Methamphetamine (MA) is a highly addictive substance that has caused serious public health problems globally.1,2 Recent population surveys report that 16% of people in the 20–29-year age group have used amphetamines.3 Of particular concern is emerging evidence of serious long-term effects of amphetamine use, including depression, anxiety, psychosis and memory disturbance.4
Neuroimaging studies demonstrate abnormalities in abstinent MA users.5-8 The aim of this study was to determine the prevalence of clinically occult cerebral abnormalities in active amphetamine users presenting to our emergency department (ED).9
All patients received standard clinical management and a mini-mental state examination (MMSE) was performed.10 Detailed data on their drug use were collected. When possible, an MRI was performed during the time of patients’ attendance in the ED, or shortly after discharge. Any patient who was identified on MRI as having an abnormality was referred to the RPH neurology service for assessment.
Thirty-eight patients were enrolled, and 30 had MRI scans. Of these 30 patients, 19 were male, and their mean age was 26.7 years (SD, 5.4 years; range 19–41). The median time from last amphetamine use to ED attendance was 13.6 hours (interquartile range [IQR], 5.8–33.2), and the median time from last use to MRI scan was 2.8 days (IQR, 1.8–4.9). Nineteen patients required admission to hospital. Clinical presentations of the 30 patients who had MRI scans are shown in Box 1.
The mean age at which the 30 patients who did have an MRI scan first started to use amphetamines was 18.0 years (SD, 3.1 years; range 13–26). Twenty-three were classified as serious drug users, and the remainder as recreational users. Box 2 shows a detailed history of their drug use. Other drugs used included cocaine (n = 6), heroin (n = 5), lysergic acid diethylamide (LSD; “acid”) (n = 5) and hallucinogenic mushrooms (n = 1). Twenty-five patients were cigarette smokers (mean usage was 21 cigarettes/day for 11 years), and two were ex-smokers.
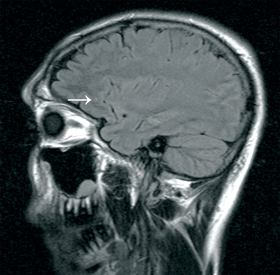
Six patients had abnormalities showing on their MRI scan. Four of these six were male, with a median age of 29 years. The most common MRI abnormality seen (n = 4) was an unidentified bright object (UBO). This is a T2 hyperintensity in subcortical white matter, without a corresponding diffusion-weighted imaging abnormality. Three of the four UBOs seen were in the frontal lobes (Box 3). One patient had cortical atrophy, and one had hippocampal oedema and sclerosis.11 Five of the 23 serious users had abnormalities showing on their MRI scans.
In our pilot study we found that one in five patients had abnormality on their MRI brain scan, the most common being a UBO. Katzman reported only five UBOs as incidental findings in 1000 asymptomatic volunteers (mean age 30.6 years).12 Most UBOs in our study were in the frontal region of the brain; this finding is consistent with reports of frontal abnormalities in MA users.5,13 Whether or not these UBOs are associated with long-term clinical effects is unknown, but our finding supports the possibility of amphetamine use being a risk for cognitive decline, and perhaps an increased risk of stroke.
1 Clinical presentations of the 30 patients who had magnetic resonance imaging scans after using amphetamines
Received 10 November 2009, accepted 22 April 2010
- Daniel M Fatovich1
- David L McCoubrie2
- Swithin J Song3
- David M Rosen4
- Nick D Lawn5
- Frank F Daly6
- Royal Perth Hospital, Perth, WA.
This study was supported by a grant from the RPH Medical Research Foundation. We are grateful for the support of the MRI team.
The RPH Medical Research Foundation funded the MRI scans.
- 1. Gunter TD. Control of methamphetamine misuse. BMJ 2007; 334: 1176-1177.
- 2. United Nations Office on Drugs and Crime. World Drug Report 2009. New York, 2009. http://www.unodc.org/unodc/en/data-and-analysis/WDR-2009.html (accessed Jul 2010).
- 3. Australian Institute of Health and Welfare. 2007 National Drug Strategy Household Survey. Canberra, AIHW: 2008. http://www.aihw.gov.au/publications/index.cfm/title/10579 (accessed Jul 2010).
- 4. Meredith CW, Jaffe C, Ang-Lee K, Saxon AJ. Implications of chronic methamphetamine use: a literature review. Harv Rev Psychiatry 2005; 13: 141-154.
- 5. Berman S, O’Neill J, Fears S, et al. Abuse of amphetamines and structural abnormalities in the brain. Ann N Y Acad Sci 2008; 1141: 195-220.
- 6. Chang L, Alicata D, Ernst T, Volkow N. Structural and metabolic brain changes in the striatum associated with methamphetamine abuse. Addiction 2007; 102: 16-32.
- 7. Ernst T, Chang L, Leonido-Yee M, Speck O. Evidence for long-term neurotoxicity associated with methamphetamine abuse: A 1H MRS study. Neurology 2000; 54: 1344-1349.
- 8. Thompson PM, Hayashi KM, Simon SL, et al. Structural abnormalities in the brains of human subjects who use methamphetamine. J Neurosci 2004; 24: 6028-6036.
- 9. Gray SD, Fatovich DM, McCoubrie DL, Daly FF. Amphetamine-related presentations to an inner-city tertiary emergency department: a prospective evaluation. Med J Aust 2007; 186: 336-339. <MJA full text>
- 10. Dick JP, Guiloff RJ, Stewart A, et al. Mini-mental state examination in neurological patients. J Neurol Neurosurg Psychiatry 1984; 47: 496-499.
- 11. Gardner H, Lawn N, Fatovich DM, Archer JS. Acute hippocampal sclerosis following ecstasy ingestion. Neurology 2009; 73: 567-569.
- 12. Katzman GL, Dagher AP, Patronas NJ. Incidental findings on brain magnetic resonance imaging from 1000 asymptomatic volunteers. JAMA 1999; 282: 36-39.
- 13. Bae SC, Lyoo IK, Sung YH, et al. Increased white matter hyperintensities in male methamphetamine abusers. Drug Alcohol Depend 2006; 81: 83-88.





Abstract
Objectives: To determine the prevalence of occult brain abnormalities in magnetic resonance imaging of active amphetamine users.
Design, setting and participants: Prospective convenience study in a tertiary hospital emergency department (ED). Patients presenting to the ED for an amphetamine-related reason were eligible for inclusion. We collected demographic data, drug use data, and performed a mini-mental state examination (MMSE).
Main outcome measures: The proportion of patients with an abnormality on their MRI scan.
Results: Of 38 patients enrolled, 30 had MRI scans. Nineteen were male and their mean age was 26.7 ± 5.4 years (range 19–41 years). The mean age of first amphetamine use was 18 years (range 13–26 years). Sixteen patients used crystal methamphetamine (mean amount 2.5 g/week), nine used amphetamine (“speed”) (mean amount 2.9 g/week), and 23 used ecstasy (mean amount 2.3 tablets/week). Marijuana was smoked by 26 (mean amount 5.9 g/week), and 28 drank alcohol (mean amount 207 g/week). The median MMSE score was 27/30 (interquartile range, 26–29). Abnormalities on brain MRI scans were identified in six patients, most commonly an unidentified bright object (n = 4).
Conclusion: In this pilot study of brain MRI of young people attending the ED with an amphetamine-related presentation, one in five had an occult brain lesion. While the significance of this is uncertain, it is congruent with evidence that amphetamines cause brain injury.