The Victorian Cancer Registry estimates that the number of men diagnosed with cancer will increase by 75% in the next 10 years, largely due to prostate cancer — the most common cancer diagnosis in men.1 Algorithms have been developed to calculate risk of prostate cancer progression at time of diagnosis for men with localised disease, classifying men into four groups: very low, low, intermediate and high risk of recurrence (Appendix 1).2,3 The National Comprehensive Cancer Network (NCCN) guidelines suggest that many men with very low-risk clinically localised disease should be managed with active surveillance; men with low-risk and intermediate-risk disease should be managed with active surveillance or with external beam radiation therapy (EBRT), radical prostatectomy (RP), brachytherapy, or a combination of these treatments; and men with high-risk disease should be managed with EBRT and androgen deprivation therapy (ADT), or EBRT, high dose rate brachytherapy and ADT, or RP and pelvic node dissection.2
Few studies have compared the effectiveness of treatments for localised disease.4 The recently published Prostate Cancer Intervention Versus Observation Trial (PIVOT) — a randomised control trial comparing RP with watchful waiting in 731 US patients with localised prostate cancer disease — showed that RP was associated with reduced all-cause mortality for men with a prostate-specific antigen (PSA) level greater than 10 ng/mL but that it offered no survival benefit to men with a PSA level of 10 ng/mL or less.5 A systematic review comparing disease-free survival for low dose rate brachytherapy with other treatments for men with clinically localised prostate cancer found no significant difference when compared with RP.6
Little is known about current patterns of care for men diagnosed with prostate cancer in Australia and whether treatment correlates with clinical practice guidelines. A population-based patterns-of-care survey conducted in Victoria in 1993 showed that 11% of men diagnosed with prostate cancer had received the diagnosis following informal screening and 25% received definitive curative therapy.7 New South Wales data show that the annual number of PSA tests performed more than doubled, from 184 350 tests in 1996 to 433 187 in 2006.8 The impact that this has had on patterns of care is unknown.
Our aim was to use the Prostate Cancer Registry to assess patterns of care for men diagnosed with prostate cancer at contributing hospitals in Victoria.
The Prostate Cancer Registry was established as a rapid case-ascertainment registry in 2009 to monitor patterns and quality of care for Victorian men diagnosed with prostate cancer. Details of the registry, including methods for data collection, are described elsewhere.9 Initially, four hospitals (three metropolitan, one regional) contributed data, accounting for about 25% of all Victorian patients with newly diagnosed prostate cancer (calculated based on 2009 incident notification data supplied to the Victorian Cancer Registry). Addition of 13 hospitals in 2010 increased capture to about 75% of newly diagnosed patients in Victoria. With Human Research Ethics Committee approval from the hospitals that contributed data to the Prostate Cancer Registry, prostate cancer notifications that were submitted by these hospitals to the Victorian Cancer Registry are also forwarded to the Prostate Cancer Registry.
Frequencies were used to describe treatment modalities and risk groups, using NCCN risk of disease progression categories (Appendix 1). While the NCCN includes a very low-risk category, this could not be calculated because the Prostate Cancer Registry does not collect PSA density, which is a required parameter in the calculation for very low risk. The Gleason score documented in the histopathology report at the time of biopsy was used in the NCCN algorithm. Where no clinical T stage was recorded but the Gleason score was ≤ 6 and PSA level was < 10 ng/mL, the patient was classified as having disease that was at low risk of progression. Patients were categorised as having no active treatment if their clinical records recorded “active surveillance” or “watchful waiting” and also if there was no documentation of active surveillance or watchful waiting but no treatment was delivered in the within 12 months of diagnosis.
A Pearson’s χ2 test was used to compare the association between presence of treatment within 12 months of diagnosis and categorical predictor variables of age at diagnosis, risk of disease progression at diagnosis and type of hospital and hospital location attended at diagnosis. These variables were then included in a multivariate logistic regression model. To compare categorical data for the 2008–2011 cohort with 1993 data,7 a two-sided Fisher exact test was used. Stata/IC 11.0 (StataCorp) was used for all analyses and a two-sided P value < 0.05 was considered to be statistically significant.
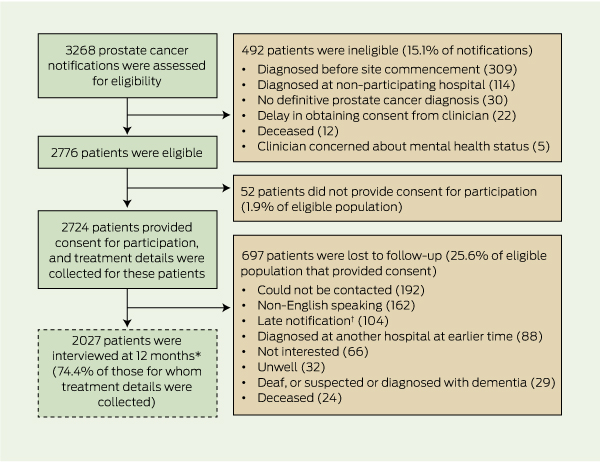
The recruitment of patients for the study is summarised in Box 1. A total of 3268 notifications were received before 1 February 2012 by the Prostate Cancer Registry for the period August 2008 to February 2011. In total, 492 men (15.1%) were ineligible. Of the 2776 who were eligible and for whom treatment data were sought, 52 (1.9%) did not provide consent for participation, leaving 2724 men (98.1%) for whom treatment data were collected and follow-up data were sought. As 697 men were lost to follow-up, confirmation of treatment and PSA levels were obtained for 2027 of the eligible men who provided consent for participation (74.4%).
Hospital characteristics and patient characteristics at time of diagnosis are shown in Box 2. Notifications from 11 public and six private hospitals were progressively added to the Prostate Cancer Registry. Most patients were diagnosed at metropolitan hospitals, and there were similar numbers of patients from public hospitals and private hospitals. Mean age at diagnosis was 66.2 years (SD, 8.9 years; range, 46.5–96.6 years). Median PSA level at diagnosis was 6.8 ng/mL (interquartile range, 4.7–10.6 ng/mL; range, 0.7–57 ng/mL). A PSA level of ≤ 4.0 ng/mL is considered normal. Most patients (92.9% [2531/2724]) were classified as having clinically localised disease, almost half were classified as being at intermediate risk of disease progression (44.1% [1201/2724]), and intermediate-risk disease affected almost half (47.5% [1201/2531]) of those who had clinically localised disease.
The principal treatment types according to risk of disease progression are summarised in Box 3. Nearly half of men with clinically localised disease (46.1% [1168/2531]) had RP, and more than half of those with intermediate risk of disease progression (54.5% [655/1201]) had RP. Of the 1195 men who had RP, 780 (65.3%) had open RP, 239 (20.0%) had robot-assisted laparoscopic RP, 88 (7.4%) had laparoscopic RP, five (0.4%) had laparoscopic RP converted to open RP and for 83 (6.9%) approach was not known. For patients with clinically localised disease, there was no significant association between disease risk category and surgical approach (P = 0.47). In total, 558 men (20.5% of those for whom treatment data were collected) were recorded as having received ADT. While 163 men received ADT alone, it was prescribed as an adjuvant or salvage therapy for men receiving EBRT (259), EBRT and high dose rate brachytherapy (60), RP and EBRT (30), RP alone (38), and with high dose rate brachytherapy, low dose rate brachytherapy or chemotherapy (8). Overall, 71.0% of men (1933/2724) received surgery, radiotherapy and/or brachytherapy, and 25.6% of men (698/2724) had EBRT. A notable proportion of those with high-risk localised disease (12.1% [72/594]) received no active treatment, and 40.6% of those with low risk of progression (299/736) received no active treatment.
Frequencies of the categories of age at diagnosis, risk of disease progression at diagnosis and type and location of hospital that provided the prostate cancer notification for men who received active treatment within 12 months of diagnosis and those who did not are shown in Box 4. Results of multivariate logistic regression analysis are shown in Box 5. When men with locally advanced disease were excluded from the model, having a prostate cancer notification made by a private hospital remained an independent factor associated with not receiving treatment (odds ratio, 1.39; 95% CI, 1.12–1.72). When only men with low-risk disease were included in the model, the odds ratio increased to 1.42 (95% CI, 1.16–1.75).
A comparison of the patterns of care for 2008–2011 and for 1993 is shown in Appendix 2. Median PSA level at diagnosis declined from 20.1 ng/mL in 1993 to 6.8 ng/mL in 2008–2011, and the proportion of men receiving no active treatment declined from 35.6% (373/1048) in 1993 to 22.7% (619/2724) in 2008–2011.7
From 1993 to 2008–2011, there were notable declines in the median PSA level at diagnosis and the percentage of men receiving no active treatment. However, this comparison is based on different methods of data collection — treatment for men in the 1993 study was determined by surveying the treating clinicians 3 years after diagnosis, whereas our 2008–2011 treatment data were collected 12 months after diagnosis. The proportion of men who had no active treatment in our study (22.7%) is similar to that in a US Medicare-linked study (about 23% for men diagnosed in 2007)10 and a South Australian study monitoring men treated in the public health system (about 20% of men diagnosed from 1998 to 2007).11 Men with low-risk disease in our study were four times more likely to receive no treatment compared with men with low-risk disease in 40 urology practices in the United States and recruited to the Cancer of the Prostate Strategic Urologic Research Endeavor (CAPSURE) registry (40.6% v 9%).12 This suggests that there is less overtreatment of patients with low-risk disease in Victoria compared with the US.
Our finding that almost half of men with clinically localised disease had RP is similar to the 50% rate of RP described for men recruited to the CAPSURE registry.12 In contrast, much lower rates have been reported elsewhere: 12%–16% in US Medicare-linked reviews (which captured only men older than 64 years and those with a disability or end-stage renal disease),10,13 11% in a population-based dataset from Northern England,14 23% in the study of men in South Australia who were treated in the public health system,11 and 13% in the previous study of men in Victoria.7
Our finding that 71.0% of men received surgery, radiotherapy and/or brachytherapy contrasts with results of the 1993 Victorian study, in which 25% of men received initial treatment with curative intent.7 Extrapolating this to statewide data using the Victorian Cancer Registry15 would equate to a sevenfold increase in the number of men having RP, from about 280 in 19937 to 2180 in 2010. Similarly, the proportion of men treated with EBRT and high dose rate brachytherapy has increased from 12.0% to 25.6%, or from about 230 men7 to 1370 men over the same period. In future, we expect that the proportion of men receiving robot-assisted laparoscopic RP (20.0% of those who had RP in our study) will increase, as the Prostate Cancer Registry did not receive notifications from facilities performing robot-assisted laparoscopic RP before December 2010.
We identified a sixfold decline in hormonal therapy as front-line treatment for prostate cancer compared with the 1993 Victorian study (38.8% to 6.0%).7 This downward trend was also reported in the South Australian study, although it was less pronounced (OR, 0.58 [95% CI, 0.40–0.82] for men diagnosed between 2004 and 2006 compared with those diagnosed between 1998 and 2000).11
The apparent change in patterns of prostate cancer management in Victoria is consistent with results of studies undertaken in Queensland16 and New South Wales.17 In contrast, the institutionally based South Australian registry shows a consistent proportion of men treated with RP (23% over the 1998–2007 study period), or possibly a slight decline.11
We found that the probability of receiving treatment declined as men aged and as the likelihood of disease progression increased, which is consistent with previous findings.7,12 Of particular interest was our finding that having a prostate cancer notification made by a private hospital was an independent factor for not receiving radical treatment, even after age and of risk of disease progression were taken into account. This contrasts the results of a US study, which showed that having private health insurance was associated with higher rates of treatment compared with not having private health insurance.18
2 Hospital characteristics and patient characteristics at diagnosis of prostate cancer (n = 2724)
* National Comprehensive Cancer Network risk categories determined taking into account prostate-specific antigen level, Gleason score and clinical stage.7 |
|||||||||||||||
3 Frequency of principal treatment types according to risk of disease progression* at diagnosis (n = 2724)
4 Age at diagnosis, risk of disease progression and type and location of hospital that made the prostate cancer notification for men who received active treatment within 12 months of diagnosis and those who did not (n = 2724)
* National Comprehensive Cancer Network risk categories determined taking into account prostate-specific antigen level, Gleason score and clinical stage.7 |
|||||||||||||||
5 Results of multivariate logistic regression analysis of factors associated with men not receiving active treatment within 12 months of diagnosis (n = 2724)
* Reference category. † National Comprehensive Cancer Network risk categories determined taking into account prostate-specific antigen level, Gleason score and clinical stage.7 |
|||||||||||||||
Received 8 August 2012, accepted 4 March 2013
- Sue M Evans1
- Jeremy L Millar2
- Ian D Davis3
- Declan G Murphy4,5
- Damien M Bolton6
- Graham G Giles7
- Mark Frydenberg8,9
- Nick Andrianopoulos1
- Julie M Wood1
- Albert G Frauman10
- Anthony J Costello4
- John J McNeil1
- 1 Department of Epidemiology and Preventive Medicine, Monash University, Melbourne, VIC.
- 2 Department of Radiation Oncology, Alfred Hospital, Melbourne, VIC.
- 3 Eastern Health Clinical School, Monash University and Eastern Health, Melbourne, VIC.
- 4 Australian Prostate Cancer Research Centre, Epworth Healthcare, Melbourne, VIC.
- 5 Division of Cancer Surgery, Peter MacCallum Cancer Centre, Melbourne, VIC.
- 6 Department of Urology, Austin and Repatriation General Hospital, Austin Health, Melbourne, VIC.
- 7 Cancer Control Research Institute, Cancer Council Victoria, Melbourne, VIC.
- 8 Department of Surgery, Monash University, Melbourne, VIC.
- 9 Department of Urology, Cabrini Health, Melbourne, VIC.
- 10 Department of Clinical Pharmacology and Therapeutics, University of Melbourne, Melbourne, VIC.
This project was co-funded through the Priority-driven Collaborative Cancer Research Scheme by the Prostate Cancer Foundation of Australia, the radiation oncology section of the Australian Government Department of Health and Ageing and Cancer Australia.
No relevant disclosures.
- 1. Thursfield V, Farrugia H. Cancer in Victoria: statistics and trends 2011. Melbourne: Cancer Council Victoria, 2012. http://www.cancervic.org. au/downloads/cec/cancer-in-vic/CCV-statistics-trends-2011.pdf (accessed Mar 2013).
- 2. National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology: prostate cancer [subscription only]. http://www.nccn.org/professionals/physician_gls/pdf/prostate.pdf (accessed May 2013).
- 3. D’Amico AV. Combined-modality staging for localized adenocarcinoma of the prostate. Oncology (Williston Park) 2001; 15: 1049-1059.
- 4. Wilt TJ, MacDonald R, Rutks I, et al. Systematic review: comparative effectiveness and harms of treatments for clinically localized prostate cancer. Ann Intern Med 2008; 148: 435-448.
- 5. Nielsen ME. How bad are positive margins after radical prostatectomy and how are they best managed? J Urol 2009; 182: 1257-1258.
- 6. Peinemann F, Grouven U, Hemkens LG, et al. Low-dose rate brachytherapy for men with localized prostate cancer. Cochrane Database Syst Rev 2011; (7): CD008871.
- 7. Frydenberg M, Giles GG, Mameghan H, et al. Prostate cancer in Victoria in 1993: patterns of reported management. Med J Aust 2000; 172: 270-274.
- 8. Smith DP, Supramaniam R, Marshall VR, Armstrong BK. Prostate cancer and prostate-specific antigen testing in New South Wales. Med J Aust 2008; 189: 315-318. <MJA full text>
- 9. Evans SM, Millar JL, Wood JM, et al. The Prostate Cancer Registry: monitoring patterns and quality of care for men diagnosed with prostate cancer. BJU Int 2013; 111 (4 Pt B): E158-E166.
- 10. Dinan MA, Robinson TJ, Zagar TM, et al. Changes in initial treatment for prostate cancer among Medicare beneficiaries, 1999–2007. Int J Radiat Oncol Biol Phys 2012; 82: e781-e786.
- 11. Beckmann K, Pinnock CB, Tamblyn DJ, et al. Clinical and socio-demographic profile of an Australian multi-institutional prostate cancer cohort. Asia Pac J Clin Oncol 2009; 5: 247-256.
- 12. Cooperberg MR, Broering JM, Carroll PR. Time trends and local variation in primary treatment of localized prostate cancer. J Clin Oncol 2010; 28: 1117-1123.
- 13. Kapoor DA, Zimberg SH, Ohrin LM, et al. Utilization trends in prostate cancer therapy. J Urol 2011; 186: 860-864.
- 14. Fairley L, Baker M, Whiteway J, et al. Trends in non-metastatic prostate cancer management in the Northern and Yorkshire region of England, 2000–2006. Br J Cancer 2009; 101: 1839-1845.
- 15. Thursfield V, Farrugia H. Cancer in Victoria: statistics and trends 2010. Melbourne: Cancer Council Victoria, 2011. http://www.cancervic.org. au/downloads/cec/cancer-in-vic/CCV-Statistics-trends.pdf (accessed Mar 2013).
- 16. Baade PD, Youlden DR, Coory MD, et al. Urban–rural differences in prostate cancer outcomes in Australia: what has changed? Med J Aust 2011; 194: 293-296. <MJA full text>
- 17. Hayen A, Smith DP, Patel MI, O’Connell DL. Patterns of surgical care for prostate cancer in NSW, 1993–2002: rural/urban and socio-economic variation. Aust N Z J Public Health 2008; 32: 417-420.
- 18. Wu X, Chen VW, Andrews PA, et al. Initial treatment patterns for clinically localized prostate cancer and factors associated with the treatment in Louisiana. J La State Med Soc 2005; 157: 188-194.





Abstract
Objective: To describe patterns of care for men diagnosed with prostate cancer in Victoria, Australia, between 2008 and 2011.
Design, setting and patients: Men who were diagnosed with prostate cancer at 11 public and six private hospitals in Victoria from August 2008 to February 2011, and for whom prostate cancer notifications were received by the Prostate Cancer Registry.
Main outcome measures: Characteristics of men diagnosed with prostate cancer; details of treatment provided within 12 months of diagnosis, according to National Comprehensive Cancer Network risk categories; and characteristics of men who did not receive active treatment within 12 months of diagnosis.
Results: Treatment details were collected for 98.1% of men who were assessed as eligible to participate in the study (2724/2776) and were confirmed by telephone 12 months after diagnosis for 74.4% of them (2027/2724). Most patients (2531/2724 [92.9%]) were diagnosed with clinically localised disease, of whom 1201 (47.5%) were at intermediate risk of disease progression. Within 12 months of diagnosis, 299 of the 736 patients (40.6%) who had been diagnosed as having disease that was at low risk of progression had received no active treatment, and 72 of 594 patients (12.1%) who had been diagnosed as having disease that was at high risk of progression had received no active treatment. Of those diagnosed as having intermediate risk of disease progression, 54.5% (655/1201) had undergone radical prostatectomy. Those who received no active treatment were more likely than those who received active treatment to be older (odds ratio [95% CI], 2.96 [2.01–4.38], 10.94 [6.96–17.21] and 32.76 [15.84–67.89], respectively, for age 65–74 years, 75–84 years and ≥ 85 years, compared with < 55 years), to have less advanced disease (odds ratio [95% CI], 0.20 [0.16–0.26], 0.09 [0.06–0.12] and 0.05 [0.02–0.90], respectively, for intermediate, high and very high-risk [locally advanced] or metastatic disease, compared with low-risk disease) and to have had their prostate cancer notified by a private hospital (odds ratio [95% CI], 1.35 [1.10–1.66], compared with public hospital).
Conclusion: Our data reveal a considerable “stage migration” towards earlier diagnosis of prostate cancer in Victoria and a large increase in the use of radical prostatectomy among men with clinically localised disease.