Persistence with long-term medication in hypertension or dyslipidaemia (hypercholesterolaemia) in the community is known to be unsatisfactory.1-8 Out of 49 000 patients in Australia who were initiated on antihypertensive therapy in 2004–2006, 19% failed to collect the first repeat prescription at 1 month and 50% had discontinued therapy within 20 months.8 Overall tablet burden and dosing frequency are predictors of poor compliance,9,10 and this has stimulated the development of single-pill, fixed-dose combinations administered once daily. We analysed Medicare Australia Pharmaceutical Benefits Scheme (PBS) claims over the period December 2006 – March 2010 for a single-pill, fixed-dose tablet combination of amlodipine (a calcium channel blocker [CCB]) and atorvastatin (a statin). It is prescribed for the management of hypertension and dyslipidaemia together. Using these data as a broad surrogate of persistence, we derived a picture of persistence in Australian patients prescribed amlodipine and atorvastatin together (AA) in one pill, in comparison with those prescribed the separate components as two-pill, dual-drug therapy.
The PBS database generated information on 6204 patients who were prescribed amlodipine and atorvastatin as two-pill therapy, and on 4146 patients who were prescribed AA as single-pill therapy. Their demographic features by selected prior treatments are summarised in Box 1.
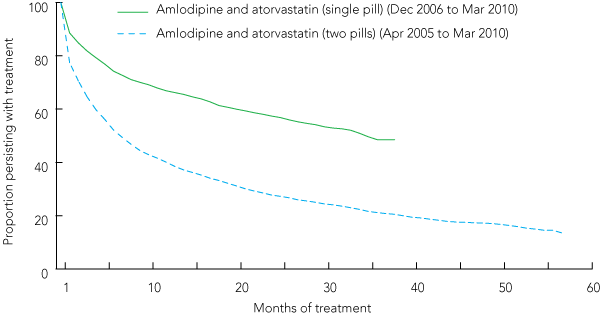
Persistence curves for those initiated on single-pill AA or dual-pill amlodipine and atorvastatin are shown in Box 2. Apparent persistence was far superior in those initiated on single-pill AA. Median persistence time (MPT) on single-pill AA was 35 months (95% CI, 33 to ≥ 38 months), and was only 7 months (95% CI, 6–8 months) on amlodipine and atorvastatin. Patients switching from amlodipine and atorvastatin to single-pill AA were censored at that point, and this would slightly overestimate the extent of cessation on two-pill therapy. The hazard ratio for cessation of amlodipine and atorvastatin (two pills) versus AA (one pill) was 2.17 (95% CI, 2.05–2.13; P < 0.0001), that is, a 117% higher rate of cessation. Since much of the persistence data for amlodipine and atorvastatin preceded that for single-pill AA by 20 months in real time, the respective proportions of patients failing to collect their first repeat prescription after 1 month was highly informative: 23% for the two-pill combination and only 11% for the single-pill prescription. The corresponding proportions of patients failing to persist with treatment at 12 months were 59% and 33%.
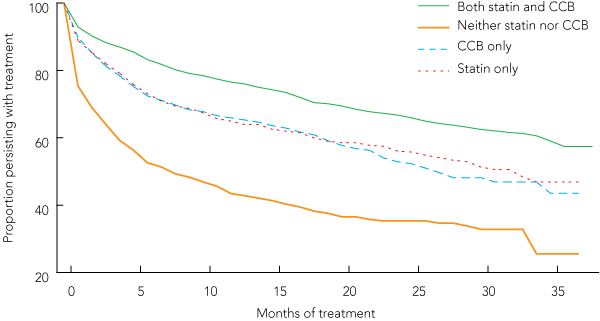
Persistence with single-pill AA treatment varied according to the treatment used in the 3 months before initiation (see Box 3). The MPT was only 8 months (95% CI, 6–11 months) in those having used neither a CCB nor a statin, 32 months (95% CI, 28 to ≥ 38 months) in those using a statin only, 27 months (95% CI, 23 to ≥ 38 months) in those using a CCB only, and ≥ 38 months (confidence intervals could not be derived) in those using both a CCB and a statin. The corresponding proportions of patients failing to collect their first repeat prescription at 1 month were 25%, 11%, 10% and 7%. The corresponding proportions of patients failing to persist with treatment at 12 months were 57%, 35%, 34% and 23%.
The MPT using the single pill was only 8 months (95% CI, 6–12 months) in those not previously using any antihypertensive drugs, compared with ≥ 37 months (95% CI, 29 to ≥ 38 months) in those using one or more antihypertensive drugs. The MPT on single-pill AA varied with age. This was 17 months (95% CI, 6–30 months) in those aged < 50 years, 28 months (95% CI, 19 to ≥ 38 months) in those 50–59 years, 36 months (95% CI, 34 to ≥ 38 months) in those aged 60–69 years, ≥ 38 months (95% CI, 35 to ≥ 38 months) in those aged 70–79 years, and 25 months (95% CI, 18–32 months) in those aged ≥ 80 years.
Findings from the stepwise multivariate model of AA, single-pill cessation are presented in Box 4. Taking prior CCB and statin therapy as a reference group, if neither a CCB nor a statin were used in prior therapy, treatment cessation on single-pill AA increased by 165% (95% CI, 127%–210%; P < 0.0001). If a CCB only or a statin only had been used in prior therapy, cessation on single-pill AA increased by 55% (95% CI, 31%–83%; P < 0.0001) and 48% (95% CI, 29%–70%; P < 0.0001), respectively. Taking the use of two antihypertensive drugs as a reference group, in those naive to prior antihypertensive therapy, cessation of single-pill AA increased by 38% (95% CI, 18%–62%; P < 0.001), while in patients using three or more antihypertensive agents prior to treatment, cessation of single-pill AA increased by 15% (95% CI, 2%–29%; P < 0.02). Females were 13% (95% CI, 2%–25%; P < 0.02) more likely to cease single-pill AA treatment than males. Cessation of single-pill AA treatment was significantly greater in younger age groups, but this was significant only in the univariate model.
We confirmed that long-term persistence with a single-pill combined formulation in the management of hypertension and dyslipidaemia in the Australian population is far superior to that observed when patients have used the separate component drugs. Since patients in the amlodipine and atorvastatin group were censored if they switched to single-pill AA treatment, the switch being a logical clinical step, this will have slightly understated the apparent persistence in the two-pill group. If patients had not been censored at the point of switch, their apparent excess cessation, compared with single-pill AA, would have been reduced from 117% to 110%. Furthermore, the proportion failing to collect the first repeat prescription was twofold greater in the two-pill group. Our findings confirm the thrust of conclusions from United States health maintenance and managed care organisations.11-13
By examination of treatment in the 3 months before initiation, we have been able to identify which patients are more likely to persist with treatment. Patients appear to be more likely to persist on a single, combination pill if they have used one or both components previously, or if they have received any antihypertensive therapy, as noted by others.12 In univariate analysis, poorer persistence was noted on the single pill in the youngest and oldest subgroups, consistent with a pattern we previously reported in patients newly treated with antihypertensive drugs.8
Other contributors to poor persistence include the cost of medication and the risk of adverse events.10 The former is perhaps less relevant in our study as the patients paid only a nominal amount for medication. Adverse events with antihypertensive drugs do contribute to poor persistence.14
There were some limitations in our approach. There was an absence of medical histories; we had no information on the degree of blood pressure and cholesterol control; only concession card holders were studied (yet they still represented around 65% of Australian patients using these medications); and we did not adjust for any temporal effects in the data.
It must follow that poor persistence will impact on the clinical outcomes. Cardiovascular disease outcomes in the second Australian National Blood Pressure Study were 20%–23% higher in patients reporting poor compliance with their medication.15 It has been calculated that hypertensive patients taking antihypertensive and statin therapy at real-world adherence levels can be expected to receive about 50% of the potential benefit seen in clinical trials.16 In a recent US study using similar drugs, those on a single-pill regimen were more adherent and showed a lower cardiovascular event rate. Incidence rates per 100 person-years were 1.39 on a single pill, 2.21 on a two-pill regimen; and 2.26 in the non-adherent group on any therapy.17
What can be done to improve persistence? Drugs with fewer side effects, more convenient once-daily dosing schedules, a greater number of single-pill combination products and better patient education are potential solutions. Ultimately, better persistence and better control of hypertension, dyslipidaemia and other chronic diseases or risk factors will rest on a partnership between health professionals and the individual patient.
1 Characteristics of patients and their prior treatments at study entry*
Amlodipine and atorvastatin (two-pill therapy) (n = 6204) |
AA (single-pill therapy) (n = 4146) |
P |
|||||||||||||
Male |
2858 (46%) |
1921 (46%) |
ns |
||||||||||||
Prescriptions from general practitioner |
4589 (74%) |
3789 (91%) |
|
||||||||||||
Prescriptions from specialist |
1615 (26%) |
357 (9%) |
< 0.0001 |
||||||||||||
Age distribution (years) |
|
|
|
||||||||||||
< 50 |
239 (4%) |
165 (4%) |
|
||||||||||||
50–59 |
510 (8%) |
352 (8%) |
|
||||||||||||
60–69 |
1591 (26%) |
1139 (27%) |
|
||||||||||||
70–79 |
2602 (42%) |
1732 (42%) |
|
||||||||||||
≥ 80 |
1262 (20%) |
758 (18%) |
ns |
||||||||||||
Mean age, years (SD) |
71.2 (10.3) |
70.7 (10.3) |
< 0.007 |
||||||||||||
Treatment during previous 3 months |
|
|
|
||||||||||||
Amlodipine only |
1814 (29%) |
751 (18%) |
< 0.0001 |
||||||||||||
Atorvastatin only |
3063 (49%) |
993 (24%) |
< 0.0001 |
||||||||||||
Neither of above |
918 (15%) |
1358 (33%) |
< 0.0001 |
||||||||||||
Amlodipine and atorvastatin |
409 (7%) |
1044 (25%) |
na |
||||||||||||
Other statins |
523 (8%) |
982 (24%) |
< 0.0001 |
||||||||||||
Other calcium channel blockers |
562 (8%) |
633 (15%) |
< 0.0001 |
||||||||||||
Antihypertensive drugs previously being used |
|
|
|
||||||||||||
Nil |
665 (11%) |
472 (11%) |
ns |
||||||||||||
1 |
1397 (23%) |
826 (20%) |
< 0.002 |
||||||||||||
2 |
2007 (32%) |
1195 (29%) |
< 0.0001 |
||||||||||||
≥ 3 |
2135 (34%) |
1653 (40%) |
< 0.0001 |
||||||||||||
AA = amlodipine+atorvastatin. ns = not significant. na = not applicable. * Data are frequency except age. Frequencies are contrasted by χ2, age by t test. |
|||||||||||||||
2 Persistence curves for AA single-pill therapy versus amlodipine and atorvastatin two-pill therapy*
 | |||||||||||||||
|
AA = amlodipine+atorvastatin. * Patients using two-pill amlodipine and atorvastatin therapy were censored (discontinued from our dataset) if they switched to single-pill amlodipine and atorvastatin. | |||||||||||||||
3 Persistence curves for AA single-pill therapy, according to prior therapy
 | |||||||||||||||
|
AA = amlodipine+atorvastatin. CCB = calcium channel blocker. | |||||||||||||||
4 Stepwise multivariate analysis of patients discontinuing AA single-pill therapy*
|
Univariate HR (95% CI) |
P |
Multivariate HR (95% CI) |
P |
|||||||||||
Female |
1.18 (1.07–1.31) |
< 0.002 |
1.13 (1.02-1.25) |
< 0.02 |
|||||||||||
Male |
1.00 |
|
1.00 |
|
|||||||||||
Specialist treatment |
1.10 (0.92–1.31) |
ns |
|
|
|||||||||||
GP treatment |
1.00 |
|
|
|
|||||||||||
Age (years) |
|
|
|
|
|||||||||||
< 50 |
1.76 (1.39–2.22) |
< 0.0001 |
|
|
|||||||||||
50–69 |
1.33 (1.11–1.59) |
< 0.002 |
|
|
|||||||||||
60–69 |
1.05 (0.93–1.19) |
ns |
|
|
|||||||||||
70–79 |
1.00 |
|
|
|
|||||||||||
≥ 80 |
1.38 (1.21–1.58) |
< 0.0001 |
1.36 (1.20-1.54) |
< 0.0001 |
|||||||||||
Treatment in previous 3 months |
|
|
|
|
|||||||||||
Statin only† |
1.48 (1.30–1.68) |
< 0.0001 |
1.48 (1.29–1.70) |
< 0.0001 |
|||||||||||
CCB only† |
1.53 (1.29–1.81) |
< 0.0001 |
1.55 (1.31–1.83) |
< 0.0001 |
|||||||||||
Neither CCB nor statin† |
2.78 (2.44–3.16) |
< 0.0001 |
2.65 (2.27–3.10) |
< 0.0001 |
|||||||||||
CCB and statin† |
1.00 |
|
1.00 |
|
|||||||||||
Antihypertensive drugs previously used |
|
|
|
|
|||||||||||
Nil |
2.22 (1.90–2.59) |
< 0.0001 |
1.38 (1.18–1.62) |
< 0.0001 |
|||||||||||
1 |
1.21 (1.04–1.40) |
< 0.02 |
|
|
|||||||||||
2 |
1.00 |
|
|
|
|||||||||||
≥ 3 |
1.04 (0.92–1.18) |
ns |
1.15 (1.02–1.29) |
< 0.02 |
|||||||||||
AA = amlodipine + atorvastatin as a single pill. HR = hazard ratio. GP = general practitioner. ns = not significant. CCB = calcium channel blocker. * In the stepwise approach, only items of statistical significance remain in the final model. † The statistics on prior statin or CCB therapy relate to the curves in Box 3. |
|||||||||||||||
Received 4 November 2010, accepted 10 May 2011
- Leon A Simons1
- Michael Ortiz1,2
- Gordon Calcino3
- 1 St Vincent’s Hospital, University of New South Wales, Sydney, NSW.
- 2 Abbott Products, Sydney, NSW.
- 3 HI Connections, Canberra, ACT.
Raw data for the study were supplied by Medicare Australia. Leon Simons contributed his time in an honorary capacity. Michael Ortiz is an employee of Abbott Products. Gordon Calcino is an independent contractor. The manufacturer of the amlodipine and atorvastatin single pill had no involvement in any aspect of this study or report.
None relevant to this article declared (ICMJE disclosure forms completed).
- 1. Simons LA, Levis G, Simons J. Apparent discontinuation rates in patients prescribed lipid-lowering drugs. Med J Aust 1996; 164: 208-211.
- 2. Caro JJ, Salas M, Speckman JI, et al. Persistence with treatment for hypertension in actual practice. CMAJ 1999; 160: 31-37.
- 3. Simons LA, Simons J, Mcmanus P, Dudley J. Discontinuation rates for use of statins are high. BMJ 2000; 321: 1084.
- 4. Benner JS, Glynn RJ, Mogun H, et al. Long-term persistence in use of statin therapy in elderly patients. JAMA 2002; 288: 455-461.
- 5. Degli Espositi E, Sturani A, Di Martino M, et al. Long-term persistence with antihypertensive drugs in new patients. J Hum Hypertens 2002; 16: 439-444.
- 6. Osterberg L, Blaschke T. Adherence to medication. N Engl J Med 2005; 353: 487-497.
- 7. Elliott WJ, Plauschinat CA, Skrepnek GH, Gause D. Persistence, adherence, and risk of discontinuation with commonly prescribed antihypertensive drug monotherapies. J Am Board Fam Med 2007; 20: 72-80.
- 8. Simons LA, Ortiz M, Calcino G. Persistence with antihypertensive medication: Australia-wide experience, 2004–2006. Med J Aust 2008; 188: 224-227. <MJA full text>
- 9. Kiortsis DN, Giral P, Bruckert E, Turpin G. Factors associated with low compliance with lipid-lowering drugs in hyperlipidemic patients. J Clin Pharm Ther 2000; 25: 445-451.
- 10. Chapman RH, Benner JS, Petrilla AA, et al. Predictors of adherence with antihypertensive and lipid-lowering therapy. Arch Intern Med 2005; 165: 1147-1152.
- 11. Bangalore S, Kamalakkannan G, Parkar S, Messerli FH. Fixed-dose combinations improve medication compliance: a meta-analysis. Am J Med 2007; 120: 713-719.
- 12. Hussein MA, Chapman RH, Benner JS, et al. Does a single-pill antihypertensive/lipid-lowering regimen improve adherence in US managed care enrolees? Am J Cardiovasc Drugs 2010; 10: 193-202.
- 13. Gupta AK, Arshad S, Poulter NR. Compliance, safety, and effectiveness of fixed-dose combinations of antihypertensive agents: a meta-analysis. Hypertension 2010; 55: 399-407.
- 14. After the diagnosis: adherence and persistence with hypertension therapy. Am J Manag Care 2005; 11 (13 Suppl): S395-S399.
- 15. Nelson MR, Reid CM, Ryan P, et al. Self-reported adherence with medication and cardiovascular disease outcomes in the second Australian National Blood Pressure Study (ANBP2). Med J Aust 2006; 185: 487-489. <MJA full text>
- 16. Cherry SB, Benner JS, Hussein MA, et al. The clinical and economic burden of nonadherence with antihypertensive and lipid-lowering therapy in hypertensive patients. Value Health 2009; 12: 489-497.
- 17. Chapman RH, Yeaw J, Roberts CS. Association between adherence to calcium-channel blocker and statin medications and likelihood of cardiovascular events among US managed care enrollees. BMC Cardiovasc Disord 2010; 10: 29.





Abstract
Objective: To study patient persistence on therapy for hypertension and dyslipidaemia using a single-pill combination compared with a two-pill approach.
Design and setting: Post-hoc observational assessment of Pharmaceutical Benefits Scheme claim records covering the period April 2005 to March 2010.
Participants: A 10% random sample of Australian long-term concession card holders was analysed. The patients studied had commenced on either amlodipine and atorvastatin as two individual pills, or a single pill containing both amlodipine and atorvastatin (AA), with neither combined approach having been dispensed to them in the previous 6 months.
Main outcome measures: The proportions of patients failing to fill their first repeat prescription after 1 month or failing to persist with treatment at 12 months, and the median persistence time (MPT) were measured.
Results: Of 4146 patients prescribed the AA single pill, 11% failed to fill the first repeat prescription and 33% had ceased treatment by 12 months (MPT, 35 months). Of 6204 patients prescribed amlodipine and atorvastatin as two pills, 23% failed to fill the first repeat prescriptions and 59% had ceased treatment by 12 months (MPT, 7 months). In a multivariate model, cessation of single-pill therapy increased by 165% if there was no prior therapy, but only increased by 48%–55% if there was no prior therapy with a calcium channel blocker or statin. MPT on the single pill was 8 months in those without prior antihypertensive therapy, but was ≥ 37 months in those with any prior antihypertensive therapy.
Conclusion: A single-pill combination drug is associated with superior long-term persistence compared with two-pill therapy in the management of hypertension and dyslipidaemia.