PAHs) — large hydrocarbons associated with thermal degradation — for which certified reference material standards were available.
E‐liquid analyses
Our analytical methods are fully described in online Supporting Information 2. Briefly, analytical standards for each chemical were purchased from Sigma‐Aldrich Australia, Rowe Scientific, or Cayman Chemical. As a simple solvent does not reflect the sample matrix of commercial e‐liquids, standards were prepared for analysis in 50% propylene glycol/50% glycerol.
Analytes were extracted from the sample matrix by solid phase micro‐extraction, which enables solventless extraction using a fused silica or stainless steel fibre coated with a thin film polymer. Headspace (gas phase) analysis using solid phase micro‐extraction removes a large fraction of the interference by the propylene glycol/glycerol matrix (> 90% of the sample by mass), and has previously been used for the analysis of volatile compounds, additives, and flavours in e‐liquids.13 We used a divinylbenzene/carboxen/polydimethylsiloxane fibre for all analyses (Supelco). Polar compounds were separated on a 30 m × 0.25 mm HP‐INNOWax (polyethylene glycol) analytical column (Agilent J&W), with a film thickness of 0.25 µm and 4‐bromophenol‐2,3,5,6‐d4 as internal standard. Non‐polar compounds and PAHs were analysed on a 30 m × 0.25 mm Zebron ZB‐5MS analytical column (Phenomenex) with a film thickness of 1 µm and biphenyl‐d10 as internal standard. Analytes were detected by mass spectrometry (Agilent 6890N GC/Agilent 5973 network mass selective detector) in electron impact ionisation mode (70 eV); to optimise sensitivity, compounds were quantified in selected ion monitoring mode.
E‐liquid ageing
To evaluate chemical changes resulting from vaping (aerosol generation from evaporation and condensation), which can cause thermal decomposition, oxidation, and polymerisation of e‐liquid components, we developed a method for simulating the ageing of e‐liquids (Supporting Information 2). After accelerated ageing, e‐liquids were re‐analysed using the same techniques as for fresh e‐liquids.
Ethics approval
Ethics approval was not required for this study.
Results and discussion
Excipients and solvents
Propylene glycol and glycerol were the main ingredients by proportion in each e‐liquid. The propylene glycol/glycerol ratio was not specified for three e‐liquids. Most e‐liquids were labelled as including 30% propylene glycol/70% glycerol, but the actual value was within 10 percentage points of the labelled amount for only 11 (propylene glycol) or 21 (glycerol) of these 62 e‐liquids; in one case, the propylene glycol/glycerol ratio was the reverse of that indicated on the label. The mean propylene glycol content was 77.4% of the label value (standard deviation [SD], 18.5%; range 43.7‒126.8%), the mean glycerol content was 111.8% of the label value (SD, 9.7%; range, 88.5‒137.5%) (Box 1). The magnitude of these differences was consistent with manufacturers mixing propylene glycol and glycerol on a volume‐for‐volume rather than a weight‐for‐weight basis; as the density of propylene glycol is 1.04 g/cm³ and that of glycerol is 1.26 g/cm³, volume‐based mixing will increase the amount of glycerol by about 21% of the intended level.
Benzyl alcohol, a solvent/flavour enhancer, was found in 42 of 65 e‐liquids and 32 of 65 aged e‐liquids, at levels of up to 1687 mg/L (Supporting Information 1). Benzyl alcohol, also found in e‐liquids by other studies,8,14,15 is a dermal sensitising agent and skin allergen that elicits severe reactions in some people.16
Nicotine
Nicotine was found in trace amounts in six fresh e‐liquids (maximum, 3.25 mg/L) but not in aged e‐liquids (Box 2; Supporting Information 1). In our earlier study, six of ten e‐liquids contained nicotine (maximum, 2900 mg/L).6 The results for our more recent samples may indicate cleaner manufacturing processes, or that nicotine was present as nicotine salts rather than as freebase nicotine.17 Nicotine is relatively common in “nicotine‐free” e‐liquids,18 with implications for health and addiction. Nicotine in Australian “nicotine‐free” e‐liquids could be the result of accidental contamination or poor quality control during manufacture.
Nicotyrine, formed by the dehydrogenation and oxidation of nicotine,5 is not usually detected in e‐liquids unless they have been exposed to air, in which case it accumulates over time. We detected it in seven fresh (including two that also contained nicotine) and nine aged e‐liquids (maximum, 2.9 µg/L), indicating that they had previously contained nicotine.
Flavouring chemicals
A range of flavouring chemicals were detected in fresh and aged e‐liquids (Box 3; Supporting Information 1). Some, including furfural, thymol, and 4‐(4‐methoxyphenyl)‐2‐butanone were found only infrequently or at very low levels. Benzaldehyde, added to e‐liquids for its almond‐like flavour, was detected in 60 fresh and 61 aged e‐liquids at concentrations ranging from 11.4 µg/L to 17.3 mg/L. Benzaldehyde inhibits microsomal cytochrome P450 2A6 (CYP2A6)19 — increasing systemic nicotine exposure and blood nicotine concentrations in smokers20 — reduces phagocytosis,10 and is an inhalation irritant.21 Benzaldehyde can also react with propylene glycol in e‐liquids, producing aldehyde propylene glycol acetals9 that activate airway irritant receptors.22
Other flavouring chemicals we found frequently or at high concentrations were:
- menthol: 44 fresh (maximum, 205 mg/L) and 50 aged e‐liquids (maximum, 176 mg/L);
- ethyl maltol: 58 fresh (maximum, 2583 mg/L) and 52 aged e‐liquids (maximum, 4084 mg/L);
- trans‐cinnamaldehyde: 48 fresh (maximum, 97.9 mg/L) and 38 aged e‐liquids (maximum, 142.5 mg/L); and
- ethyl vanillin: 59 fresh (maximum, 2192 mg/L) and 50 aged e‐liquids (maximum, 3393 mg/L).
The high frequency of detection and the high concentrations of these chemicals have concerning health implications. Menthol enhances the addictive properties of nicotine23 and inhibits nicotine metabolism.20 Menthol was detected in most e‐liquids, but only a small proportion were labelled as being “menthol”‐ or “ice”‐flavoured. Conversely, one “menthol” e‐liquid contained no menthol, and may have instead contained potentially carcinogenic analogues such as pulegone, or synthetic “coolants” such as N‐ethyl‐p‐menthane‐3‐carboxamide.
Ethyl maltol is added to e‐liquids as a sweetener. The effects of heating and inhaling it are largely unknown, but it increases free radical formation in e‐cigarette aerosols.4 Free radicals induce oxidative stress, which affect cell survival and proliferation, and inflammation. Ethyl maltol reacts with iron and copper (potentially present in e‐liquids as coil residue) to produce toxic hydroxypyranone complexes.12
trans‐Cinnamaldehyde impairs innate immune cell function in the lung,3 suppresses bronchial airway epithelial cell ciliary motility and mitochondrial function,7 inhibits microsomal CYP2A6, impairs neutrophil, macrophage and natural killer cell function, and reduces oxidative burst when heated and inhaled.10
Ethyl vanillin is used widely in foods, beverages, cosmetics, and drugs for its potent vanilla odour and flavour. In e‐liquids, it reduces oxidative burst10 and inhibits in vitro free radical formation.4 Like benzaldehyde, both trans‐cinnamaldehyde and ethyl vanillin react with propylene glycol in e‐liquids to produce aldehyde propylene glycol acetals.9
Other chemicals
We found 2‐chlorophenol in 27 fresh and 30 aged samples, at concentrations of up to 206 mg/L (Box 2; Supporting Information 1). Similar chemicals have been identified as pesticide or herbicide residues or decomposition by‐products in canola oil,24 from which glycerol is derived. While not as ubiquitous as in our earlier study,6 this acutely toxic chemical, used in disinfectants and insecticides, remains a problem for the e‐liquid manufacturing process.
Polycyclic aromatic hydrocarbons
PAHs are produced during the thermal decomposition of organic material, including tobacco and fossil fuels. Lower temperature thermal decomposition in e‐cigarettes generally produces a greater proportion of low molecular weight PAHs, such as acenaphthylene, fluorene, and anthracene. Most PAHs are known or suspected carcinogens, and exposure has been linked with a range of adverse health effects in humans.25 The health effects of inhaling specific individual PAHs, however, have not been studied in detail. We generally detected PAHs at very low levels (Box 4; Supporting Information 1). The accelerated ageing process did not increase the levels of any PAH; those of benzo[a]pyrene, benzo[b]fluoranthene, benzo[k]fluoranthene, and pyrene were all markedly lower in aged than in fresh e‐liquids; indeed, benzo[a]pyrene and benzo[b]fluoranthene were not detected in aged e‐liquids. These high molecular weight PAHs may have been chemically modified to low molecular weight PAHs (which we did not analyse), or PAHs formed during the ageing process may have been preferentially volatilised and not recovered.
Health effects of inhaled e‐liquid aerosols
To estimate the potential effect on health of inhaling aerosols generated from e‐liquids, some assumptions must be made. As acute inhalation toxicity levels (median lethal concentrations that kill 50% of a test animal population [LC50]) have not been established for most of the chemicals we detected, we cannot calculate no observed adverse effect levels (NOAELs). Of the chemicals with established inhalation LC50 values, we found that four (benzaldehyde, menthol, 2‐chlorophenol, benzyl alcohol) were frequently detected in e‐liquids at concentrations that exceeded the inhalation LC50. However, the inhalation LC50 is measured in air, and it is not possible to directly convert concentrations in e‐liquids to concentrations in air without assumptions about puff volume and rate of use. Further research is needed to determine whether aerosols generated from these e‐liquids contain these chemicals at relevant concentrations.
Conclusion
Our findings support and build upon earlier reports on the chemical composition of e‐liquids. We found that a range of harmful chemicals are present, and that the heating/cooling/ageing process can affect e‐liquid chemical composition. We acknowledge some limitations to our investigation, including the fact that we measured only a pre‐determined selection of chemical analytes. Future studies would benefit from a discovery approach to identifying novel chemicals in e‐liquids. Further, we acknowledge that the chemical composition of e‐liquids is not entirely representative of the aerosol inhaled by e‐cigarette users. Nevertheless, our finding that every e‐liquid tested contained one or more chemicals potentially harmful to health provides a clear motivation for further investigations.
Box 1 – Propylene glycol and glycerol content of 62 e‐liquids, as proportions of the labelled amount*
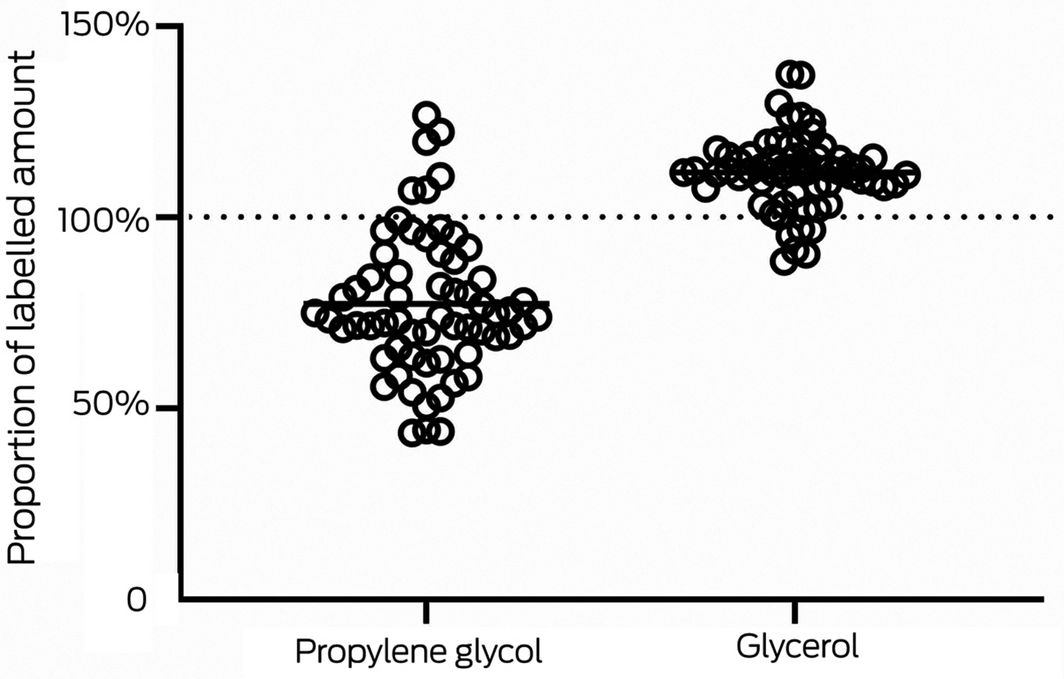
* The solid horizontal lines indicate the respective mean levels of propylene glycol or glycerol.
Box 2 – Concentrations of non‐flavouring chemicals in 65 Australian e‐liquids, fresh and after ageing
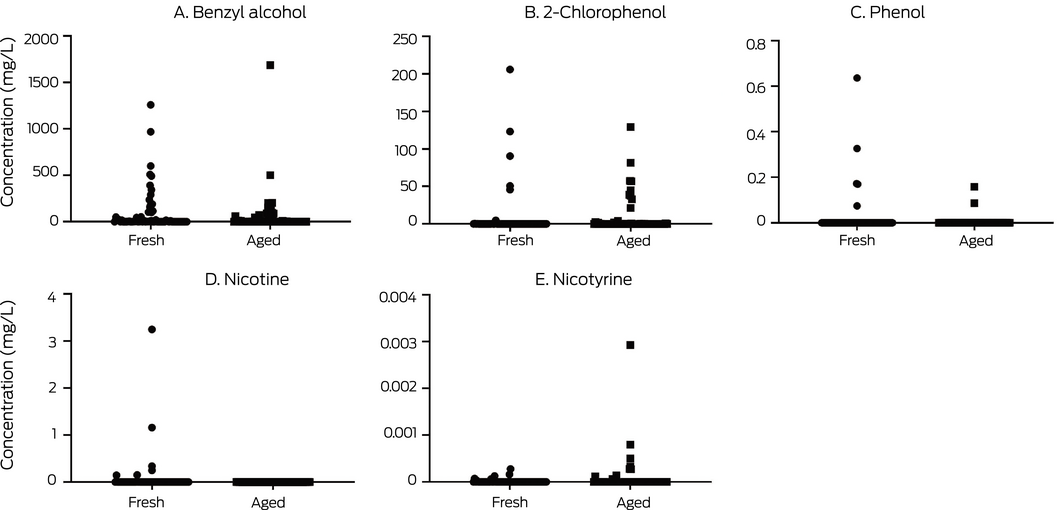
The values for individual products are included in the online Supporting Information 1.
Box 3 – Concentration of flavouring chemicals in 65 Australian e‐liquids, fresh and after ageing
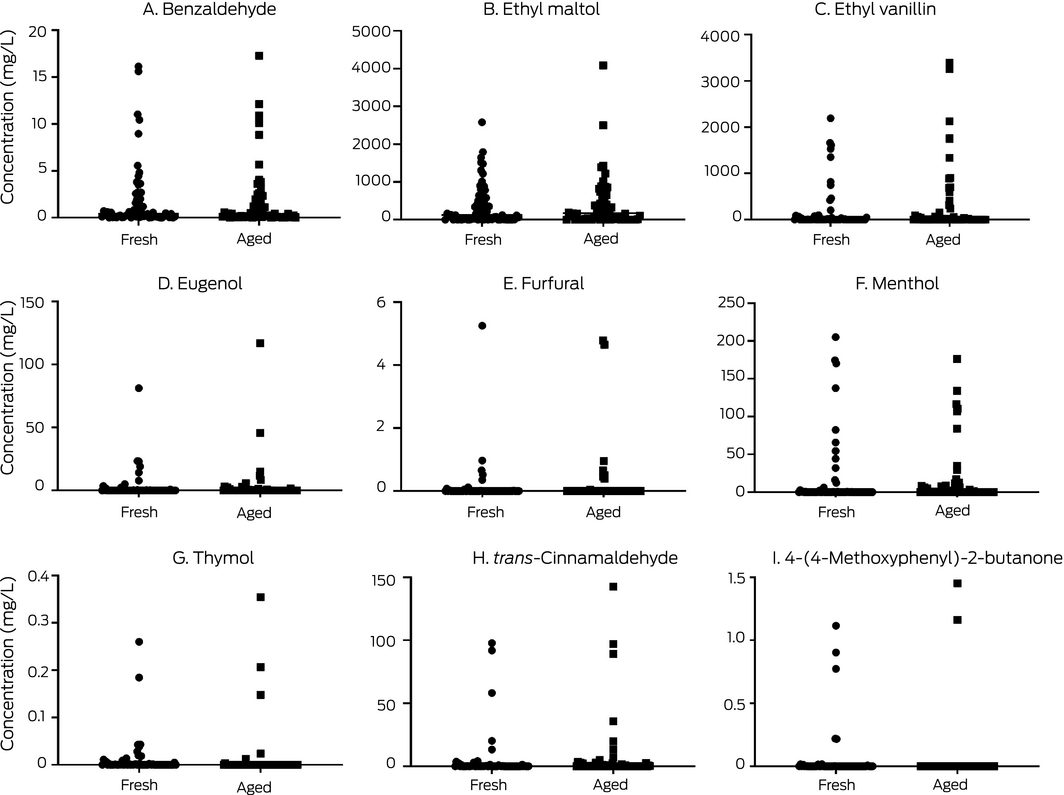
The values for individual products are included in the online Supporting Information 1.
Box 4 – Concentration of polycyclic aromatic hydrocarbons in 65 Australian e‐liquids, fresh and after ageing
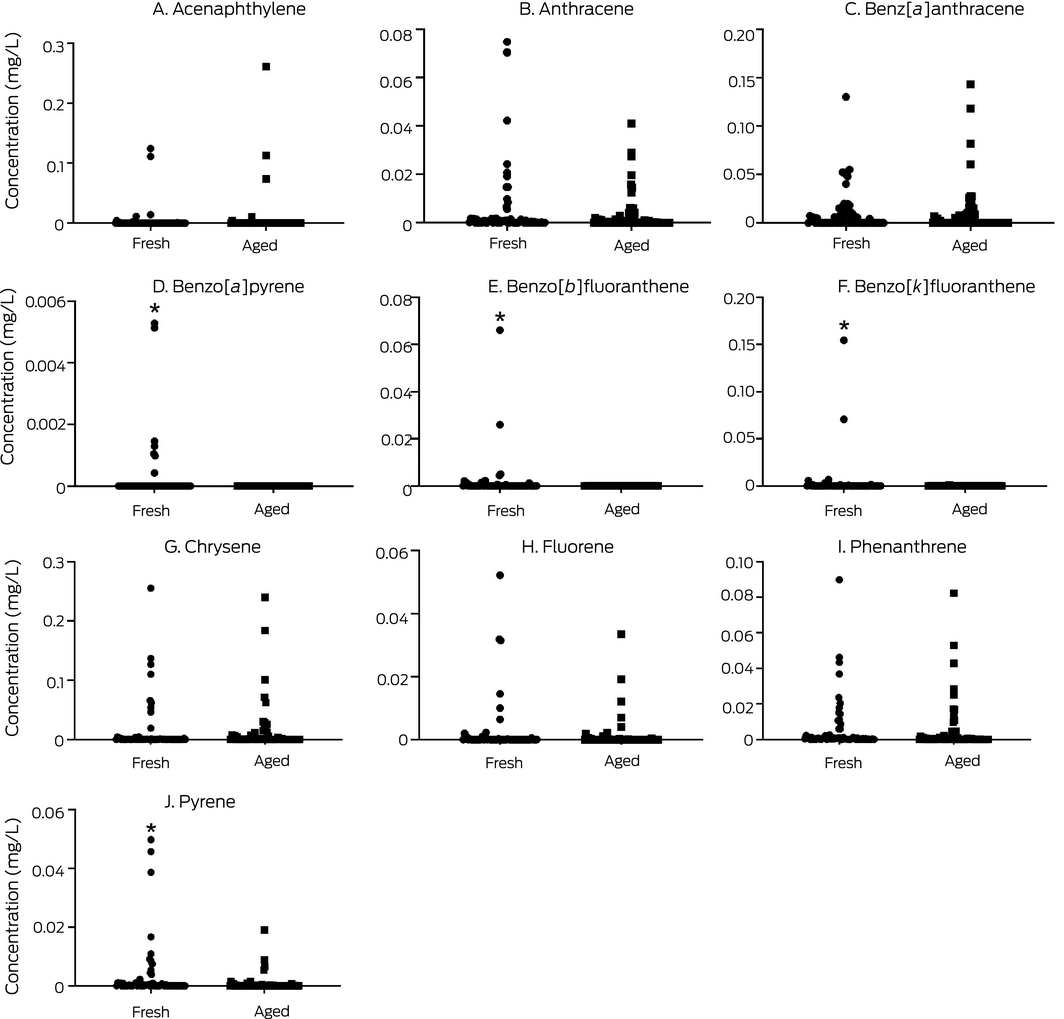
The values for individual products are included in the online Supporting Information 1. * Statistically significantly higher median concentration in fresh than aged samples (Mann–Whitney test; P < 0.05).
Received 18 February 2021, accepted 19 June 2021
- Alexander Larcombe1,2
- Sebastien Allard3
- Paul Pringle3
- Ryan Mead‐Hunter2,4
- Natalie Anderson1,2
- Benjamin Mullins2,4
- 1 Telethon Kids Institute, Perth, WA
- 2 Curtin University, Perth, WA
- 3 Curtin Water Quality Research Centre, Curtin University, Perth, WA
- 4 Curtin Institute for Computation, Curtin University, Perth, WA
This investigation was commissioned by Lung Foundation Australia and the Minderoo Foundation, and was funded by the Minderoo Foundation, Lung Foundation Australia, Cancer Council WA, and the Scottish Masonic Charitable Foundation.
No relevant disclosures.
- 1. Worku D, Worku E. A narrative review evaluating the safety and efficacy of e‐cigarettes as a newly marketed smoking cessation tool. SAGE Open Med 2019; 7: 2050312119871405.
- 2. Girvalaki C, Tzatzarakis M, Kyriakos CN, et al. Composition and chemical health hazards of the most common electronic cigarette liquids in nine European countries. Inhal Toxicol 2018; 30: 361–369.
- 3. Clapp PW, Pawlak EA, Lackey JT, et al. Flavored e‐cigarette liquids and cinnamaldehyde impair respiratory innate immune cell function. Am J Physiol Lung Cell Mol Physiol 2017; 313: L278–L292.
- 4. Bitzer ZT, Goel R, Reilly SM, et al. Effect of flavoring chemicals on free radical formation in electronic cigarette aerosols. Free Radic Biol 2018; 120: 72–79.
- 5. Son Y, Wackowski O, Weisel C, et al. Evaluation of e‐vapor nicotine and nicotyrine concentrations under various e‐liquid compositions, device settings, and vaping topographies. Chem Res Toxicol 2018; 31: 861–868.
- 6. Chivers E, Janka M, Franklin P, et al. Nicotine and other potentially harmful compounds in “nicotine‐free” e‐cigarette liquids in Australia. Med J Aust 2019; 210: 127–128. https://www.mja.com.au/journal/2019/210/3/nicotine‐and‐other‐potentially‐harmful‐compounds‐nicotine‐free‐e‐cigarette
- 7. Clapp PW, Lavrich KS, van Heusden CA, et al. Cinnamaldehyde in flavored e‐cigarette liquids temporarily suppresses bronchial epithelial cell ciliary motility by dysregulation of mitochondrial function. Am J Physiol Lung Cell Mol Physiol 2019; 316: L470–L486.
- 8. Czoli CD, Goniewicz ML, Palumbo M, et al. Identification of flavouring chemicals and potential toxicants in e‐cigarette products in Ontario, Canada. Can J Public Health 2019; 110: 542–550.
- 9. Erythropel HC, Jabba SV, DeWinter TM, et al. Formation of flavorant–propylene glycol adducts with novel toxicological properties in chemically unstable e‐cigarette liquids. Nicotine Tob Res 2019; 21: 1248–1258.
- 10. Hickman E, Herrera CA, Jaspers I. Common e‐cigarette flavoring chemicals impair neutrophil phagocytosis and oxidative burst. Chem Res Toxicol 2019; 32: 982–985.
- 11. Greenhalgh EM, Grace C, Scollo MM. Electronic cigarettes (e‐cigarettes) (InDepth 18B). In: Scollo MM, Winstanley MH editors. Tobacco in Australia: facts and issues. Melbourne: Cancer Council Victoria, 2018. https://www.tobaccoinaustralia.org.au/chapter‐18‐harm‐reduction/indepth‐18b‐e‐cigarettes/18b‐9‐legal‐status (viewed Sept 2021).
- 12. Li Z, Lu J, Wu C, et al. Toxicity studies of ethyl maltol and iron complexes in mice. Biomed Res Int 2017; 2017.
- 13. Bansal V, Hashemi B, Raza N, et al. Review of the analytical methods for and clinical impact of additives and flavors used in electronic cigarettes. Expos Health 2020; 12: 593–615.
- 14. Hutzler C, Paschke M, Kruschinski S, et al. Chemical hazards present in liquids and vapors of electronic cigarettes. Arch Toxicol 2014; 88: 1295–1308.
- 15. Schober W, Szendrei K, Matzen W, et al. Use of electronic cigarettes (e‐cigarettes) impairs indoor air quality and increases FeNO levels of e‐cigarette consumers. Int J Hyg Environ Health 2014; 217: 628–637.
- 16. Chow ET, Avolio AM, Lee A, Nixon R. Frequency of positive patch test reactions to preservatives: the Australian experience. Australas J Dermatol 2013; 54: 31–35.
- 17. Ghosh A, Beyazcicek O, Davis ES, et al. Cellular effects of nicotine salt containing e‐liquids. J Appl Toxicol 2020; 41: 493–505.
- 18. Omaiye EE, Cordova I, Davis B, Talbot P. Counterfeit electronic cigarette products with mislabeled nicotine concentrations. Tob Regul Sci 2017; 3: 347–357.
- 19. Adams T, Cohen SM, Doull J, et al; Expert Panel of the Flavor and Extract Manufacturers Association. The FEMA GRAS assessment of benzyl derivatives used as flavor ingredients. Food Chem Toxicol 2005; 43: 1207–1240.
- 20. Benowitz NL, Herrera B, Jacob P. Mentholated cigarette smoking inhibits nicotine metabolism. J Pharmacol Exp Ther 2004; 310: 1208.
- 21. Kosmider L, Sobczak A, Prokopowicz A, et al. Cherry‐flavoured electronic cigarettes expose users to the inhalation irritant, benzaldehyde. Thorax 2016; 71: 376.
- 22. Gerhold KA, Bautista DM. TRPA1: irritant detector of the airways. J Physiol 2008; 586: 3303.
- 23. Valentine GW, DeVito EE, Jatlow PI, et al. Acute effects of inhaled menthol on the rewarding effects of intravenous nicotine in smokers. J Psychopharmacol 2018; 32: 986–994.
- 24. Abdel‐Gawad H, Hegazi B. Fate of 14C‐ethyl prothiofos insecticide in canola seeds and oils. J Environ Sci Health Part B 2010; 45: 116–122.
- 25. Kim KH, Jahan SA, Kabir E, Brown RJC. A review of airborne polycyclic aromatic hydrocarbons (PAHs) and their human health effects. Environ Int 2013; 60: 71–80.





Abstract
Objectives: To assess the chemical composition of electronic cigarette liquids (e‐liquids) sold in Australia, in both their fresh and aged forms.
Design, setting: Gas chromatography–mass spectrometry analysis of commercial e‐liquids sold in Australia (online and physical stores).
Main outcome measures: Chemical composition of 65 Australian e‐liquids — excipients/solvents, flavouring chemicals, other known e‐liquid constituents (including nicotine), and polycyclic aromatic hydrocarbons — before and after an accelerated ageing process that simulated the effects of vaping.
Results: The measured levels of propylene glycol and glycerol often diverged from those recorded on the e‐liquid label. All e‐liquids contained one or more potentially harmful chemicals, including benzaldehyde, menthol, trans‐cinnamaldehyde, and polycyclic aromatic hydrocarbons. Nicotine or nicotyrine were detected in a small proportion of e‐liquids at extremely low concentrations.
Conclusions: Australian e‐liquids contain a wide variety of chemicals for which information on inhalation toxicity is not available. Further analyses are required to assess the potential long term effects of e‐cigarette use on health.