Visual field defects typically caused by lesions in the optic nerve or optic chiasm may be caused indirectly by subdural haematomas. A 61-year-old man survived transtentorial herniation caused by subdural haematomas that resulted from shaking his head in an attempt to remove a cockroach impacted in his external auditory canal. Bilateral incomplete posterior cerebral artery infarction of both inferior tips of the occipital lobe resulted in bilateral superior quadrantanopia. There are no previous reports of tentorial herniation causing this permanent outcome. (MJA 2011; 194: 420-422)
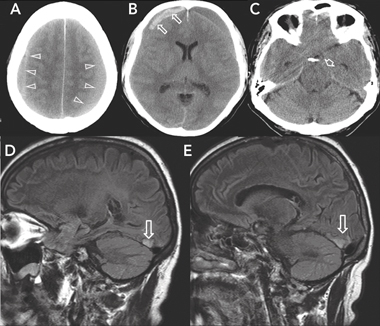
A cerebral computed tomography (CT) scan showed large bilateral frontoparietal subdural haematomas (Box 1, A). The right-sided haematoma had mixed hyperdense and isodense areas suggestive of acute on subacute bleeding, while the left frontal haematoma was isodense. The CT scan also showed marked mass effect (Box 1, B) and early tentorial herniation of the uncus (Box 1, C) without tonsillar descent. There was a delay of 8 hours, including transfer of the patient to a neurosurgery centre, before surgery. The patient’s medical notes showed that before undergoing surgery he had developed bilateral extensor Babinski responses and had a GCS score of 14/15. One hour before the operation, the nursing records indicated “GCS 13/15, patient too drowsy and disoriented to consent [to surgery]”. No further imaging was undertaken and no further notes were made before surgery.
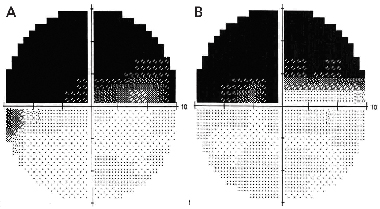
During routine follow-up 10 weeks later, the patient reported persistent difficulty with reading since the operation and that he was experiencing problems differentiating colours. His visual acuity was found to be 6/5 in the right eye and 6/18 in the left, with symptoms of defective colour vision (3/14 Ishihara plates correctly identified with the right eye, and 4/14 with the left). A minor cataract was found in his left eye, but there was no other intraocular abnormality in either eye. His optic discs appeared healthy without pallor in both eyes (and remained so on further follow-up several years later). Computerised visual field perimetry testing conducted at 10 weeks showed bilateral superior altitudinal defects (Box 2). Routine visual evoked potentials showed normal latencies. However, multifocal visual evoked potentials showed normal latencies with decreased amplitude in superior fields bilaterally.
Magnetic resonance imaging (MRI) of the brain and orbits conducted 6 months after the surgery showed chronic infarction involving the inferior tip of both occipital lobes medially, more marked on the right side (Box 1, D and E). There was evidence of residual blood products from the evacuated bilateral subdural haematomas, with no evidence of new bleeding. There was no abnormality of the optic nerves or optic chiasm, and venous drainage on magnetic resonance venography was normal.
Subdural haematoma is an uncommon cause of bilateral homonymous hemianopia.1 It can occur as the result of direct local injury to the occipital lobes, or alternatively, mass effect from subdural haematoma can result in tentorial herniation, potentially compromising the blood supply of the posterior cerebral artery (PCA) and placing the regions it supplies at risk. Visual field defects as a consequence of this indirect effect of subdural haematoma have been reported in some surviving patients, but posterior cerebral infarction is more commonly recognised in postmortem examinations of the brain.2-4
Altitudinal visual field defects are typically due to optic nerve lesions and are usually unilateral. Theoretically, the minimal lesion required to produce a bilateral superior quadrantanopia is one located in the inferior optic chiasm; however, this occurs rarely. Typically, injury causes splaying of the chiasm, which is more commonly associated with bitemporal hemianopia. Transtentorial herniation is a well recognised cause of uncal and brainstem displacement and third cranial nerve palsy, but it can also cause stretching and compression of the PCA — thereby compromising the blood supply and leading to infarction.4,5 Survival from tentorial herniation with residual bilateral homonymous hemianopia has been occasionally reported in the literature.6-9 To date, there is no documentation of it causing bilateral superior quadrantanopia. The regions of the brain at risk secondary to PCA ischaemia or infarction are the inferior and medial temporal and occipital cortices. The actual area of involvement depends on which tributaries (and distributions) are affected. The calcarine branch of the medial occipital artery, which supplies the medial occipital lobe, is most commonly involved.10,11
In our patient, there was evidence of mass effect and tentorial herniation secondary to the enlarging subdural haematoma. This would have placed the PCA blood supply at risk. The MRI findings of bilateral infarction of the inferior tip of the medial occipital lobe, together with field test findings of bilateral superior altitudinal defects, confirm the involvement of the visual cortex, which is topographically organised with the cortical representation of the superior field localised inferiorly.5,12 The affected region corresponds to the striate cortex and most posterior part of the prestriate cortex. Both areas are involved in aspects of colour visual processing, suggesting an explanation for the patient’s defective colour vision despite a normal optic chiasm and optic nerves, although a congenital cause cannot be excluded.13
A diagnosis of acute on subacute subdural haematoma fits well with the timing of the patient’s violent attempts to remove a foreign body from his ear about 7 weeks previously, and with subsequent delayed re-bleeding and increasing supratentorial pressure resulting in uncal herniation and infarction of the part of the primary visual cortex known to correlate with the superior visual fields. A preferred method of removing a cockroach from the ear canal is to drown it with non-toxic vegetable or mineral oil so it can then be easily extracted with forceps. Cautious suction can also be used, taking care not to cause barotrauma by occluding the canal. Lignocaine 2% also rapidly kills the insect or results in it leaving the canal, but entry of lignocaine through any perforation into the middle ear can produce severe sustained vertigo with nystagmus for some hours.14-17
1 Initial computed tomography scan of the brain (A–C), and magnetic resonance images 6 months later (D, E)
Provenance: Not commissioned; externally peer reviewed.
- 1. Soza M, Tagle P, Kirkham T, et al. Bilateral homonymous hemianopia with sparing of central vision after subdural hematoma. Can J Neurol Sci 1987; 14: 153-155.
- 2. Keane JR. Blindness following tentorial herniation. Ann Neurol 1980; 8: 186-190.
- 3. Sato M, Tanaka S, Kohama A, et al. Occipital lobe infarction caused by tentorial herniation. Neurosurgery 1986; 18: 300-305.
- 4. Miller JD, Adams JH. The pathophysiology of raised intracranial pressure. In: Adams JH, Duchen LW, editors. Greenfield’s neuropathology. 5th ed. London: Edward Arnold, 1992: 79.
- 5. Ropper AH. Chapter 268. Coma. In: Fauci AS, Braunwald E, Kasper DL, et al. Harrison’s principles of internal medicine. Harrison’s Online, 17th ed, 2011. http://accessmedicine.com/content.aspx?aid=2888141 (accessed Mar 2011).
- 6. Chungpieboonpatana A, Ratanalert S, Ladpli S. Occipital lobe infarction following descending transtentorial herniation. J Med Assoc Thai 1988; 71: 553-560.
- 7. McOmish D, Pike J. Occipital lobe infarction following descending trans-tentorial brain herniation. Australas Radiol 1981; 25: 9-12.
- 8. Muthukumar N, Gurunathan J, Jawahar G, et al. Occipital lobe infarction caused by tentorial herniation. J Indian Med Assoc 1992; 90: 161.
- 9. Tanaka R, Miyasaka Y, Yada K, et al. Bilateral homonymous hemianopsia due to tentorial herniation, with sparing of central vision: case report. Neurosurgery 1992; 31: 787-790.
- 10. Hoyt WF. Vascular lesions of the visual cortex with brain herniation through the tentorial incisura. Neuro-ophthalmologic considerations. Arch Ophthalmol 1960; 64: 44-57.
- 11. Namura S, Kang Y, Matsuda I, et al. Magnetic resonance imaging of sequelae of temporal lobe herniation secondary to traumatic acute subdural hematoma: Kernohan’s notch and posterior cerebral artery territory infarctions contralateral to the supratentorial lesion — case report. Neurol Med Chir (Tokyo) 1997; 37: 32-35.
- 12. Chapter 15. The visual pathway. Bron AJ, Tripathi RC, Tripathi BJ, editors. Wolff’s anatomy of the eye and orbit. 8th ed. London: Hodder Arnold, 1997: 579-593.
- 13. Hurlbert A. Colour vision: primary visual cortex shows its influence. Curr Biol 2003; 13: R270-R272.
- 14. Ryan C, Ghosh A, Wilson-Boyd B, et al. Presentation and management of aural foreign bodies in two Australian emergency departments. Emerg Med Australas 2006; 18: 372-378.
- 15. O’Toole K, Paris PM, Stewart RD, Martinez R. Removing cockroaches from the auditory canal: controlled trial. N Engl J Med 1985; 312: 1197.
- 16. Cantrell H. More on removing cockroaches from the auditory canal. N Engl J Med 1986; 314: 720.
- 17. Warren J, Rotello LC. Removing cockroaches from the auditory canal: a direct method. N Engl J Med 1989; 320: 322.





None identified.