Trachoma, or “sandy blight”, disappeared from urban Australia in the early 20th century,1 and from regional communities during the 1930s. Despite a chance comment in 1688 by a Dutch navigator, William Dampier, trachoma was not a feature of traditional Aboriginal life.2 Traditional Aboriginal culture is based on good vision and visual representation, not on blindness and visual disability. Trachoma was reported in Aboriginal communities in the 1930s, and studied systematically by Father Frank Flynn, an army chaplain and ophthalmologist in the Northern Territory during the Second World War.3 Dame Ida Mann clearly showed trachoma to be a widespread problem in the 1950s and 1960s.4 The extent of trachoma in Aboriginal communities was further defined by Fred Hollows and the National Trachoma and Eye Health Program (NTEHP) in the 1970s.5
However, without coordinated and sustained effort, trachoma control has limited impact,6 and blinding endemic trachoma still persists in Aboriginal communities7-10 despite the knowledge that has been gathered about its control.11,12
The sampling method has been described in detail elsewhere.13 Briefly, we grouped Australian Bureau of Statistics (ABS) Indigenous Areas (IAREs) into five strata based on the Accessibility/Remoteness Index of Australia:14 “major city”, “inner regional”, “outer regional”, “remote”, and “very remote”. The very remote stratum was then subdivided into “very remote coastal” and “very remote inland”. Within each of these six strata, five sample areas or communities were randomly selected using 2006 ABS census data15 to identify geographical areas containing about 300 Indigenous people. The sample size of 1500 children aged 5 to 15 years and 1500 adults aged 40 years and older was selected based on the ability to detect changes in vision impairment;13 it also conformed to the World Health Organization’s recommendation for the assessment of trachoma of 50 children per cluster.16 Fieldwork was undertaken between January and December 2008.
A standardised questionnaire collected data on demographics, general health, eye health and health service utilisation, and a standardised eye examination was performed.13 Trachoma was graded in each eye using a × 2.5 loupe according to the WHO simple grading system,17 where:
trachomatous inflammation, follicular (TF) was defined as the presence of five or more follicles in the upper tarsal conjunctiva;
trachomatous inflammation, intense (TI) as pronounced inflammatory thickening of the tarsal conjunctiva that obscures more than half of the normal deep tarsal vessels;
trachomatous trichiasis (TT) as the presence of at least one eyelash rubbing on the eyeball;
trachomatous scarring (TS) as the presence of scarring in the tarsal conjunctiva; and
corneal opacity (CO) as easily visible corneal opacity over the pupil.
The trachoma graders were given self-directed trachoma grading training using an interactive CD-ROM.18 Photographs of the everted left tarsal plate were taken with a Nikon D40 digital camera using an 80 mm lens and extension rings (Nikon Corporation, Tokyo, Japan).
All trachoma photographs were graded twice by the same experienced grader using a fine grading system.19 Follicles 1 (F1) was defined as one or two small follicles in the upper tarsal conjunctiva; Follicles 2 (F2) as greater than two but fewer than five follicles of 0.5 mm in diameter in the upper tarsal conjunctiva (ie, less severe than TF); Scarring 1 (S1) as a small amount of early scarring apparent, but not clearly visible; and Scarring 2 (S2) as a moderate amount of early scarring apparent, but not clearly visible (ie, less severe than TS). Grading was performed on a laptop computer without knowledge of the clinical or photograding.
Primary ethics approval was obtained from the Human Research Ethics Committee of the Royal Victorian Eye and Ear Hospital. Further separate formal ethics approval was obtained from the Aboriginal Health and Medical Research Council of New South Wales, the Aboriginal Health Council of South Australia, the Menzies School of Health Research, the Central Australian Human Research Ethics Committee, the Western Australian Aboriginal Health Information and Ethics Committee, ACT Health (12 November 2007), the Tasmanian Scientific Research Advisory Committee, the Tasmanian Health and Medical Human Research Ethics Committee, and the Queensland Aboriginal and Islander Health Council.20 The protocol was also reviewed and approved by the Board of the National Aboriginal Community Controlled Health Organisation and by each community.
The unadjusted rate of active trachoma (TF) in children was 3.8%; after adjustment for sampling, the rate was 2.8%. All the children with TI also had TF, so rates for TF and TI are not presented separately (Box 1). Two adults, both women, had TF; both came from very remote communities, and one also had TI. The prevalence of TF among children in these two communities was 10.1% and 23.0%, respectively. In 14 communities, no children had TF; sporadic single cases of TF occurred in one major city, and one inner regional, two remote, and two very remote coastal sites (Box 2). In 10 communities, two or more children had TF, but the prevalence in five of these communities was less than 5%. However, in five communities, all in the two very remote strata, the prevalence of TF was 5% or greater.
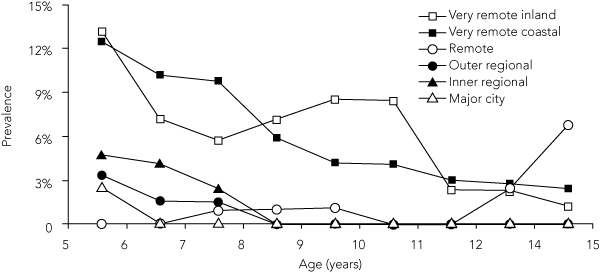
The age-specific rates show a peak prevalence of TF among the youngest children and a progressive overall decrease with increasing age (Box 3). In Australia, the prevalence of TF is usually presented for 5 to 9 year olds;9 862 children aged 5 to 9 years were examined for our study. The overall prevalence of TF in these children mirrored that found among all children 5 to 15 years. However, the prevalence of TF among children aged 5 to 9 years was higher in several communities — six of the 10 very remote communities had a prevalence greater than 5%; the highest was 25.7%. The overall prevalence of TF among children aged 5 to 9 years was 5.2%.
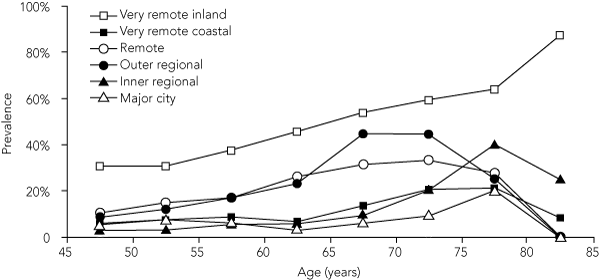
Scarring was uncommon among children, even in communities with a higher prevalence of TF (Box 2). Conversely, TS was common among adults and widely spread, occurring in all strata; in three very remote communities, over half the adults were affected (Box 2). The age-specific rates of scarring increased with age, although in most strata, rates fell in the oldest age groups (Box 4).
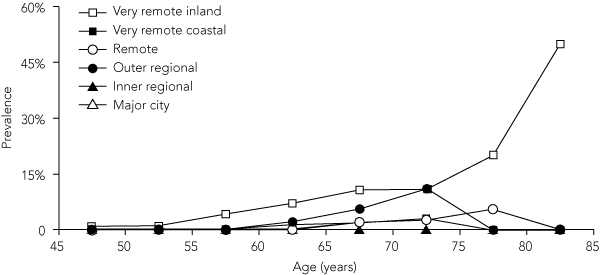
Seven men and nine women had TT. Their ages ranged from 51 to 88 years. Three of these participants also had CO — two women aged 64 and 88 years and a man aged 83 years. One had visual acuity of < 6/18 in one eye and < 6/60 in the other, the second had visual acuity of < 6/60 in both eyes, and the third had light perception in one eye and no light perception in the other. A fourth participant had visual acuity of < 6/12 associated with TT. An additional participant with TT was blind from cataract. The age-specific rates show a gradual increase of TT in the older age groups (Box 5). No child was found to have TT or CO.
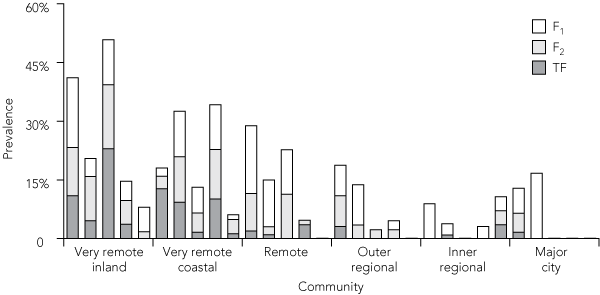
Overall, 6.2% of children had F1, 4.8% had F2 and 3.8% had TF. None of these signs were seen in six communities, and F1 alone was detected in three communities (Box 6). F2 occurred in five communities without TF, although in the remaining 16 communities TF was accompanied by F1 and F2 (Box 6). There was a significant correlation between the presence of F2 and TF (r = 0.72; P < 0.001). There was only a weak, non-significant correlation between F1 and TF (r = 0.34; P = 0.07).
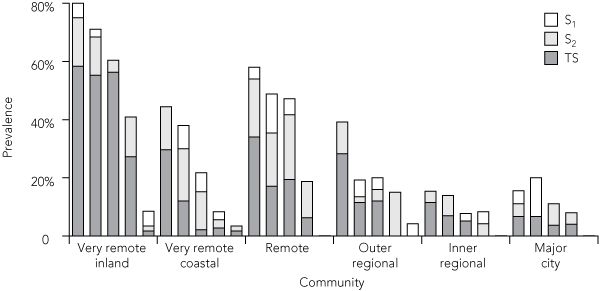
Overall, 3.6% of adults had S1, 8.5% had S2 and 15.7% had TS. In three communities, no adults had any signs of scarring (Box 7). S1 alone was found in one community, S2 occurred alone in one community and together with S1 in another, and TS was present in 24 (Box 7). These fine grading signs were clustered strongly. The correlation between the presence of S2 and TS was strong (r = 0.49; P = 0.006), but there was no correlation between S1 and TS (r = − 0.002; P = 0.99).
These data are the first national trachoma data for Australia since the 1980 report of the NTEHP.5 The overall prevalence of TF among children aged 5 to 15 years reported by the NTEHP was 17.6%. However, 53.9% of TF was the least severe composite grade, “FTC”,5 which was not severe enough to be graded as TF according to our criteria. The adjusted prevalence of TF (“FTA” [the most severe composite grade] and “FTB”) equivalent to TF/TI would be 9.5%. This compares with the overall crude prevalence of 3.8% found in our study. For those aged over 40 years, the NTEHP reported 56.3% had cicatricial trachoma; 40.0% of these cases were graded “CTC”, which would be less severe than TS according to our criteria. The NTEHP also found TT among 6.0% of those over 40 years — of whom 41.0% were blind (2.5% of this age group examined).5 We found national rates for TS of 15.7% and 1.4% for TT. In our small sample, two adults (0.17%) were blind from trachoma.
The rates of trachoma reported from screening of Aboriginal school children in outback communities in the Northern Territory, South Australia and Western Australia in 2006 and 20079,10 are generally similar to our findings.
Scarring was uncommon among children, even as measured by fine grading. Children with scarring were all from remote or very remote communities except for one 11-year-old boy in an urban area with S1, which may not have been related to trachoma. The high rates of TS among adults and the much lower rates of TS among children in the same communities suggest a real decrease in the severity of trachoma. However, without full cross-sectional data, this observation cannot be confirmed. The infrequency of TI among children (11/64 [17%] of those with TF) also suggests a decrease in the severity of TF. In some communities, specific trachoma control activities have been implemented.10 In addition, over the years, there have been obvious improvements in housing, water and sanitation and health care in general, including the ready availability and use of antibiotics to treat childhood infections. If the apparent decrease in severity of trachoma is real, it will make the recent Australian Government commitment to eliminate trachoma much more achievable.21
However, trachoma remains a significant public health problem in many communities, including half of the very remote communities in our study. Significant rates of TT still exist. The observed rate among older adults (1.4%) gives an adjusted population rate of 0.2%, double the WHO threshold.22 Participants with TS who were at risk of TT were found in 24 of the 30 communities, including major cities and regional areas (presumably, these participants grew up in endemic, more remote areas) and participants with TT were found in four of the six strata. This emphasises the general and ongoing need to carefully check older Indigenous people for signs of past trachoma and TT.23 Based on the observed prevalence, we estimate that throughout Australia there are 4778 Indigenous children (5 to 9 years) with TF, 16 314 adults (40 years and older) with TS, 1488 with TT and 273 with CO.
1 Prevalence of active trachoma among Indigenous children (aged 5–15 years) in 30 communities around Australia, 2008
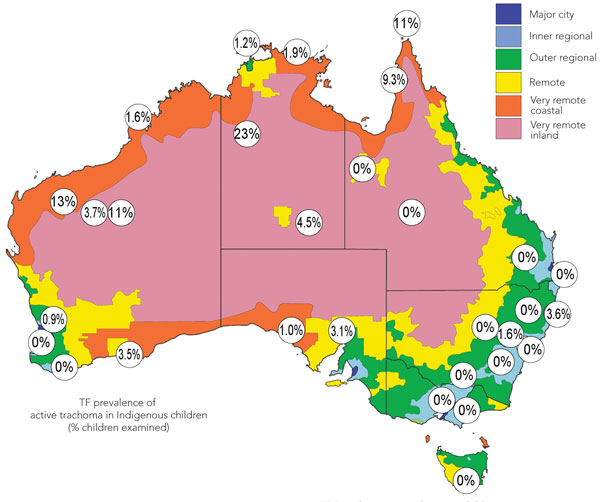
2 Prevalence of trachomatous disease among Indigenous children and adults in 30 communities around Australia, 2008
3 Prevalence of follicular trachomatous inflammation among Indigenous children, by remoteness area (3-year rolling average)
4 Prevalence of trachomatous scarring among Indigenous adults, by remoteness area (15-year rolling average)
5 Prevalence of trachomatous trichiasis among Indigenous adults, by remoteness area (15-year rolling average)
Received 23 July 2009, accepted 9 November 2009
- Hugh R Taylor1,2
- Sarah S Fox2,3
- Jing Xie2,3
- Ross A Dunn2,3
- Anna-Lena M R Arnold2,3
- Jill E Keeffe2,3
- 1 Melbourne School of Population Health, University of Melbourne, Melbourne, VIC.
- 2 Vision Cooperative Research Centre, Sydney, NSW.
- 3 Centre for Eye Research Australia, University of Melbourne, Melbourne, VIC.
We acknowledge the contributions of the National Indigenous Eye Health Survey Advisory Committee (Professor Michael Wooldridge, Professor Ian Anderson, Sandra Bailey, Stephanie Bell, Joe Chakman, Amanda Davis, Dr William Glasson, Mick Gooda, Elissa Greenham, Belinda Sullivan, Professor Debbie Sweeney), the Steering Committee (Dr Robert Casson, Rowan Churchill, Associate Professor Mark Gillies, Dr William Glasson, Dr Timothy Henderson, Professor Brian Layland, Dr Mark Loane, Associate Professor Ian McAllister, Dr Tharmalingam Mahendrarjah, Dr Richard Mills, Dr Nigel Morlet, Helen Morrissey, Dr Henry Newland, Dr Richard Rawson, Gerd Schlenther), and thank Mitasha Marolia, Anthea Burnett, Dr Tomer Shemesh, the many Aboriginal health workers and eye health coordinators who assisted with this, especially Barbara O’Connor, Desley Culpin and Phyllis Tighe; and the many volunteers who helped with the field work, including Dr Jan Lovie Kitchen, Nora Ley and Carmel McInally.
None identified.
- 1. Barrett JW, Orr WF. Trachoma in the state of Victoria. Intercolonial Med J 1909; 20 Sep 20: 450-455.
- 2. Taylor HR. Trachoma: a blinding scourge from the bronze age to the twenty-first century. Melbourne: Centre for Eye Research Australia, 2008.
- 3. Flynn F. Trachoma among natives of the Northern Territory of Australia. Med J Aust 1957; 11: 269-277.
- 4. Mann I. Culture, race, climate and eye disease. Springfield, Ill: Charles C Thomas, 1966.
- 5. Royal Australian College of Ophthalmologists. The National Trachoma and Eye Health Program of the Royal Australian College of Ophthalmologists. Sydney: RACO, 1980.
- 6. Taylor HR. Eye health in Aboriginal and Torres Strait Islander communities. Canberra: Commonwealth of Australia, 1997.
- 7. Stocks NP, Hiller JE, Newland H, McGilchrist CA. Trends in the prevalence of trachoma, South Australia, 1976 to 1990. Aust N Z J Public Health 1996; 20: 375-381.
- 8. Mak DB, O’Neill LM, Herceg A, McFarlane H. Prevalence and control of trachoma in Australia, 1997–2004. Commun Dis Intell 2006; 30: 236-247.
- 9. Tellis B, Keeffe JE, Taylor HR. Surveillance report for active trachoma, 2006: National Trachoma Surveillance and Reporting Unit. Commun Dis Intell 2007; 31: 366-374.
- 10. Tellis B, Keeffe JE, Taylor HR. Surveillance report for active trachoma, 2007: National Trachoma Surveillance and Reporting Unit. Commun Dis Intell 2008; 32: 388-399.
- 11. Taylor HR. Trachoma in Australia. Med J Aust 2001; 175: 371-372. <MJA full text>
- 12. Mak D. Guidelines for the public health management of trachoma in Australia. Canberra: Communicable Disease Network Australia, 2006.
- 13. Fox SS, Arnold A-L, Keeffe JE, Taylor HR. Sampling and recruitment methodology for a National Eye Health Survey of Indigenous Australians. Aust N Z J Public Health 2010. In press.
- 14. Australian Bureau of Statistics. Australian Standard Geographical Classification (ASGC) — electronic publication, 2005. (ABS Cat. No. 1216.0.) http://www.abs.gov.au/AUSSTATS/abs@.nsf/lookup/0D204FD3DCD90564CA256 F19001303A2?opendocument (accessed Jan 2010).
- 15. Australian Bureau of Statistics. Experimental estimates of Aboriginal and Torres Strait Islander Australians, Jun 2006. (ABS Cat. No. 3238.0.55.001.) http://www.abs.gov.au/AUSSTATS/abs@.nsf/Lookup/3238.0.55.001Main+ Features1Jun%202006? (accessed Jan 2010).
- 16. Negrel AD, Taylor HR, West S. Guidelines for the rapid assessment for blinding trachoma. Geneva: World Health Organization, 2001. (Report No. WHO/PBD/GET/00.8.)
- 17. Thylefors B, Dawson CR, Jones BR, et al. A simple system for the assessment of trachoma and its complications. Bull World Health Organ 1987; 65: 477-483.
- 18. Wright HR, McDonnell C, Squire S, et al. Trachoma grading — self directed learning [CD-ROM]. Melbourne: Centre for Eye Research Australia, 2007.
- 19. Roper KG, Taylor HR. Comparison of clinical and photographic assessment of trachoma. Br J Ophthalmol 2009; 93: 811-814.
- 20. Taylor HR, Fox SS. Ethical hurdles in Indigenous research. Aust N Z J Public Health 2008; 32: 489-490.
- 21. Kirby T. Nicola Roxon: Australia’s Minister for Health and Ageing. Lancet 2009; 373: 1073.
- 22. World Health Organization. Report of the 2nd Global Scientific Meeting on Trachoma. 2003; Aug 25-27; Geneva.
- 23. Taylor HR. Trichiasis: out of mind, out of sight. Aust J Rural Health 2009; 17: 171.





Abstract
Objective: To determine the prevalence of trachoma among Indigenous Australians.
Design, setting and participants: A national, stratified, random cluster sample survey of Indigenous children (5–15 years) and adults (≥ 40 years) in 30 communities across Australia. Data collection was undertaken in 2008.
Main outcome measures: Results based on a standardised protocol that included trachoma grading and double grading of photographs of the tarsus.
Results: 1694 Indigenous children and 1189 Indigenous adults were examined. Recruitment rates were 84% for children and 72% for adults. The overall rate of follicular trachomatous inflammation among children was 3.8%, ranging from 0.6% in major cities to 7.3% in very remote areas; 50% of communities in very remote areas had endemic rates (> 5%). Trachomatous scarring (TS) occurred among 15.7% of adults, trachomatous trichiasis (TT) among 1.4% and corneal opacity (CO) among 0.3%. TS was found in all regions and TT in all except major cities and inner regional areas. The highest community rates for TS were 58.3%; for TT, 14.6%; and for CO, 3.3%.
Conclusion: Blinding endemic trachoma remains a major public health problem in many Aboriginal and Torres Strait Islander communities. Although active trachoma is predominantly seen in very remote communities, scarring and blinding sequelae occur among Indigenous people across the country. The Australian Government’s recent commitment to eliminate blinding trachoma is welcomed and much needed.