This is the first report of endovascular therapy for acute ischaemic stroke in a patient with atrial fibrillation who was taking the direct thrombin inhibitor dabigatran for stroke prevention. As more of these agents will become available soon, endovascular therapy is likely to be used more frequently for acute stroke.
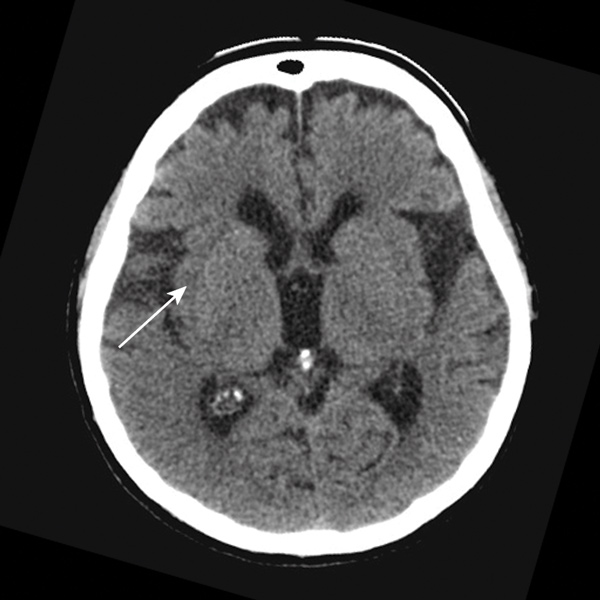
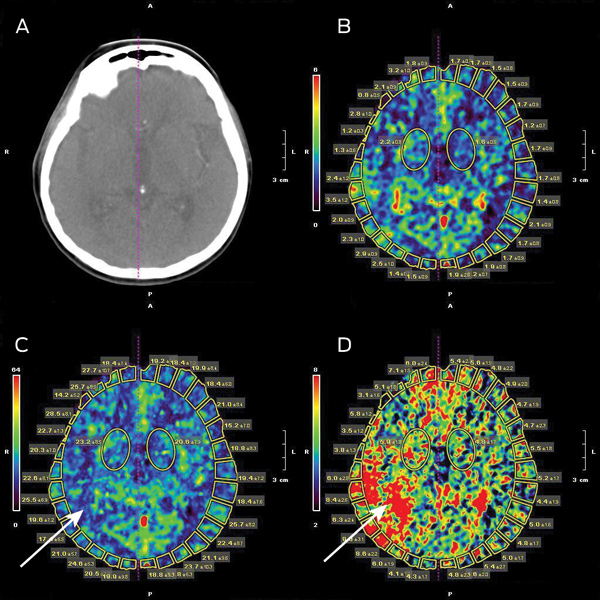
The local hospital acute stroke treatment protocol was followed, commencing with attendance by a code stroke team. Conventional non-contrast computed tomography (CT) of the brain performed 2 hours after symptom onset showed subtle and early obscuration of the right posterior insular cortex (Box 1). Findings on perfusion CT (Aquilion One, Toshiba Medical Systems, Otawara, Japan) were consistent with a large area of ischaemic penumbra in a right posterior middle cerebral artery (MCA) distribution (Box 2), corresponding to a right MCA (M2 segment) thrombotic occlusion on CT angiography.
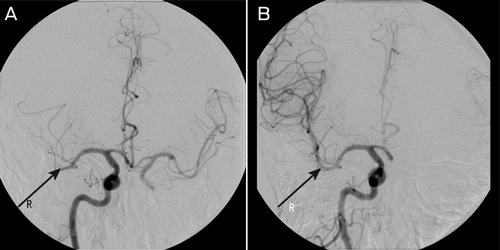
A clot retrieval device (Solitaire AB Neurovascular Remodeling Device, 4 × 15 mm; ev3 Inc, Irvine, Calif, USA) was used to achieve reperfusion to the right MCA beyond the M2 segment (Box 3). The procedure was performed under general anaesthesia. Initial diagnostic imaging was performed using a 5-French diagnostic catheter and heparin (3000 IU) was delivered intravenously. A 6-French shuttle sheath (Cook Medical, Brisbane, Qld) was positioned within the common carotid artery, and then a 6-French neuron guide catheter was positioned within the inferior right internal carotid artery. A Transcend floppy guidewire (0.014"; Boston Scientific, Natick, Mass, USA) and Rebar 18 microcatheter (ev3 Inc, Irvine, Calif, USA) were advanced into the right MCA and two occluded branches selected. The microguidewire was passed through the thrombus and the microcatheter was advanced over the wire about 5 mm beyond the thrombus. The position of the microcatheter was confirmed by contrast injection. The stent was then advanced to the distal tip of the microcatheter and then unsheathed, both within and just beyond the thrombus. The distal microcatheter marker was positioned proximal to the uncovered stent. The unsheathed Solitaire AB stent and microcatheter were removed as a unit while suction was applied to the guiding catheter. This procedure was repeated with each pass. Angiographic assessment was performed after each pass with the Solitaire AB stent to assess for revascularisation status, vessel trauma or distal emboli. Two passes as described using the Solitaire AB stent were performed within each of the two affected vessels achieving TIMI (Thrombolysis In Myocardial Infarction) grade 2 flow or better.
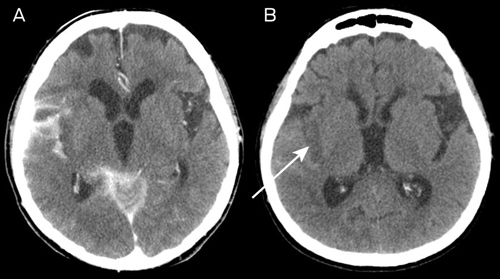
The NIHSS scores immediately after the procedure and at 24 hours were 17 and 8, respectively. A routine CT scan of the brain at 24 hours showed hyperdensity in the subarachnoid space (which had largely resolved on further CT imaging by Day 11) as well as infarction, predominantly within the posterior insular cortex (Box 4).
Intravenous administration of rtPA is now considered standard therapy in acute stroke, and is being widely used around the world and in Australia.1 However, the concomitant use of anticoagulants, including dabigatran, contraindicates use of rtPA because of a theoretical risk of intracerebral haemorrhage. Despite this, two recent case reports have shown benefit of using rtPA in patients taking dabigatran who have had a stroke, although its safety requires confirmation in a larger cohort.2,3 Endovascular approaches using mechanical clot extraction devices do not expose patients to thrombolytic agents, and thus their use may have fewer haemorrhagic sequelae in stroke patients taking dabigatran.4
To the best of our knowledge, this is the first case report of endovascular therapy for acute ischaemic stroke in a patient taking dabigatran. This situation is likely to occur more frequently in the near future as direct thrombin inhibitors including dabigatran become available as alternatives to warfarin for stroke prevention in patients with atrial fibrillation.5 It should be emphasised that, unlike warfarin, the INR and aPTT are not very useful indicators of the anticoagulant effect of dabigatran, and pathology laboratories are currently establishing appropriate monitoring strategies for this new group of anticoagulants.6
The Solitaire device used in our patient was originally designed for cerebral aneurysm bridging. However, a recent retrospective review of patients treated endovascularly with the Solitaire AB stent suggests that this procedure appears to be safe and able to deliver favourable clinical outcomes in patients with proximal cerebral vessel occlusion,7 albeit without anticoagulation therapy. Currently, no guideline exists on the safety of treatments for acute stroke in patients taking dabigatran. Although there is anecdotal evidence of success with intravenous rtPA therapy in this setting, endovascular therapy holds promise because the risk of bleeding is theoretically lower. This is important because there is no practical immediate reversal of dabigatran currently available. There is preliminary evidence of the safety of endovascular therapy in acute stroke;7 however, data on its safety in patients taking anticoagulants are lacking. Such data would enable the development of a consensus guideline for this increasingly prevalent clinical problem.
- 1. Leyden JM, Chong WK, Kleinig T, et al. A population-based study of thrombolysis for acute stroke in South Australia. Med J Aust 2011; 194: 111-115. <MJA full text>
- 2. De Smedt A, De Raedt S, Nieboer K, et al. Intravenous thrombolysis with recombinant tissue plasminogen activator in a stroke patient treated with dabigatran. Cerebrovasc Dis 2010; 30: 533-534.
- 3. Matute MC, Guillán M, García-Caldentey J, et al. Thrombolysis treatment for acute ischaemic stroke in a patient on treatment with dabigatran. Thromb Haemost 2011; 106: 178-179.
- 4. Lee L, Chryssidis S, Jannes J. Rescue therapy with local intra-arterial urokinase after poor clinical response with intravenous recombinant tissue plasminogen activator in acute ischaemic stroke. Intern Med J 2011; 41: 570-573.
- 5. Jaffer AK. Update in hospital medicine: studies likely to affect inpatient practice in 2011. Cleve Clin J Med 2011; 78: 430-434.
- 6. Favaloro EJ, Lippi G, Koutts J. Laboratory testing of anticoagulants: the present and the future. Pathology 2011; 43: 682-692.
- 7. Miteff F, Faulder KC, Goh AC, et al. Mechanical thrombectomy with a self-expanding retrievable intracranial stent (Solitaire AB): experience in 26 patients with acute cerebral artery occlusion. Am J Neuroradiol 2011; 32: 1078-1081.





No relevant disclosures.