Early detection of diabetic retinopathy (DR) and timely treatment are integral to preventing vision-threatening DR in people with diabetes.1,2 The National Health and Medical Research Council (NHMRC) guidelines for the management of DR recommend biannual screening in those with no previous diagnosis and annual screening (at least) for those who have had a diagnosis of DR.3 However, population-based studies show that 30%–50% of Australians with diabetes do not undergo adequate screening.4-6 Despite the implementation of successful national screening programs worldwide,7,8 comprehensive DR screening strategies are lacking in Australia. We propose a novel screening strategy using pathology collection centres (PCCs) as screening sites. A PCC offers the potential to opportunistically screen people with diabetes for DR, as up to 90% of people with diabetes regularly attend PCCs for diabetes-related tests.9
Patients’ sociodemographic details were collected, including diabetes type, duration of diabetes and reasons for not having biannual DR screening. Presenting distance binocular visual acuity was assessed with an eye chart. In a darkened room, two 45° non-mydriatic photographs were taken for each eye, centred on the optic disc and macula.10 Images were sent to the Centre for Eye Research Australia for assessment. Patients were asked to grade their satisfaction with the photographic procedure, on a 5-point Likert scale, from “very satisfied” to “very dissatisfied”, and the degree to which our screening service would give them more control over the management of their diabetic eye care, from “not at all” to “a considerable degree”. Questionnaire items were based on published literature.11-13 Images were graded by trained graders and validated by an ophthalmologist. The presence of DR was defined with the Early Treatment Diabetic Retinopathy Study severity scale10 and classified as none, minimal non-proliferative DR (NPDR), mild NPDR, moderate NPDR, severe NPDR, proliferative DR (PDR) and high-risk PDR.14 Follow-up recommendations were made in accordance with the NHMRC guidelines.3
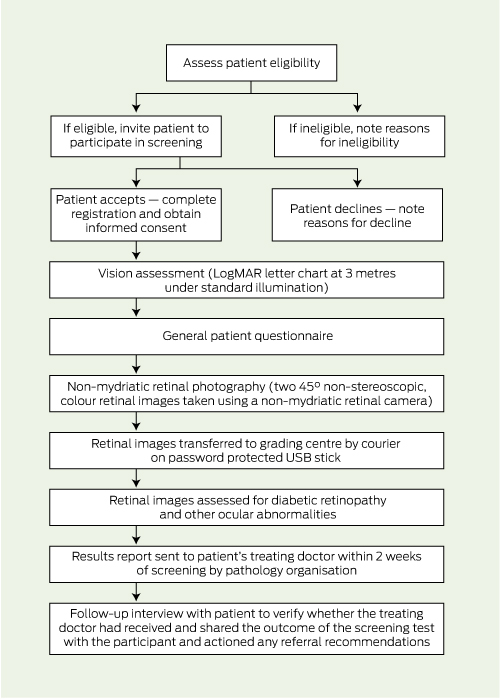
A report outlining the results of the screening and follow-up recommendations was sent electronically to the patient’s treating doctor by the pathology organisation within 2 weeks of screening. Two weeks after the distribution of reports, we contacted 50% of patients at random for a follow-up telephone interview to verify whether the treating doctor had received and shared the outcome of the screening test with the patient and provided any recommended referrals. Box 1 shows a flow chart of the testing protocol.
We were able to obtain two gradable images for 138 (74.2%) of the 186 eyes examined (ie, well centred images with good focus and illumination). The interpretation of images of 41 eyes (22.0%) was limited because of poor image quality of one or both fields as a result of small pupils, media opacity, poor fixation or the absence of one field. The images of seven eyes (3.8%) were not able to be graded. Inter- and intra-grader reliability for any referrable level of DR were assessed by a κ statistic and found to be 1. Screening results and referral recommendations for the 93 patients are presented in Box 2. Sixteen patients (17.2%) were diagnosed with DR in at least one eye. The median time for photography was 6 minutes (IQR, 10 minutes). Most patients were “satisfied” to “very satisfied” with the photographic procedure (91 patients; 97.8%) and only two patients (2.2%) reported that the screening service would not give them more control over the management of their diabetic eye care.
Our finding that a third of diabetic patients had not undertaken biannual DR screening is similar to non-adherence rates found in other studies.4,6,15 Being busy, being recently diagnosed with diabetes and being unaware of the need for DR screening were identified as barriers to DR screening that were largely overcome by our screening model in this sample. Our finding that 17.2% of patients had DR is also comparable with the findings of other DR screening studies.15,16 Interestingly, we found no cases of PDR; however, the median known duration of diabetes was 4 years, which might account for this. Although our program successfully enrolled patients with diabetes who did not participate in biannual DR screening, our model needs to be revised to ensure that the GP and patient are well informed of the screening results to help patients make informed decisions about their diabetic eye care.
1 Protocol for diabetic retinopathy screening in pathology collection centres, and follow-up procedures
Received 18 July 2012, accepted 21 November 2012
- Melanie F Larizza1
- Lauren A Hodgson1
- Eva K Fenwick1
- Ryo Kawasaki1
- Ralph Audehm2
- Jie Jin Wang1
- Tien Y Wong1,3
- Ecosse L Lamoureux1
- 1 Centre for Eye Research Australia, Melbourne, VIC.
- 2 Dianella Community Health, Melbourne, VIC.
- 3 Singapore Eye Research Institute, Singapore.
We would like to acknowledge our funding sources — the NHMRC Centres for Clinical Research Excellence (NHMRC grants 454461, 529902 and 529923), and Diabetes Australia — Vic. Other research collaborators included St Vincent’s Pathology and the Royal Victorian Eye and Ear Hospital. The Centre for Eye Research Australia receives Operational Infrastructure Support from the Victorian government.
No relevant disclosures.
- 1. Ferris FL. How effective are treatments for diabetic retinopathy? JAMA 1993; 269: 1290-1291.
- 2. Rohan TE, Frost CD, Wald NJ. Prevention of blindness by screening for diabetic retinopathy: a quantitative assessment. BMJ 1989; 299: 1198-1201.
- 3. National Health and Medical Research Council. Guidelines for the management of diabetic retinopathy. Canberra: Australian Commonwealth Government, 2008. http://www.nhmrc.gov.au/guidelines/publications/di15 (accessed Jul 2012).
- 4. McCarty CA, Lloyd-Smith CW, Lee SE, et al. Use of eye care services by people with diabetes: The Melbourne Visual Impairment Project. Br J Ophthalmol 1998; 82: 410-414.
- 5. Muller A, Vu HT, Ferraro JG, et al. Utilization of eye care services in Victoria. Clin Experiment Ophthalmol 2006; 34: 445-448.
- 6. Tapp RJ, Zimmet PZ, Harper CA, et al. Diabetes care in an Australian population: frequency of screening examinations for eye and foot complications of diabetes. Diabetes Care 2004; 27: 688-693.
- 7. Arun CS, Al-Bermani A, Stannard K, Taylor R. Long-term impact of retinal screening on significant diabetes-related visual impairment in the working age population. Diabet Med 2009; 26: 489-492.
- 8. Bäcklund LB, Algvere PV, Rosenqvist U. New blindness in diabetes reduced by more than one-third in Stockholm County. Diabet Med 1997; 14: 732-740.
- 9. National Institute of Clinical Studies. Evidence–practice gaps report volume 1: a review of developments 2004–2007. Canberra: National Health and Medical Research Council, 2008.
- 10. Grading diabetic retinopathy from stereoscopic color fundus photographs — an extension of the modified Airlie House classification. ETDRS report number 10. Early Treatment Diabetic Retinopathy Study Research Group. Ophthalmology 1991; 98 (5 Suppl): 786-806.
- 11. Eiser JR, Eiser C, Riazi A, et al. Screening for diabetic retinopathy is well received by patients and may improve self-management intentions. Diabetic Med 2001; 18: 835-841.
- 12. Okoli U, Mackay K. An evaluation of diabetic retinopathy screening models. J Public Health Med 2002; 24: 190-195.
- 13. Spurling GKP, Askew DA, Hayman NE, et al. Retinal photography for diabetic retinopathy screening in Indigenous primary health care: the Inala experience. Aust N Z J Public Health 2010; 34 Suppl 1: S30-S33.
- 14. Diabetic retinopathy study. Report number 6. Design, methods, and baseline results. Report number 7. A modification of the Airlie House classification of diabetic retinopathy. Prepared by the Diabetic Retinopathy. Invest Ophthalmol Vis Sci 1981; 21: 1-226.
- 15. Harper CA, Livingston PM, Wood C, et al. Screening for diabetic retinopathy using a non-mydriatic retinal camera in rural Victoria. Aust N Z J Ophthalmol 1998; 26: 117-121.
- 16. Levy J, Lifshitz T, Goldfarb D, et al. Screening for diabetic retinopathy with a mobile non-mydriatic digital fundus camera in Southern Israel. Isr Med Assoc J 2011; 13: 137-140.





Abstract
Objective: To evaluate the feasibility of a novel diabetic retinopathy (DR) screening model using a pathology collection centre (PCC) as a screening site.
Design and setting: Cross-sectional pilot study conducted in one urban PCC in Victoria between 1 September 2009 and 15 March 2010. Trained pathology staff screened participants for DR using undilated, non-stereoscopic colour fundus photography.
Participants: Participants were patients who spoke English, were aged at least 12 years, had type 1 or type 2 diabetes, and did not undertake biannual DR screening.
Main outcome measures: Proportion of patients who do not participate in biannual DR screening; proportion of patients who accept DR screening while attending a PCC; proportion of gradable images; patient acceptance of the screening model; and effectiveness of the follow-up pathway.
Results: Over 6 months, 289 English-speaking patients with diabetes attended the PCC, of whom 99 (34.3%) had not undertaken biannual DR screening. Of these, 93 (93.9%) accepted our screening service. Overall, retinal images were sufficient for interpretation in 88 patients (94.6%) and the median time for photography was 6 minutes (interquartile range, 10 minutes). Two, eight and six cases of minimal, mild and moderate non-proliferative DR (NPDR) in the worst eye were found, respectively. Dissemination of screening results to treating doctors and patients was found to be suboptimal and will need to be revised.
Conclusion: DR screening in one urban PCC appears to be a feasible alternative for diabetic patients who do not undertake biannual screening for DR.