Alcohol consumption during pregnancy may contribute to birth defects, growth and developmental abnormalities, and fetal mortality.1-3 In 2009, the National Health and Medical Research Council (NHMRC) revised its guidelines, recommending that the safest option for pregnant women, and women planning a pregnancy, was to not consume alcohol at all.2
Although Australia conducts routine National Drug Strategy Household Surveys, changes made in 2010 to the survey questions about alcohol use during pregnancy — to incorporate drinking before and after knowledge of pregnancy — preclude direct comparisons over time.4 Thus, we aimed to describe the prevalence and distribution of alcohol use during pregnancy in an Australian population over the 5-year period 2007 to 2011.
We undertook a cross-sectional repeated sample, trend analysis of alcohol consumption patterns during pregnancy. Data were obtained from baseline surveys of pregnant women participating in the Griffith Study of Population Health: Environments for Healthy Living (EFHL study) from 2007 to 2011. This longitudinal birth cohort study annually recruits participants from public maternity hospitals in the Logan–Beaudesert, Gold Coast and Tweed health districts in south-east Queensland and north-east New South Wales.5
Participants completed a baseline, self-administered questionnaire, which included items from the 2004 National Drug Strategy Household Survey6 relating to alcohol consumption, modified for pregnancy. Images and information about different alcohol types and what constitutes a standard drink were provided.6 Participants were asked about alcohol consumption (any level) and high-risk alcohol consumption (five or more standard drinks on any one occasion) at early, mid and late pregnancy (0–13, 14–26 and 27–42 weeks, respectively). They were also asked about low-level alcohol use, which was defined as consuming between half a standard drink and two standard drinks on any occasion. Data on maternal age, education, marital status, income, smoking and recreational drug use were also collected.5
From 2007 to 2011, 2743 pregnant women were enrolled in the EFHL study (age range, 16–52 years). Alcohol consumption data were available for 2731 of them, and 1206 (44.2%) reported drinking alcohol at some time during pregnancy. In total, 917 women (33.7%) reported consuming alcohol after the first trimester of pregnancy, when they would have been aware of their pregnancy, and 68 women (2.5%) reported drinking at high-risk levels after the first trimester (Box 1).
The proportion of women consuming low levels of alcohol after the first trimester significantly increased with increasing age (P < 0.001), increasing levels of education (P = 0.01) and increasing household income (P < 0.001). Women who smoked cigarettes and used recreational drugs during pregnancy were more likely to consume alcohol (P < 0.001 for both) (Box 1).
High-risk drinking after the first trimester was reported by 3.1% of women younger than 25 years and 4.0% of those in the lowest household income quintile (Box 1). Also, high-risk drinking after the first trimester was associated with lower levels of education (P = 0.011) and single-parent status (P = 0.001), was 5.4 times more likely among women who smoked than among non-smokers, and was 7.5 times more likely among women who used recreational drugs than among non-drug users (Box 1).
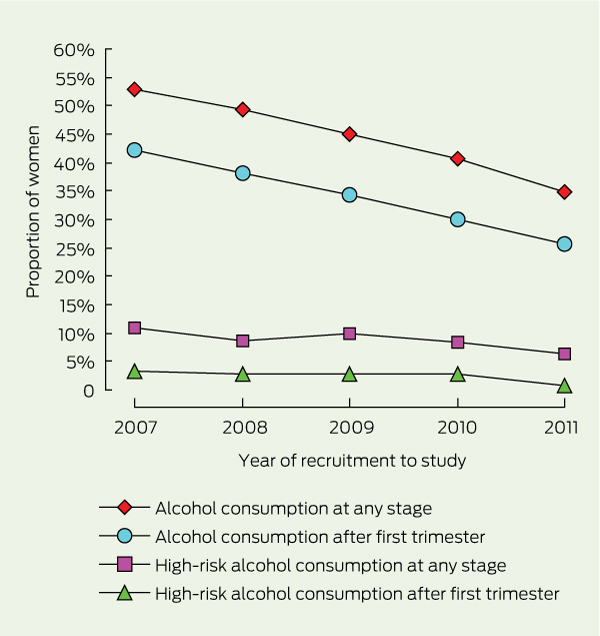
From 2007 to 2011, the proportion of women who reported drinking alcohol during pregnancy significantly decreased; 251 of 475 women (52.8%) reported alcohol use in 2007, compared with 168 of 483 (34.8%) in 2011 (P < 0.001) (Box 2). The proportion of women who drank alcohol after the first trimester of pregnancy declined from 200 of 474 women (42.2%) in 2007 to 124 of 481 (25.8%) in 2011 (Box 2). The proportion of women who consumed alcohol during every trimester of pregnancy almost halved from 2007 (99/474; 20.9%) to 2011 (53/481; 11.0%). However, for those who reported high-risk consumption — at all or after the first trimester — there was no statistically significant change over the 5 years (P = 0.12).
This study shows a steady and statistically significant decline in the proportion of women who reported drinking alcohol during pregnancy from 2007 to 2011. The proportion of women who continued to drink alcohol after their first trimester of pregnancy also declined. It is possible that increased media emphasis on the negative effects of alcohol during pregnancy7,8 may have increased underreporting of alcohol consumption during the study period, thus resulting in this overall decline. Although alcohol consumption in the general population also declined during the study period,9 this study showed no change in alcohol consumption after the first trimester for older women, single parents, low-income women, women with a trade or apprenticeship education, or women who used recreational drugs. Further, the proportion of women who reported high-risk drinking (five standard drinks or more on any one occasion) did not change over the 5-year period. This finding may have been related to the small sample size, but it is also possible that public health programs and national policies are not reaching women who are most at risk of having babies with alcohol-related birth defects.
Consistent with some other risk behaviour,10-12 there was a socioeconomic differential in drinking patterns during pregnancy: low-level alcohol consumption after the first trimester increased with increasing age, education and income, and high-risk consumption after the first trimester was more common in single women and women who did not complete school. It is particularly concerning that women who continued high-risk drinking after the first trimester were also much more likely to smoke cigarettes or use recreational drugs, which are also associated with adverse pregnancy outcomes.13,14
Given that the sociodemographic composition of the cohorts did not change across the 5 years of recruitment,15 the findings are not likely to be a result of random variation in participant sampling or demographic shifts in the study region. Nevertheless, it is possible that the study was biased slightly towards women with lower levels of alcohol consumption, because inclusion in the study was dependent on reaching the third trimester of pregnancy.2 Women who had experienced early fetal loss, which may have been associated with high levels of alcohol consumption, were not included.
Received 25 November 2012, accepted 30 May 2013
- Cate M Cameron1,2
- Tamzyn M Davey1
- Elizabeth Kendall1,2,3
- Andrew Wilson4
- Roderick J McClure5
- 1 Griffith Health Institute, Griffith University, Logan, QLD.
- 2 Population and Social Health Research Program, Griffith University, Logan, QLD.
- 3 Centre of National Research on Disability and Rehabilitation Medicine, Brisbane, QLD.
- 4 Menzies Centre for Health Policy, University of Sydney, Sydney, NSW.
- 5 Monash Injury Research Institute, Monash University, Melbourne, VIC.
This research is part of the EFHL study, which receives core funding from Griffith University and is also funded by an Australian Research Council Discovery Project grant (DP110105423). The EFHL study was conceived by Roderick McClure, Cate Cameron, Judy Searle and Ronan Lyons. We gratefully acknowledge the chief investigators, project, administrative and research staff, and hospital antenatal and birth suite midwives of the participating hospitals for helping to conduct the study. Cate Cameron was supported by a Public Health Fellowship (ID 428254) from the NHMRC.
No relevant disclosures.
- 1. Andersen AM, Andersen PK, Olsen J, et al. Moderate alcohol intake during pregnancy and risk of fetal death. Int J Epidemiol 2012; 41: 405-413.
- 2. National Health and Medical Research Council. Australian guidelines to reduce health risks from drinking alcohol. Canberra: NHMRC, 2009. http://www.nhmrc. gov.au/guidelines/publications/ds10 (accessed Jun 2013).
- 3. Burd L, Klug MG, Li Q, et al. Diagnosis of fetal alcohol spectrum disorders: a validity study of the fetal alcohol syndrome checklist. Alcohol 2010; 44: 605-614.
- 4. Callinan S, Room R. Alcohol consumption during pregnancy: results from the 2010 National Drug Strategy Household Survey. Canberra: Foundation for Alcohol Research and Education, 2012. http://www. health infonet.ecu.edu.au/key-resources/bibliography/?lid=23341 (accessed Jun 2013).
- 5. Cameron CM, Scuffham PA, Spinks A, et al. Environments for Healthy Living (EFHL) Griffith birth cohort study: background and methods. Matern Child Health J 2012; 16: 1896-1905.
- 6. Australian Institute of Health and Welfare. 2004 National Drug Strategy Household Survey: detailed findings. Canberra: AIHW, 2005. (AIHW Cat. No. PHE 66; Drug Statistics Series No. 16.) http://www.aihw.gov. au/publication-detail/?id=6442467781 (accessed Aug 2013).
- 7. Centre for Health Promotion. Pregnancy and alcohol don’t mix. Adelaide: Centre for Health Promotion, 2007. http://www.healthinfonet.ecu.edu.au/key-resources/promotion-resources?lid=19111 (accessed Aug 2013).
- 8. Queensland Health. Young women and alcohol campaign. Make up your own mind about drinking. http://www.health.qld.gov.au/atod/prevention/young_women.asp (accessed Mar 2013).
- 9. Australian Institute of Health and Welfare. 2010 National Drug Strategy Household Survey report. Canberra: AIHW, 2011. (AIHW Cat. No. PHE 145; Drug Statistics Series No. 25.) http://www.aihw.gov.au/publication-detail/?id=32212254712 (accessed Aug 2013).
- 10. Bergman MM, Scott J. Young adolescents’ wellbeing and health-risk behaviours: gender and socio-economic differences. J Adolesc 2001; 24: 183-197.
- 11. Barreto SM, Figueiredo RC. Chronic diseases, self-perceived health status and health risk behaviors: gender differences. Rev Saude Publica 2009; 43 Suppl 2: 38-47.
- 12. Tuinstra J, Groothoff JW, van den Heuvel WJ, Post D. Socio-economic differences in health risk behavior in adolescence: do they exist? Soc Sci Med 1998; 47: 67-74.
- 13. Hackshaw A, Rodeck C, Boniface S. Maternal smoking in pregnancy and birth defects: a systematic review based on 173 687 malformed cases and 11.7 million controls. Hum Reprod Update 2011; 17: 589-604.
- 14. Wong S, Ordean A, Kahan M; Society of Obstetricians and Gynecologists of Canada. SOGC clinical practice guidelines: substance use in pregnancy: no. 256, April 2011. Int J Gynaecol Obstet 2011; 114: 190-202.
- 15. Cameron CM, Scuffham PA, Shibl R, et al. Environments For Healthy Living (EFHL) Griffith birth cohort study: characteristics of sample and profile of antenatal exposures. BMC Public Health 2012; 12: 1080.





Abstract
Objective: To describe the prevalence and distribution of alcohol consumption during pregnancy in an Australian population over a 5-year period.
Design, setting and participants: Cross-sectional repeated sample, trend analysis of aggregated and stratified alcohol consumption patterns during pregnancy. Pregnant women were enrolled from 2007 to 2011 in the Griffith Study of Population Health: Environments for Healthy Living, a birth cohort study being conducted in south-east Queensland and north-east New South Wales.
Main outcome measures: Sociodemographic and alcohol consumption data were self-reported at enrolment. Alcohol measures included alcohol consumption (any level) and high-risk alcohol consumption, both during pregnancy (at any stage) and after the first trimester of pregnancy.
Results: Of 2731 pregnant women for whom alcohol consumption data were available, a decrease in alcohol consumption was observed over the study period; 52.8% reported alcohol use in 2007 compared with 34.8% in 2011 (P < 0.001). The proportion of women who drank alcohol after the first trimester of pregnancy declined from 42.2% in 2007 to 25.8% in 2011. However, high-risk drinking patterns — at all or after the first trimester — did not change over the 5 years (P = 0.12). Low-level alcohol consumption was associated with older women (P < 0.001), more highly educated women (P = 0.01), and women from higher-income households (P < 0.001). In contrast, high-risk consumption after the first trimester was associated with lower levels of education (P = 0.011) and single-parent status (P = 0.001).
Conclusions: This study showed a steady and statistically significant decline in the proportion of women who reported drinking alcohol during pregnancy from 2007 to 2011. To further reduce these levels, we need broad public health messages for the general population and localised strategies for high-risk subpopulations.
Trial registration: Australian New Zealand Clinical Trials Registry ACTRN12610000931077.