Minority groups in developed countries, including Indigenous Australians,1-3 have poorer asthma outcomes than the general population, with higher rates of emergency department visits and asthma-related deaths.1 Asthma education for patients, including information on medications and written plans for responding to changes in asthma symptoms, is essential for improving asthma outcomes.4-6 Child-specific asthma education can increase management skills, reduce symptoms, and improve school performance.7,8 Specific education programs (eg, home-based, or culture-specific) are likely to be more effective then generic ones.9
While it is accepted worldwide that Indigenous health care workers (IHCWs) play an important role in educating Indigenous people about illnesses,9,10 no controlled studies have examined the effect on health outcomes of interventions conducted by IHCWs. We have reported previously that children with asthma in the Torres Strait region of northern Australia have more severe disease than children in urban areas.11 Building on our previous work in this community,11,12 we conducted a randomised controlled trial of an education intervention by IHCWs for children with asthma.
Spirometry was performed in the standing position using a noseclip and a spirometer (calibrated daily) approved by the American Thoracic Society. Predicted values of Hibbert and colleagues were used.13 Clinical asthma was defined as repeated episodes of wheeze with dyspnoea that responded to bronchodilators. In children aged 3–6 years, two or more episodes of wheezy illness associated with cough and shortness of breath, and documented amelioration of symptoms and clinical signs after administration of a bronchodilator, supported a diagnosis of asthma.14 Severity of asthma was classified as persistent, frequent episodic, or infrequent episodic, based on the clinical pattern in the past 12 months.14
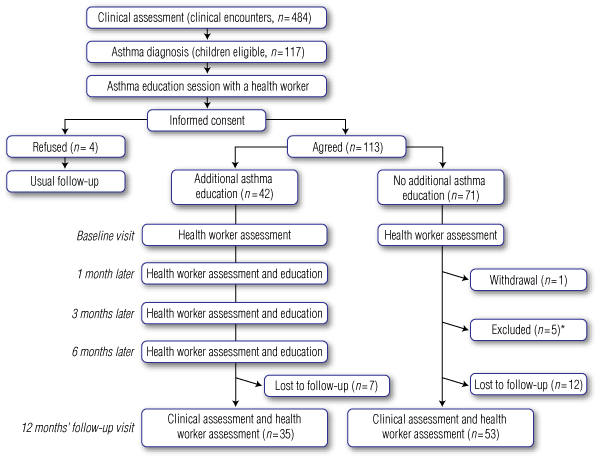
Before enrolment, all children had an asthma education session with a trained IHCW using the adapted asthma booklets. The intervention group had three additional education sessions with a trained IHCW at 1, 3 and 6 months after the baseline visit, using the same educational resources. Adherence to the study protocol (Box 1) was monitored by checking data collected during these visits. Control group children were not visited by the IHCWs. At 12 months, all children completing the study had a follow-up clinical consultation.
The primary endpoint was the number of unscheduled visits to hospital or a doctor because of asthma exacerbation in the 12 months’ follow-up. Secondary analysis focused on measures of quality of life (QoL)15,16 and the functional severity index for asthma;17 asthma knowledge and use of asthma action plans (AAPs); and school days missed because of wheezing.
Data on all acute exacerbations of asthma requiring treatment in a health facility were collected from the child’s medical records (at health clinics and Thursday Island Hospital) from 12 months before to 12 months after each child’s baseline visit. Data for secondary outcomes were collected at the baseline visit and at the 12 months’ follow-up, and also at each education session in the intervention group. Children and their carers were asked about the written AAP and what to do if asthma symptoms worsen. Carers of children taking regular medication were assessed for basic knowledge of asthma medications (eg, dosage, how the drugs work) and drug delivery technique.11 The functional severity index,17 a measure of functional impairment caused by asthma over a period of 12 months was scored (total score, 0–24; higher scores indicate worse impairment), and children and carers completed the Paediatric Asthma Quality of Life Questionnaire.15,16
Consecutive patients with a confirmed asthma diagnosis were eligible for our study. At the clinic where eligibility was assessed, informed consent was obtained from parents or guardians, and children were randomly allocated to the intervention group (additional education sessions), or to the control group (no additional education sessions) (Box 1). A randomly generated list (using a computer-generated permuted block design) within age strata (< 7 and ≥ 7 years) was used for study allocation; allocation group was revealed after enrolment. Staff collecting data from the medical records during the follow-up did not know the children and were blinded to the study allocation; other clinical study staff and the children were aware of the study allocation.
As study enrolment was slower than anticipated, we modified the intervention allocation based on estimates of asthma prevalence12 and guided by the sample size and power calculations. If a child was allocated to “additional education” but there was no trained IHCW in the child’s community to administer the intervention, we changed the allocation to “no additional education” (n = 7). If “no additional education” was not possible, either because a sibling had been allocated to “additional education” (n = 8) or the IHCW at the community health centre was familiar with the intervention (n = 5), we changed allocation to “additional education”.
To check whether bias was introduced by per-protocol analysis instead of intention-to-treat analysis, we conducted multivariate analysis including the variables “treatment allocation as per protocol”, “randomisation” (randomised v non-randomised), the interaction term “randomisation”*“treatment allocation as per protocol”, and “asthma severity at baseline”, and for each outcome variable. As P > 0.05 applied to the interaction term in all models run (ranging from 0.093 for carers’ knowledge of asthma medication to 0.999 for carers’ knowledge of how preventers and relievers work), we proceeded with per-protocol analysis without adjusting for randomisation. As siblings from the same families were included in the study (11 children), we repeated the analysis including one randomly chosen child from each family. The results of subgroup analysis were very similar to those of the whole sample, so we have only presented data for the whole cohort.
We enrolled 113 of 117 (97%) eligible children (aged 1–17 years) between April 2005 and March 2007, five were excluded for practical reasons (Box 1): 88 children (81%) with completed follow-up are included here; 35 in the additional education group and 53 in the control group; 98% were Indigenous children. Follow-up was completed by March 2008.
The intervention and control groups were comparable at baseline (Box 2 and Box 3) except for asthma severity, which appeared to be worse in the control group. There were no significant differences between the intervention and control groups in ethnicity, or parents’ highest level of education. One in 10 children possessed an AAP, two-thirds of the carers could not name their child’s medication, half did not know the dosage, and most could not explain how asthma medications worked (Box 3).
Half the children (52%) had no asthma episode that required a visit to hospital or a doctor in the follow-up period (Box 4). The median number (range) of unscheduled hospital or doctor visits was 1.0 (0–4) for the intervention group and 0 (0–4) for controls (P = 0.25). There were eight asthma-related hospital admissions (four in each group). The mean difference between the number of episodes of asthma for the intervention group (mean, 1.0) and controls (mean, 0.7) was 0.30 (95% CI, 0.22–0.39).
In the intervention group, compared with the control group, there was a significant improvement in asthma knowledge, and more carers knew where their child’s AAP was kept and were able to interpret it (Box 5); more intervention group carers could accurately recall their child’s medication dosage (difference, 37%; 95% CI, 19%–54%), knew how β2 agonists worked (difference, 55%; 95% CI, 38%–72%), and knew how preventers and relievers worked (difference, 31%; 95% CI, 11%–51%). They also reported having used their AAP (difference, 32%; 95% CI, 4%–60%) and could describe it accurately (difference, 29%; 95% CI, 9%–49%).
Among the whole group, there was a decrease in the number of unscheduled hospital or doctor visits from the 12-month period before (median [range], 1.0 [0–7]) to the 12-month period after study entry (median, 0.0 [0–4]; P = 0.038) (Box 4). Likewise, the median (range) functional severity index score for the total cohort was reduced from 9.0 (0–22) to 5.0 (0–24); (P < 0.01). The median (range) QoL scores for carers increased from 5.2 (1.9–7.0) to 6.8 (1.9–7.0); (P < 0.01), and the QoL scores for children increased slightly from 6.0 (2.6–7.0) to 6.9 (2.5–7.0); (P = 0.053). These results were similar when the intervention and control groups were analysed separately.
Several studies have shown that asthma education improves asthma outcomes: self-management education reduced hospitalisations, emergency room visits, days off work or school, and improved QoL scores.6 A review of four studies examined the effect of culture-specific programs on asthma outcomes in ethnic minorities and found significantly improved QoL measures and asthma knowledge, as well as significantly reduced hospital or emergency department visits.9 Our findings for improvement in asthma knowledge are consistent with this review, but in our study there was no change in clinic presentations. This may relate to sample size, or our inclusion of all grades of asthma; children with severe disease may have benefited more from asthma education provided by IHCWs.
To our knowledge, this is the only culture-specific study in an affluent country of an education intervention by IHCWs for Indigenous children. It provides a model for addressing the gap in health outcomes between Indigenous and non-Indigenous Australians. We have previously raised the issue of inequity in asthma management in children in the Torres Strait.11
The before-and-after differences in QoL score, functional severity index and number of asthma exacerbations are consistent with findings from other studies evaluating asthma programs delivered in relevant dialects or using health workers with the same ethnic background as the patients.18-20 The change in QoL score from 5.2 to 6.8 is clinically important (the minimum important difference for the questionnaire we used is 0.5) and ≥ 1.5 is classified as a large change.21 The decrease in functional severity index was also a clinically important indication of a reduction in asthma severity.
Our response and follow-up rates were high, limiting the potential for selection bias. We took steps to reduce recall bias by using face-to-face interviews and a standardised protocol for data collection.11,15-17 Our study had limited power to detect small differences between the groups with certainty; consequently, there may have been differences that the study did not detect, and we did not achieve our planned sample size despite the change in allocation. Moreover, the effect of random measurement error will tend to bias estimates towards the null, which should be borne in mind when interpreting weak and statistically non-significant associations. The open nature of our study was unavoidable and, although it is unlikely that the pattern of care delivered by local clinic staff changed, it is possible that families in the control group became aware of the intervention. Given the nature of the setting (small Indigenous communities), it would be inevitable that trained IHCWs would educate any family they had contact with (not just intervention group families), leading to “cross-group contamination”. Also, all children had an “intervention” at baseline: the specialist assessment and an education session with a trained IHCW. This potentially diminished the impact of additional asthma education sessions by IHCWs. We suspect these factors also contributed to the differences in the before-and-after comparison being more marked than those between the intervention and control groups.
The non-significant unequal distribution in asthma severity between the intervention and control groups (Box 2) was a limitation. This was caused mostly by children from outer islands generally having more severe asthma. As we could not feasibly conduct the study in the outer islands, these children were re-allocated to the control group.
4 Number of visits to hospital or a doctor because of wheezing — 12 months before and after study entry
- Patricia C Valery1
- Ian B Masters2
- Brett Taylor3
- Yancy Laifoo4
- Peter K O’Rourke1
- Anne B Chang5,6
- 1 Queensland Institute of Medical Research, and Australian Centre for International and Tropical Health, University of Queensland, Brisbane, QLD.
- 2 Department of Respiratory Medicine, Royal Children’s Hospital, Brisbane, QLD.
- 3 The Asthma Foundation of Queensland, Brisbane, QLD.
- 4 Thursday Island Primary Health Care Centre, Thursday Island, QLD.
- 5 Menzies School of Health Research, Charles Darwin University, Darwin, NT.
- 6 Queensland Children’s Respiratory Centre and Queensland Children’s Medical Research Institute, Royal Children’s Hospital, Brisbane, QLD.
We thank Valerie Logan for technical support; the District Health staff, Saimo Gela and Debra Nona, for help with data collection; and Janelle Stirling (former coordinator of the Queensland Institute of Medical Research Indigenous Health Research Program), Vanessa Clements and Torres Strait personnel for their help with culturally appropriate, paediatric asthma education packages. We also thank Gail Garvey (coordinator of the Queensland Institute of Medical Research Indigenous Health Research Program). We are grateful to the children and their families for participating in the study. We also acknowledge the support of the Torres Strait communities and the District Health Council — Torres Strait Health and Northern Peninsula Area, Queensland Health.
None identified.
- 1. Mannino DM, Homa DM, Akinbami LJ, et al. Surveillance for asthma — United States, 1980–1999. MMWR Surveill Summ 2002; 51: 1-13.
- 2. Anderson HR, Gupta R, Strachan DP, Limb ES. 50 years of asthma: UK trends from 1955 to 2004. Thorax 2007; 62: 85-90.
- 3. Australian Centre for Asthma Monitoring. Asthma in Australia 2008. Canberra: Australian Institute of Health and Welfare, 2008. (AIHW Asthma Series No. 3, Cat. No. ACM 6.)
- 4. British guideline on the management of asthma: a national clinical guideline. Edinburgh: Scottish Intercollegiate Guidelines Network, 2008.
- 5. Global Initiative for Asthma. Global strategy for asthma management and prevention 2008 (update). 2008. http://www.ginasthma.org (accessed Apr 2010).
- 6. Gibson PG, Powell H, Coughlan J, et al. Self-management education and regular practitioner review for adults with asthma. Cochrane Database Syst Rev 2003; (1): CD001117.
- 7. Evans D, Clark NM, Feldman CH, et al. A school health education program for children with asthma aged 8-11 years. Health Educ Q 1987; 14: 267-279.
- 8. Brown JV, Bakeman R, Celano MP, et al. Home-based asthma education of young low-income children and their families. J Pediatr Psychol 2002; 27: 677-688.
- 9. Bailey EJ, Cates CJ, Kruske SG, et al. Culture-specific programs for children and adults from minority groups who have asthma. Cochrane Database Syst Rev 2009; (2): CD006580.
- 10. Ratima M, Fox C, Fox B, et al. Long-term benefits for Maori of an asthma self-management program in a Maori community which takes a partnership approach. Aust N Z J Public Health 1999; 23: 601-605.
- 11. Chang A, Shannon C, O’Neil M, et al. Asthma management in indigenous children of a remote community using an indigenous health model. J Paediatr Child Health 2000; 36: 249-251.
- 12. Valery P, Chang A, Shibasaki S, et al. High prevalence of asthma in five remote indigenous communities in Australia. Eur Respir J 2001; 17: 1089-1096.
- 13. Hibbert ME, Lannigan A, Landau LI, Phelan PD. Lung function values from a longitudinal study of healthy children and adolescents. Pediatr Pulmonol 1989; 7: 101-109.
- 14. Phelan PD, Olinsky A, Oswald H. Asthma: classification, clinical patterns and natural history. In: Phelan PD, ed. Clinical paediatrics. Vol. 3. London: Bailliere Tindall, 1995: 307-318.
- 15. Juniper EF, Guyatt GH, Feeny DH, et al. Measuring quality of life in children with asthma. Qual Life Res 1996; 5: 35-46.
- 16. Juniper EF, Guyatt GH, Feeny DH, et al. Measuring quality of life in the parents of children with asthma. Qual Life Res 1996; 5: 27-34.
- 17. Rosier M, Bishop J, Nolan T, et al. Measurement of functional severity of asthma in children. Am J Respir Crit Care Med 1994; 149: 1434-1441.
- 18. Moudgil H, Marshall T, Honeybourne D. Asthma education and quality of life in the community: a randomised controlled study to evaluate the impact on white European and Indian subcontinent ethnic groups from socioeconomically deprived areas in Birmingham, UK. Thorax 2000; 55: 177-183.
- 19. Blixen CE, Hammel JP, Murphy D, Ault V. Feasibility of a nurse-run asthma education program for urban African-Americans: a pilot study. J Asthma 2001; 38: 23-32.
- 20. D’Souza W, Crane J, Burgess C, et al. Community-based asthma care: trial of a “credit card” asthma self-management plan. Eur Respir J 1994; 7: 1260-1265.
- 21. Juniper EF, Guyatt GH, Willan A, Griffith LE. Determining a minimal important change in a disease-specific Quality of Life Questionnaire. J Clin Epidemiol 1994; 47: 81-87.





Abstract
Objective: To assess the outcomes of an education intervention for childhood asthma conducted by Australian Indigenous health care workers (IHCWs).
Design and setting: Randomised controlled trial in a primary health care setting on Thursday Island and Horn Island, and in Bamaga, Torres Strait region of northern Australia, April 2005 to March 2007.
Participants: 88 children, aged 1–17 years, with asthma diagnosed by a respiratory physician (intervention group, 35; control group, 53; 98% Indigenous children).
Interventions: Children were randomly allocated to: (i) three additional asthma education sessions with a trained IHCW, or (ii) no additional asthma education. Both groups were re-assessed at 12 months.
Main outcome measures: Primary endpoint: number of unscheduled visits to hospital or a doctor caused by asthma exacerbation. Secondary outcomes: measures of quality of life (QoL) and functional severity index; asthma knowledge and understanding of asthma action plans (AAPs); and school days missed because of wheezing.
Results: The groups were comparable at baseline (except for asthma severity, which was adjusted for in the analysis). There were no significant differences in the primary outcome (number of unscheduled medical visits for asthma). School children in the intervention group missed fewer school days because of wheezing (100% < 7 days v 21% of those in the control group missed 7–14 days). Significantly more carers in the intervention group could answer questions about asthma medication, knew where their AAP was kept (84% v 56%), and were able to describe the plan (67% v 40%). In both the intervention and control groups (before-and-after comparison), there was a significantly reduced frequency of asthma exacerbations, as well as an improved QoL score and functional severity index, with no significant differences between the groups.
Conclusions: A community-based asthma education program conducted by trained IHCWs improves some important asthma outcomes in Indigenous children with asthma.
Trial registration: Australian Clinical Trials Registry ACTRN012605000718640.