The known The burden of cervical cancer is greater for Indigenous than for non-Indigenous Australian women. Participation in cervical screening is significantly lower and the prevalence of cervical abnormalities significantly higher among Indigenous than non-Indigenous women in Queensland.
The new Indigenous women are less likely to undergo clinical investigation within the recommended 2 months of a high grade abnormal Pap smear result, but by 6 months the follow-up rate was similar for Indigenous and non-Indigenous women.
The implications Improving timeliness of follow-up and increasing participation in cervical cancer prevention is important for reducing the higher burden of cervical cancer among Indigenous women.
For most Australian women, efforts to control cervical cancer have been successful; its incidence and mortality have each decreased by more than 50% since the inception of the National Cervical Screening Program (NCSP) in 1991.1 The NCSP currently recommends that women aged 18–69 years who have ever been sexually active have a Pap smear every two years, to detect and treat cervical cell abnormalities before they progress to cervical cancer.1 Current guidelines recommend clinical investigation by a specialist within 2 months of a high grade abnormal (HGA) Pap smear report,2 as in some women these abnormalities progress to cervical cancer.3 Investigation typically involves examining the cervix by colposcopy, and collecting a biopsy sample from the affected area for histological confirmation of the diagnosis.2
Among Aboriginal and Torres Strait Islander (hereafter referred to respectfully as Indigenous) women, the incidence of cervical cancer is twice and mortality four times as high as for non-Indigenous Australian women.1,4 Recent declines in the incidence of cervical cancer among Indigenous women in the Northern Territory5 and nationally4 indicate that efforts to prevent cervical cancer in Indigenous women have been partially effective. However, in the absence of routine screening data for Indigenous women, how this reduction has been achieved and what problems remain cannot be assessed.1,6
We recently reported that participation by Indigenous women in cervical screening was 20 percentage points lower than for non-Indigenous women in Queensland, and the prevalence of HGA results was twice as high.7,8 In this article we report our investigation of the adequacy of clinical follow-up for Indigenous and non-Indigenous women after a HGA Pap smear report.
Methods
We conducted a record linkage study of data from the Queensland Pap Smear Register (PSR), the Queensland Hospital Admitted Patient Data Collection (QHAPDC), and the Queensland Cancer Registry (QCR). All three datasets record Indigenous status for patients, but the PSR Indigenous status data are unreliable and incomplete. The data extraction and record linkage process have been described elsewhere.9 Briefly, the PSR was used to identify Queensland resident women aged 20–69 years who had a Pap smear between 8 February 1999 (the start of the PSR) and 31 December 2011. We linked the PSR data with hospital inpatient records of women aged 20–69 years who had ever been identified as Indigenous during a hospital admission between 1995 and 2011. The accuracy of Indigenous status in the QHAPDC is high (87%; 95% confidence interval [CI], 84–91%).10 Women were classified as Indigenous if they were linked to at least one QHAPDC record and if at least 50% of their records recorded them as being Indigenous.9,11 All other women were classified as non-Indigenous. The linked analysis dataset contained date of birth, residence (suburb, postcode), test date, test type (cytology or histology) and test result from the PSR; Indigenous status from the QHAPDC; and, where relevant, cervical cancer diagnosis date from the QCR. Women were excluded if their address details were insufficient to determine Queensland residency. There were no missing data for the variables included in this analysis.
Study population
Data from the Queensland PSR for women aged 20–68 years, with a Queensland address, and with a first HGA Pap smear (“index smear”) between 1 January 2000 and 31 December 2009 were analysed; clinical follow-up information to 31 December 2010 (biopsy histology result or cancer diagnosis date) was available. Women aged 69 years at the time of the Pap smear were excluded to ensure that 12 months’ follow-up was available. HGA was defined as a Pap smear result coded as possible or definitive cervical intra-epithelial neoplasia grade 2 or worse, adenocarcinoma in situ, or invasive cancer (based on the national cytology coding system).2 Clinical investigation was defined as a histology test or cancer diagnosis (data to 31 December 2010) following the index smear. Women who had a clinical investigation on the day of the index smear or earlier were excluded because the investigation was likely to have been related to gynaecological symptoms.
Geographical areas
Location of residence at the time of the index smear was mapped to 2011 Statistical Local Area (SLA) boundaries. SLAs were grouped according to geographic remoteness, based on the Accessibility/Remoteness Index of Australia (ARIA+).12 Area-level disadvantage was classified (in quintiles) using the Index of Relative Socio-Economic Advantage and Disadvantage (IRSAD).13
Outcome measures
The main outcome measure was time to clinical investigation following a HGA Pap smear result, calculated as days from the index smear to the first recorded clinical investigation (histology test or cancer diagnosis date). The PSR does not record colposcopies, but colposcopists are required to perform a biopsy in more than 95% of women with high grade cytological abnormalities who are not pregnant.14 Follow-up time was censored at 12 months from the index smear date. Since the PSR does not systematically include information about deaths or interstate migration, we assumed ongoing residence in Queensland for 12 months after the index smear.
Statistical analyses
All analyses were conducted in Stata 14.0 (StataCorp). Medians were reported for non-normally distributed variables and compared in Mann–Whitney tests. Trends in ordinal variables were assessed with χ2 tests in a bivariate logistic regression model. The Kaplan–Meier method was used to estimate the failure function, 1 – S(t), or the probability that the event (clinical investigation) has occurred by time t (expressed in months after the index smear). We report this probability separately for Indigenous and non-Indigenous women. Factors associated with time to investigation were analysed by Cox proportional hazards regression; the model included terms for remoteness of residence, area-level disadvantage, age group (in 5-year bands), and Indigenous status. Interactions between Indigenous status and covariates, and between time since index smear and covariates, were assessed. There was a significant interaction between Indigenous status and time to clinical investigation, which indicated a violation of the proportional hazard assumption; interval-specific follow-up rates for 2-monthly intervals (expressed per person–month) and interval-specific hazard ratios (reported as incidence rate ratios [IRRs]) were therefore calculated. Two-month intervals were chosen to reflect the current recommendation that clinical investigation be undertaken within 2 months of the HGA Pap smear, and to provide consistent time intervals over the full 12-month follow-up period. We assessed temporal trends of clinical investigation during the first 2 months for Indigenous and non-Indigenous women with the margins and lincom commands in Stata. P < 0.05 was deemed statistically significant.
Ethics approval
The study was approved by the Human Research Ethics Committees of Queensland Health (reference, HREC/15/QCH/19-957), the Northern Territory Department of Health and Menzies School of Health Research (reference, HOMER-2012-1737), and Charles Darwin University (reference, H12093). Data access and linkage were approved by the Director-General of Queensland Health, data custodians, and the Queensland Research Linkage Group.
Results
The Queensland PSR contained records for 1 219 034 women aged 20–69 years and resident in Queensland who had a Pap smear during 2000–2009. After excluding 215 women (the index smear was taken on the same date as a cancer diagnosis or histology, was preceded by cancer, or was taken at age 69), the study cohort consisted of 34 980 women with HGA Pap smear results, 1592 (4.6%) of whom were classified as Indigenous after linkage to hospital records. A total of 790 women with HGA Pap smear results had a record of invasive cervical cancer recorded on the QCR (of a total of 1352 cancer diagnoses recorded on the QCR for 2000–2009). Indigenous women were younger than non-Indigenous women at their index smear (median age, 28 years; interquartile range [IQR], 23–36 years v 31 years; IQR, 25–40 years; P < 0.001) and more likely to live in less affluent and more remote areas (Box 1).
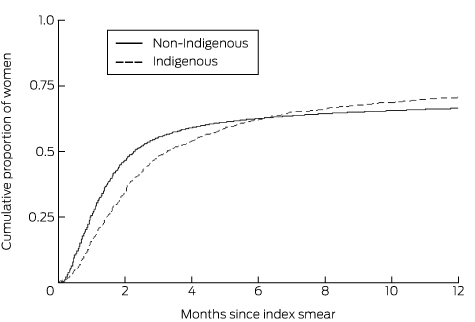
A greater proportion of Indigenous than of non-Indigenous women had a recorded clinical investigation within 12 months of a HGA Pap smear result (70.8% v 66.6%; P < 0.001). However, median follow-up time was longer for Indigenous than for non-Indigenous women (100 days; 95% CI, 91–112 days v 69 days; 95% CI, 68–70 days). The cumulative proportion of women who had a clinical investigation within 6 months of the index smear was still slightly lower for Indigenous than for non-Indigenous women, but was higher for Indigenous women thereafter (Box 2, Box 3).
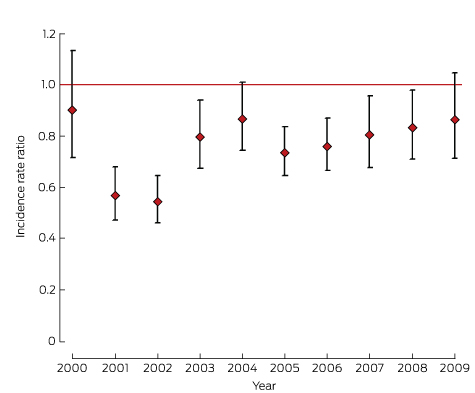
The interval-specific incidence rate of clinical investigation was also lower for Indigenous than for non-Indigenous women (incidence rate ratio [IRR] < 1.0) for the first 2 months after the index smear, but was higher thereafter (Box 4). After adjusting for area-level disadvantage, place of residence and age group, and including interactions that changed during the follow-up time, Indigenous women remained less likely than non-Indigenous women to have a clinical investigation during the first 2 months (adjusted IRR, 0.74; 95% CI, 0.68–0.81; Box 4). The adjusted IRR was lower for all years (2000–2009), with slight but not statistically significant improvement over time (Box 5).
There were 34 764 women with a possible or definitive HGA finding or cancer in situ recorded on their Pap smear result (excluding women with invasive cancer recorded in their report); an invasive lesion was recorded at clinical investigation for 674 of these women (1.9%).
Discussion
During 2000–2009, Indigenous women were less likely than non-Indigenous Australian women to have received a clinical investigation within the recommended 2-month interval after a HGA Pap smear result. However, the follow-up rate by 6 months was similar for both groups, and was subsequently higher for Indigenous women; by 12 months, a greater proportion of Indigenous women had had a clinical investigation than non-Indigenous women. The available data do not indicate why 30% of all women did not have a follow-up clinical investigation. It is possible that some had a repeat Pap smear, which would not have been counted as a clinical investigation, or that a biopsy was not collected during the colposcopy visit (but this would be unusual).
A successful cervical cancer prevention program depends on the adequate assessment, treatment and follow-up of women with cervical cell abnormalities.15 The NCSP has been unable to report on program indicators for Indigenous women, and much has consequently remained unknown about how Indigenous women use cervical screening services. Our findings show that most women with HGA results are eventually followed up, but that a lower proportion of Indigenous women have a clinical investigation within the 2-month period recommended by NHMRC guidelines.2 Significant delays in clinical investigation could result in HGAs progressing to cervical cancer.
Possible reasons for the delay in follow-up of Indigenous women may include factors related to health systems, health practitioners, and individuals. From the health system perspective, the cost, accessibility and cultural appropriateness of services will influence a woman’s decisions about attending diagnostic follow-up of an abnormal cytology result. Although Medicare provides a rebate for private outpatient services, an additional payment by the patient is usually required.16
Indigenous Australians are more likely to use the public hospital system than other Australians (90% v 57% of hospital separations are from public hospitals),17 where waiting times for dysplasia clinics can be several months. In Queensland, more than half of the Indigenous population live in outer regional, remote or very remote areas. While access to services in these areas is limited and long distance travel can impede timely follow-up,18 we found that adjusting for remoteness and area disadvantage did not remove the difference between Indigenous and non-Indigenous women in this regard.
Mainstream health care settings can be culturally uninviting. Combined with a history of marginalisation, this can create feelings of alienation, isolation, fear and mistrust of the health care system and health practitioners.19 Ineffective communication by health practitioners can compound these problems.19 Cultural views of cancer may also cause delays in seeking diagnostic or curative treatment; from an Indigenous perspective, cancer could be seen as retribution for a past wrong, and mainstream cancer treatment may be seen as loss of traditional lifestyle.19,20
Our findings are similar to those reported by BreastScreen Australia, which found that Indigenous women were less likely to attend post-screening assessment within the recommended period after mammography.21 In New Zealand, the proportion of women receiving follow-up after HGA Pap test results was lower for Māori than for European and other women at 90 days (81.0% v 83.6%) and 180 days (87.5% v 89.2%).22
The strengths of our study include near complete population-based data for cervical screening and clinical investigation from the Queensland PSR (including a 12-month follow-up period), and a reliable data source (QHAPDC) for identifying Indigenous women in the PSR. We have previously reported in detail possible reasons why our record linkage methodology may not have identified all Indigenous women.9 Briefly, accuracy of Indigenous status in Queensland hospital records is not perfect (it was estimated to be 87% in 2011, and to be higher in regional and remote areas than in metropolitan areas), but has improved.10 It is possible that a public hospital record was not available for linkage for all Indigenous women, but this is probably uncommon.9
Our follow-up classification relied on a histology test record or cancer diagnosis date. Women who had a colposcopy without a biopsy would therefore be incorrectly classified as not having had any follow-up. It is not possible to quantify the number of Indigenous or non-Indigenous women in this category because of the lack of comprehensive colposcopy data in the current PSR. However, we expect this number to be small, given that the NHMRC guidelines2 and the Colposcopy Quality Improvement Program14 recommend that a biopsy should be undertaken except in certain circumstances (eg, if a woman is pregnant). Further, we calculated the time to clinical investigation using the pathology report date, which may introduce error if a delay occurs between colposcopy and the pathology assessment.
We are unable to report on the number of Queensland residents who were followed up interstate (eg, in border centres such as Tweed Heads), so that we may have slightly underestimated the number of women investigated. This may partially explain why a lower proportion of women in Queensland were followed up than in Victoria, where it was reported in 2013 that 89% of women with a HGA on one or more of their Pap test reports were followed up within 6–9 months (by colposcopy or biopsy).23 Pre-2006 guidelines recommended that women return for annual cytological testing following a HGA finding, to confirm the absence of abnormalities; for a very small number of women included in our study, index Pap smears prior to 2006 may have been for test-of-cure, and it is uncertain whether they would be more or less compliant with follow-up than women receiving an initial HGA result. As we cannot determine which women were in this category, we could not adjust our analysis accordingly. Including data for these women may have caused us to either under- or overestimate the time to follow-up for both Indigenous and non-Indigenous women.
Our findings describe the follow-up of screened women in Queensland; similar analyses in other states are warranted, given the potential importance of this clinical and health service delivery information.
Conclusions
Major changes to the Australian NCSP will be introduced from May 2017, shifting from a Pap test every 2 years to a human papillomavirus (HPV) test every 5 years for women aged 25–74 years.24 In preparation for this new program, new draft clinical management guidelines have been published, and the pertinent screening result in the renewed program will no longer be HGA cytology, but a positive result for HPV16/18 or other high risk HPV type, together with a positive cytology triage result. Our findings will continue to be relevant for assessing the time to clinical investigation for Indigenous women, even though the screening process (and trigger for colposcopy referral) will change.25
Our finding that Indigenous women with HGA findings are less likely to receive clinical follow-up within the recommended 2-month interval is concerning. Reasons for the initial delay need to be identified and solutions developed. While our findings about clinical investigation are important, increasing participation in cervical cancer prevention programs, both screening and HPV vaccination, remain critical for closing the gap in cervical cancer rates between Indigenous and non-Indigenous women.
Box 1 – Demographic characteristics of 34 980 Queensland resident women aged 20–68 years at the time of their first high grade abnormal (HGA) Pap smear result, 2000–2009
Characteristics |
Indigenous |
Non-Indigenous |
P* |
||||||||||||
Number of women (proportion of all women with HGA result) |
1592 (4.6%) |
33 388 (95.5%) |
|||||||||||||
Age group (years) |
< 0.001 |
||||||||||||||
20–24 |
31.1% |
21.1% |
|||||||||||||
25–29 |
24.8% |
22.7% |
|||||||||||||
30–34 |
15.9% |
18.1% |
|||||||||||||
35–39 |
11.5% |
12.2% |
|||||||||||||
40–44 |
7.2% |
8.7% |
|||||||||||||
45–49 |
4.2% |
6.4% |
|||||||||||||
50–54 |
2.3% |
4.5% |
|||||||||||||
55–59 |
1.4% |
3.1% |
|||||||||||||
60–64 |
0.9% |
2.0% |
|||||||||||||
65–68 |
0.9% |
1.2% |
|||||||||||||
Area-level disadvantage (quintiles) |
< 0.001 |
||||||||||||||
1 (most disadvantaged) |
28.5% |
11.4% |
|||||||||||||
2 |
34.7% |
22.7% |
|||||||||||||
3 |
18.0% |
23.1% |
|||||||||||||
4 |
15.0% |
25.1% |
|||||||||||||
5 (least disadvantaged) |
3.8% |
17.8% |
|||||||||||||
Place of residence |
< 0.001 |
||||||||||||||
Major city |
22.6% |
60.5% |
|||||||||||||
Inner regional |
14.6% |
18.9% |
|||||||||||||
Outer regional |
42.0% |
18.4% |
|||||||||||||
Remote |
9.4% |
1.4% |
|||||||||||||
Very remote |
11.4% |
0.8% |
|||||||||||||
* χ2 test for ordinal trend in bivariate logistic regression model. | |||||||||||||||
Box 2 – Queensland resident women aged 20–68 years having a clinical investigation after a high grade abnormal Pap smear result, 2000–2009, by 2-month interval
|
Indigenous women |
Non-Indigenous women |
||||||||||||||
Women with clinical investigation |
Cumulative proportion (95% CI) |
Women with clinical investigation |
Cumulative proportion (95% CI) |
||||||||||||
Total number of women |
1592 |
33 388 |
|||||||||||||
2-month interval |
|||||||||||||||
0 to < 2 months |
542 |
34.1% (31.8–36.4%) |
15 519 |
46.5% (46.0–47.0%) |
|||||||||||
2 to < 4 months |
304 |
54.0% (51.5–56.4%) |
4223 |
59.2% (58.7–59.7%) |
|||||||||||
4 to < 6 months |
143 |
62.2% (59.8–64.6%) |
1170 |
62.8% (62.2–63.3%) |
|||||||||||
6 to < 8 months |
64 |
66.1% (63.8–68.5%) |
617 |
64.5% (64.0–65.1%) |
|||||||||||
8 to < 10 months |
43 |
68.9% (66.6–71.2%) |
380 |
65.7% (65.2–66.2%) |
|||||||||||
10 to < 12 months |
31 |
70.7% (68.5–73.0%) |
336 |
66.6% (66.1–67.1%) |
|||||||||||
Box 3 – Cumulative proportion of Queensland resident women aged 20–68 years having a clinical investigation after a high grade abnormal Pap smear result, 2000–2009
Box 4 – Interval-specific rates (per person–month) and incidence rate ratios for eligible Queensland residents aged 20–68 years* investigated after a high grade abnormal Pap smear result, 2000–2009
Time since index smear |
Interval-specific rate (with 95% CI) |
Incidence rate ratio, Indigenous v non-Indigenous women (with 95% CI) |
|||||||||||||
Indigenous |
Non-Indigenous |
Unadjusted |
Adjusted† |
||||||||||||
0–2 months |
0.20 (0.19–0.22) |
0.31 (0.30–0.31) |
0.65 (0.60–0.71) |
0.74 (0.68–0.81) |
|||||||||||
2–4 months |
0.19 (0.17–0.21) |
0.14 (0.14–0.14) |
1.32 (1.18–1.48) |
1.21 (1.08–1.36) |
|||||||||||
4–6 months |
0.10 (0.08–0.12) |
0.05 (0.04–0.05) |
2.16 (1.80–2.59) |
1.74 (1.45–2.09) |
|||||||||||
6–8 months |
0.06 (0.04–0.07) |
0.02 (0.02–0.03) |
2.28 (1.75–2.95) |
1.70 (1.31–2.20) |
|||||||||||
8–10 months |
0.04 (0.03–0.06) |
0.02 (0.02–0.02) |
2.53 (1.86–3.46) |
1.79 (1.30–2.45) |
|||||||||||
10–12 months |
0.03 (0.02–0.04) |
0.01 (0.01–0.02) |
2.64 (2.22–3.13) |
1.75 (1.46–2.09) |
|||||||||||
* Women who did not have a clinical investigation in a preceding interval. † Adjusted for age group, place of residence, and area-level disadvantage. | |||||||||||||||
Received 5 March 2016, accepted 22 August 2016
- Lisa J Whop1
- Peter D Baade2
- Julia ML Brotherton3
- Karen Canfell4
- Joan Cunningham1
- Dorota Gertig5
- Kamalini Lokuge6
- Gail Garvey1
- Suzanne P Moore1
- Abbey Diaz1
- Dianne L O'Connell4
- Patricia Valery7
- David M Roder8
- John R Condon1
- 1 Menzies School of Health Research, Charles Darwin University, Darwin, NT
- 2 Cancer Research Centre, Cancer Council Queensland, Brisbane, QLD
- 3 National HPV Vaccination Program Register, Victorian Cytology Service, Melbourne, VIC
- 4 Cancer Research Division, Cancer Council NSW, Sydney, NSW
- 5 Melbourne School of Population and Global Health, University of Melbourne, Melbourne, VIC
- 6 ANU College of Medicine, Biology and Environment, Australian National University, Canberra, ACT
- 7 QIMR Berghofer Medical Research Institute, Brisbane, QLD
- 8 Centre for Population Health Research, University of South Australia, Adelaide, SA
The National Indigenous Cervical Screening Project is funded by a National Health and Medical Research Council (NHMRC) Project Grant (104559). This project is part of the NHMRC Centre of Research Excellence in Discovering Indigenous Strategies to Improve Cancer Outcomes via Engagement, Research Translation and Training (DISCOVER-TT CRE; 1041111) and the Cancer Council NSW Strategic Research Partnership to Improve Cancer Control for Indigenous Australians (STREP Ca-CIndA; SRP13-01). We also acknowledge the ongoing support of the Lowitja Institute, Australia’s national institute for Aboriginal and Torres Strait Islander health research. Lisa Whop was supported by a Sidney Myer Health scholarship, a Menzies Enhanced Living scholarship, and a Lowitja Institute scholarship. Abbey Diaz was supported by an NHMRC postgraduate scholarship (1055587) and a DISCOVER-TT CRE-funded Menzies Enhanced Living scholarship. The NHMRC supported Joan Cunningham with a research fellowship (1058244), Patricia Valery with a career development fellowship (1083090), and Karen Canfell with a career development award (1082989). We acknowledge the staff and registrars from the Queensland Health Pap Smear Register, the Queensland Cancer Registry, and the Queensland Health Admitted Patient Data Collection for their assistance in providing the data, and the Queensland Research Linkage Group for linking the data. We gratefully acknowledge Tegan Harris for a range of contributions, including designing and building the database. The views expressed in this publication are those of the authors and do not necessarily reflect the views of the funding agencies.
Karen Canfell, Dorota Gertig and Julia Brotherton are investigators in a trial of cytology and primary human papilloma virus (HPV) screening in Australia (“Compass”), which is conducted and funded by the Victorian Cytology Service (VCS), a government-funded health promotion charity, and by Karen Canfell’s research group (formerly University of New South Wales, now at Cancer Council New South Wales). The VCS received equipment and a funding contribution for the Compass pilot study and for the main trial from Roche Molecular Systems and Ventana Inc. USA.
- 1. Australian Institute of Health and Welfare. Cervical screening in Australia 2012–2013 (AIHW Cat. No. CAN 91; Cancer Series No. 93). Canberra: AIHW, 2015. http://www.aihw.gov.au/publication-detail/?id=60129550871 (accessed Sept 2016).
- 2. National Health and Medical Research Council. Screening to prevent cervical cancer: guidelines for the management of asymptomatic women with screen detected abnormalities. Canberra: NHMRC, 2005. https://www.nhmrc.gov.au/guidelines-publications/wh39 (accessed Sept 2016).
- 3. McCredie MR, Paul C, Sharples KJ, et al. Consequences in women of participating in a study of the natural history of cervical intraepithelial neoplasia 3. Aust N Z J Obstet Gynaecol 2010; 50: 363-370.
- 4. Zhang X, Condon JR, Rumbold AR, et al. Estimating cancer incidence in Indigenous Australians. Aust N Z J Public Health 2011; 35: 477-485.
- 5. Zhang X CJ, Dempsey K, Garling L. Cancer in the Northern Territory 1991–2010: incidence, mortality and survival. Darwin: Department of Health, 2014. http://digitallibrary.health.nt.gov.au/prodjspui/handle/10137/617 (accessed Sept 2016).
- 6. Australian Institute of Health and Welfare. The inclusion of Indigenous status on pathology request forms (AIHW Cat. No. IHW 103). Canberra: AIHW, 2013. http://www.aihw.gov.au/publication-detail/?id=60129544909 (accessed Sept 2016).
- 7. Whop LJ, Baade P, Garvey G, et al. Cervical abnormalities are more common among indigenous than other Australian women: a retrospective record-linkage study, 2000–2011. PLoS One 2016; 11: e0150473.
- 8. Whop LJ, Garvey G, Baade P, et al. The first comprehensive report on Indigenous Australian women’s inequalities in cervical screening: a retrospective registry cohort study in Queensland, Australia (2000–2011). Cancer 2016; 122: 1560-1569.
- 9. Whop LJ, Diaz A, Baade P, et al. Using probabilistic record linkage methods to identify Australian Indigenous women on the Queensland Pap Smear Register: the National Indigenous Cervical Screening Project. BMJ Open 2016; 6: e009540.
- 10. Australian Institute of Health and Welfare. Indigenous identification in hospital separations data: quality report (AIHW Cat. No. IHW 90). Canberra: AIHW, 2013. http://www.aihw.gov.au/publication-detail/?id=60129543215 (accessed Sept 2016).
- 11. Australian Institute of Health and Welfare and Australian Bureau of Statistics. National best practice guidelines for data linkage activities relating to Aboriginal and Torres Strait Islander people (AIHW Cat. No. IHW 74). Canberra: AIHW, 2012. http://www.aihw.gov.au/publication-detail/?id=10737422216 (accessed Sept 2016).
- 12. Australian Institute of Health and Welfare. Rural, regional and remote health: a guide to remoteness classifications (AIHW Cat. No. PHE 53). Canberra: AIHW, 2004. http://www.aihw.gov.au/publication-detail/?id=6442467589 (accessed Sept 2016).
- 13. Australian Bureau of Statistics. Socio-Economic Indexes for Areas (SEIFA) 2011: Technical Paper (ABS Cat. No. 2033.0.55.001). Canberra: ABS, 2011. http://www.ausstats.abs.gov.au/ausstats/subscriber.nsf/0/22CEDA8038AF7A0DCA257B3B00116E34/$File/2033.0.55.001%20seifa%202011%20technical%20paper.pdf (accessed Sept 2016).
- 14. The Colposcopy Quality Improvement Program (C-QuIP): Quality program standards for audit. Diagnostic colposcopists. Updated Aug 2015. http://www.cquip.edu.au/component/docman/doc_download/38-standards-in-diagnostic-colposcopy-.html (accessed Feb 2016).
- 15. Kavanagh AM, Simpson JM. Predicting nonattendance for colposcopy clinic follow-up after referral for an abnormal Pap smear. Aust N Z J Public Health 1996; 20: 266-271.
- 16. Australian Government Department of Health. Medicare Benefits Schedule book. Canberra: Department of Health, 2015. http://www.health.gov.au/internet/mbsonline/publishing.nsf/Content/432EE55FAB58E5C4CA257D6B001AFB8A/$File/201411-MBS.pdf (accessed Feb 2016).
- 17. Australian Institute of Health and Welfare. Admitted patient care 2013–14: Australian hospital statistics (AIHW Cat. No. HSE 156; Health Services Series No. 60). Canberra: AIHW, 2015. http://www.aihw.gov.au/publication-detail/?id=60129550483 (accessed Feb 2016).
- 18. Shahid S, Finn L, Bessarab D, Thompson SC. “Nowhere to room … nobody told them”: logistical and cultural impediments to Aboriginal peoples’ participation in cancer treatment. Aust Health Rev 2011; 35: 235-241.
- 19. Shahid S, Finn LD, Thompson SC. Barriers to participation of Aboriginal people in cancer care: communication in the hospital setting. Med J Aust 2009; 190: 574-579. <MJA full text>
- 20. Shahid S, Finn L, Bessarab D, et al. Understanding, beliefs and perspectives of Aboriginal people in Western Australia about cancer and its impact on access to cancer services. BMC Health Serv Res 2009; 9: 132.
- 21. Roder D, Webster F, Zorbas H, et al. Breast screening and breast cancer survival in Aboriginal and Torres Strait Islander women of Australia. Asian Pac J Cancer Prev 2012; 13: 147-155.
- 22. Smith M, Walker R, Canfell K. National Cervical Screening Programme. Monitoring report number 40: 1 July – 31 December 2013. National Screening Unit, 2014. https://www.nsu.govt.nz/system/files/page/monitoringreport40_final.pdf (accessed Feb 2016).
- 23. Victorian Cervical Cytology Registry. Statistical report 2013. Melbourne: VCCR, 2013. http://www.vccr.org/site/VCCR/filesystem/documents/dataandresearch/StatisticalReports/VCS_StatisticsReport_2013_Web_SinglePages_Final.pdf (accessed Feb 2016).
- 24. Australian Government Department of Health. National Cervical Screening Program: overview of the renewal [website]. Updated May 2015. http://www.cancerscreening.gov.au/internet/screening/publishing.nsf/Content/overview-of-the-renewal (accessed Feb 2016).
- 25. Armstrong B, Brand A, Canfell K, et al; Cancer Council Australia Cervical Cancer Screening Guidelines Working Party. Oncogenic HPV types 16 and/or 18. In: National Cervical Screening Program: guidelines for the management of screen-detected abnormalities, screening in specific populations and investigation of abnormal vaginal bleeding [website]. Updated Aug 2016. http://wiki.cancer.org.au/australia/Clinical_question:Oncogenic_HPV_types_16_and_or_18 (accessed Aug 2016).





Abstract
Objectives: To investigate time to follow-up (clinical investigation) for Indigenous and non-Indigenous women in Queensland after a high grade abnormality (HGA) being detected by Pap smear.
Design, setting, participants: Population-based retrospective cohort analysis of linked data from the Queensland Pap Smear Register (PSR), the Queensland Hospital Admitted Patient Data Collection, and the Queensland Cancer Registry. 34 980 women aged 20–68 years (including 1592 Indigenous women) with their first HGA Pap smear result recorded on the PSR (index smear) during 2000–2009 were included and followed to the end of 2010.
Main outcome measures: Time from the index smear to clinical investigation (histology test or cancer diagnosis date), censored at 12 months.
Results: The proportion of women who had a clinical investigation within 2 months of a HGA finding was lower for Indigenous (34.1%; 95% CI, 31.8–36.4%) than for non-Indigenous women (46.5%; 95% CI, 46.0–47.0%; unadjusted incidence rate ratio [IRR], 0.65; 95% CI, 0.60–0.71). This difference remained after adjusting for place of residence, area-level disadvantage, and age group (adjusted IRR, 0.74; 95% CI, 0.68–0.81). However, Indigenous women who had not been followed up within 2 months were subsequently more likely to have a clinical investigation than non-Indigenous women (adjusted IRR for 2–4 month interval, 1.21; 95% CI, 1.08–1.36); by 6 months, a similar proportion of Indigenous (62.2%; 95% CI, 59.8–64.6%) and non-Indigenous women (62.8%; 95% CI, 62.2–63.3%) had been followed up.
Conclusions: Prompt follow-up after a HGA Pap smear finding needs to improve for Indigenous women. Nevertheless, slow follow-up is a smaller contributor to their higher cervical cancer incidence and mortality than their lower participation in cervical screening.