It is conservatively estimated that between a third and half of patients hospitalised for decompensated heart failure have heart failure with preserved systolic function (HFPSF).1 Despite the prevalence of this syndrome, there are no robust, generally agreed diagnostic criteria for diastolic heart failure. International definitions of HFPSF2-4 define patients as having symptoms (shortness of breath, fatigue, orthopnea) or clinical signs of fluid retention (pulmonary, abdominal, or peripheral), and normal to near-normal systolic function on echocardiography (left ventricular ejection fraction [LVEF] > 45%),2 with clinical improvement in response to conventional treatment for chronic heart failure (CHF), if the diagnosis is in doubt.
Patients with HFPSF have a distinct demographic profile, aetiological background and pathophysiology.1,5 Compared with patients who have a low ejection fraction, previous studies suggest that those with HFPSF are generally older, are more often women, and are more likely to have CHF of hypertensive aetiology.6,7
HFPSF also has a different natural history compared with heart failure with left ventricular systolic dysfunction (LVSD), despite producing a similar symptom burden and mortality.1,8 The multiple comorbid conditions frequently associated with HFPSF contribute substantially to the risk of hospitalisation as HFPSF progresses.8,9
To date, most of the data on the epidemiology and natural history of HFPSF, and the associated disease burden, have come from clinical trials.7 Treatment for HFPSF remains largely empirical, as most drugs and devices that have been shown to reduce morbidity and mortality in heart failure have predominantly been tested in patients with LVSD.7
Accordingly, we undertook to characterise the demographics, pharmacotherapy, readmission rates and survival of patients with HFPSF from clinical data collected over a period of 10 years.
We performed a retrospective analysis of longitudinal clinical data collected during routine management from a cohort of 2961 patients admitted to a large tertiary referral hospital with a diagnosis of CHF. Patients with CHF who were admitted to the general medical or cardiology units between 1 July 1994 and 30 June 2004 were included. Follow-up was performed from index admission to either death or study census (30 June 2005). The data included were for all index and subsequent admissions for eligible patients.
For the purpose of this study, CHF was defined according to recently published international guidelines.6-8 About 1000 casenotes for the cohort were reviewed and scored for the diagnosis of CHF using the Framingham criteria.10 This confirmed the specificity of ICD codes for principal diagnosis of CHF as being greater than 99%. Hence, the ICD codes for CHF were accepted for the remainder of the cohort.
Echocardiography data were derived from the hospital’s echocardiogram database. Standard biplane LVEF was calculated by means of Simpson’s method of disc,11 following manual tracing of endocardial borders. Preserved systolic function was defined as an LVEF over 45%2 or, in the absence of a quantitative assessment, a subjective report of normal left ventricular size and systolic function.
We used the following definitions for hospitalisations:
Length of stay — the number of days the patient occupied a bed, inclusive of admission and discharge dates.
CHF-related hospitalisations — identified by a discharge diagnosis coding of CHF in either the first or second diagnostic position for an unplanned hospitalisation.12
All-cause hospitalisations — all unplanned admissions to hospital for any cause.
Comparative baseline mortality and population data were obtained from the South Australian Department of Health.
Of the 2961 patients admitted with CHF during the 10-year admission period of this study, there were 753 with echocardiograms available for analysis. Of these, 189 (25%) showed normal left ventricular size and systolic function; these patients comprised the HFPSF cohort, while the remaining 564 were the LVSD cohort.
Box 1 shows that the mean age of patients in the HFPSF cohort was 2 years older than that of patients in the LVSD cohort.
Patients with HFPSF were much more likely to be women, and were more likely to have fewer social supports, less likely to be married and more likely to be widowed, less likely to live in their own home and more likely to be living in a nursing home (Box 1).
Box 2 shows that patients with HFPSF were less likely than those with LVSD to be prescribed an angiotensin-converting enzyme inhibitor, carvedilol, or an angiotensin II receptor antagonist. Overall, patients with HFPSF were just as likely as those with LVSD to be prescribed any other β-blockers, or spironolactone. Despite the greater prevalence of atrial fibrillation in the HFPSF cohort, prescription of digoxin did not differ between the two cohorts (Box 2).
The mean follow-up period for all 753 patients with CHF was 4.1 years (SD, 0.13 years). Despite the HFPSF group having more prognostically significant comorbid conditions, such as anaemia and atrial fibrillation, there was no significant difference in patient survival between groups (P = 0.912; Box 3).
Box 4 shows that, after controlling for sex and preserved systolic function, multivariate predictors of survival included left ventricular size, and the presence of multiple additional comorbid conditions (dementia, renal failure, cerebrovascular disease, peripheral vascular disease, chronic obstructive pulmonary disease). Younger age (< 75 years; hazard ratio [HR], 0.033; P < 0.001) and a history of hypertension (HR, 0.004; P = 0.004) were associated with improved survival.
There was an increase in the risk of re-hospitalisation among patients with HFPSF, with a median of two admissions per year compared with a median of 1.5 admissions per year for patients with LVSD (Box 1).
Patients with HFPSF were more likely than those with LVSD to be admitted for diabetes (P < 0.001), anaemia (P < 0.001) and malignancy (P < 0.001), whereas patients with LVSD were more likely to have admissions directly related to heart failure (P < 0.001). There was no significant difference in the prevalence of hypertension between the LVSD and the HFPSF groups (P = 0.23). Patients with HFPSF had a higher echocardiographic prevalence of left ventricular hypertrophy (46.0% v 29.2%; P < 0.001). There were no statistical differences between groups in any other diagnostic category of admission coding (Box 1).
We undertook this study to characterise the demographic characteristics, pharmacotherapy, readmission rates and survival of patients with HFPSF from clinical data collected for patients admitted over a period of 10 years. The demographic characteristics of our HFPSF group closely mirrored those reported in other epidemiological studies. In particular, patients with HFPSF were more likely to be women and more likely to be older (age, > 75 years).6,7
Importantly, our study is the first to identify significant differences in the social environment of these patients. Specifically, patients with HFPSF were less likely to live in their own home, or have a spouse. Lack of social and carer support may have played a significant predisposing role in hospitalisation and readmissions.14,15
Patients with HFPSF had a higher prevalence of associated comorbid conditions, such as anaemia and atrial fibrillation, and such conditions have the potential to influence readmission and survival. Comorbid conditions may also have played a role in the therapeutic decision making of clinicians, potentially adversely impacting the intensity of therapy.16
Other studies have shown that patients with HFPSF were more likely to have hypertensive aetiology,7 but we found no significant difference in the prevalence of hypertension between the LVSD and the HFPSF groups. The higher echocardiographic prevalence of left ventricular hypertrophy among HFPSF patients suggests that this result may be due to variability in the coding of hypertension, rather than accurately reflecting the true historical prevalence of this condition in each cohort.
We observed that patients with HFPSF received less intensive pharmacological management than patients with LVSD. The evidence for pharmacotherapy in HFPSF is less rigorous than in LVSD but there are recognised studies supporting the importance of HFPSF pharmacotherapies.2-4 Patients with HFPSF were less likely to be prescribed an angiotensin-converting enzyme inhibitor, despite such agents having been shown to reduce hospitalisation rates and improve functional capacity in patients with HFPSF in randomised clinical trials.17
It is well appreciated that β-blockers have numerous theoretical benefits in patients with HFPSF, including lowering of heart rate (potentially improving diastolic filling and coronary blood flow, particularly during activity), reduction in myocardial oxygen demand and, by lowering the blood pressure, regression of left ventricular hypertrophy.18 Heart rate reduction is known to be particularly important in the treatment of pulmonary congestion as a result of diastolic heart failure secondary to ischaemia, and in patients with atrial fibrillation,19 and has recently been identified as an important determinant of β-blocker efficacy in the prevention of early mortality in LVSD.
As with β-blockers, atrial fibrillation may also have contributed to the similar prescription rates for digoxin in both patients with HFPSF and LVSD, despite concerns that have been raised about the use of digoxin in HFPSF. Classically, digitalis glycosides are known to increase intracellular calcium concentrations, potentially impairing myocardial relaxation and further worsening diastolic dysfunction. It is possible, however, that the sympatho-inhibitory, pro-parasympathetic and renin, angiotensin and aldosterone suppressing actions of digoxin are beneficial in HFPSF, but this remains to be proven in the clinical setting.20
We observed a higher rate of hospital readmission in the HFPSF cohort compared with the LVSD cohort. Previous reports, however, have identified readmission rates that were lower12,21,22 or comparable6,23,24 to those of patients with LVSD. Notably, our HFPSF cohort was older, with less social support, and a greater burden of comorbid conditions, so readmission cannot be attributed solely to differences in cardiac systolic function.
It has been suggested that HFPSF confers a better prognosis than heart failure with impaired systolic function in terms of morbidity and mortality. Studies that examined cohorts comparable to those in our study (mean age, > 65 years) have reported similar mortality rates among patients with HFPSF and LVSD.6,23,24 On the other hand, previous studies in younger populations (mean age, < 65 years) have shown better survival for HFPSF than LVSD.6,23,24 Whether such differences relate to differences in the relative malignancy of the underlying aetiology among younger LVSD patients, or are the result of an excess burden of morbidity and mortality related to comorbid illnesses and social isolation in the older HFPSF population remains uncertain.
In older patients, the clinical syndrome of CHF carries a uniformly poor prognosis regardless of the level of systolic function. Our findings, and those of similar studies, serve to heighten awareness of the prognostic impact of HFPSF, particularly among older patients.7
Numerous guidelines now indicate that Doppler-based parameters (eg, the ratio of peak early-diastolic transmitral flow velocity to peak early-diastolic mitral annular velocity [E/E' ratio] and pulmonary vein velocities) may be used to further clarify the diagnosis of diastolic impairment in the diagnosis of HFPSF. However, our study has shown a significant independent survival disadvantage, regardless of these Doppler-based parameters, when clinical findings of CHF are associated with normal systolic left ventricular function. It remains to be determined whether these Doppler echocardiographic parameters, coupled with novel diagnostic biomarkers such as brain natriuretic peptide, provide incremental prognostic value in the management of patients with HFPSF in routine clinical practice.25-28
Our study has a number of limitations that require comment. These data are based on a cohort of patients from a single tertiary institution. Despite this, the clinical data available for this cohort were substantially more detailed than reported previously. The index admission was that recorded at the tertiary hospital of interest; however, readmissions data from all tertiary and some regional hospitals throughout the state were used to ensure the maximum possible readmissions data were captured.7
The clinical underutilisation of echocardiography was a considerable limitation of this study. Capturing echocardiography data was complicated by patients having been discharged before an echocardiogram was performed, and those who had used private imaging services after discharge making records inaccessible. Although there may have been a bias towards patients with more severe symptoms having echocardiogram data available, this is unlikely to alter the findings of our study with regard to the characteristics of those with impaired compared with preserved systolic function.
Prescribing data reflected what patients were dispensed from the hospital pharmacy, but do not capture medications on ward stock, such as loop diuretics, and do not take into account potential changes to pharmacotherapy after discharge.
In the context of less compelling evidence for aggressive cardiopharmacological management, our study shows that these predominantly older female patients with HFPSF had less social support and higher readmission rates for associated comorbid illnesses. We propose that the burden of reduced survival in HFPSF may relate more to comorbid conditions than suboptimal cardiac management. Therefore, we would warn clinicians to caveat anicula! — beware of the little old lady with a “normal” echocardiogram.
1 Baseline characteristics of 753 patients admitted with chronic heart failure between 1 July 1994 and 30 June 2004, and mortality and readmissions data
|
Type of heart failure |
|
|||||||||||||
Characteristic |
HFPSF |
LVSD |
P* |
||||||||||||
No. of patients |
189 |
564 |
|
||||||||||||
Female |
118 (62.4%) |
217 (38.5%) |
0.001 |
||||||||||||
Mean age in years ± SD |
77± 13 |
75± 9 |
0.004 |
||||||||||||
Marital status |
|
|
|
||||||||||||
Married |
70 (37.0%) |
281 (49.8%) |
0.005 |
||||||||||||
Widowed |
85 (45.0%) |
173 (30.7%) |
< 0.001 |
||||||||||||
Separated/divorced |
17 (9.0%) |
50 (8.9%) |
ns |
||||||||||||
Single |
7 (3.7%) |
8 (1.4%) |
ns |
||||||||||||
Living status |
|
|
|
||||||||||||
Nursing home |
34 (17.9%) |
43 (7.6%) |
< 0.001 |
||||||||||||
House/independent |
122 (64.5%) |
465 (82.4%) |
< 0.001 |
||||||||||||
Hostel |
17 (8.9%) |
23 (4.0%) |
ns |
||||||||||||
Family support/next of kin |
|
|
|
||||||||||||
Child |
99 (52.3%) |
224 (39.7%) |
0.002 |
||||||||||||
Spouse |
52 (27.5%) |
206 (36.5%) |
0.016 |
||||||||||||
Other relative |
10 (5.3%) |
26 (5.0%) |
0.473 |
||||||||||||
Biochemistry results (mean concentration) |
|
|
|
||||||||||||
Sodium (mmol/L) |
138.91 |
138.27 |
ns |
||||||||||||
Potassium (mmol/L) |
4.08 |
4.22 |
ns |
||||||||||||
Creatinine (mmol/L) |
0.13 |
0.12 |
ns |
||||||||||||
Haemoglobin (g/dL) |
118.83 |
124.26 |
ns |
||||||||||||
Comparison of mortality and readmission |
|
|
|
||||||||||||
Death within 1 year |
42 (22.2%) |
148 (26.2%) |
ns |
||||||||||||
Median readmissions per patient during follow-up period (interquartile range) |
2 (1–5) |
1.5 (1–4) |
0.032 |
||||||||||||
No. of readmissions |
642 (27.5%) |
1688 (72.4%) |
|
||||||||||||
Reason for admissions by admission diagnostic coding |
|
|
|
||||||||||||
Anaemia |
25 (3.9%)† |
39 (2.3%) |
< 0.001 |
||||||||||||
Chronic heart failure |
125 (19.5%) |
423 (25.1%) |
< 0.001 |
||||||||||||
Diabetes |
27 (4.2%)† |
31 (1.8%) |
< 0.001 |
||||||||||||
Malignancy |
27 (4.2%)† |
31 (1.8%) |
< 0.001 |
||||||||||||
Median length of stay in days during follow-up period (interquartile range) |
16 (1.5–43) |
10 (0–36) |
0.084 |
||||||||||||
HFPSF = heart failure with preserved systolic function. LVSD = left ventricular systolic dysfunction. ns = not significant. * Significance level, P ≤ 0.05. † Higher than expected. |
|||||||||||||||
2 Management of chronic heart failure for 753 patients admitted between 1 July 1994 and 30 June 2004
|
Type of heart failure |
|
Compared with I-PRESERVE trial7 |
||||||||||||
Characteristic |
HFPSF |
LVSD |
P* |
||||||||||||
No. of patients |
189 |
564 |
|
|
|||||||||||
Pharmacotherapy |
|
|
|
|
|||||||||||
Angiotensin-converting enzyme inhibitor |
117 (61.9%) |
409 (72.5%) |
0.008 |
25% |
|||||||||||
Any β-blocker |
41 (21.6%) |
127 (22.5%) |
ns |
59% |
|||||||||||
Carvedilol† |
3 (1.5%) |
50 (8.8%) |
< 0.001 |
— |
|||||||||||
Angiotensin II receptor antagonist‡ |
127 (67.1%) |
432 (76.5%) |
0.012 |
|
|||||||||||
Digoxin |
73 (38.6%) |
240 (42.5%) |
ns |
14% |
|||||||||||
Spironolactone |
35 (18.5%) |
95 (16.8%) |
ns |
15% |
|||||||||||
Calcium-channel blocker |
45 (23.8%) |
119 (21.0%) |
ns |
40% |
|||||||||||
Statin |
45 (23.8%) |
147 (26.0%) |
ns |
31% |
|||||||||||
Antiarrhythmic |
17 (8.9%) |
75 (13.2%) |
ns |
8.7% |
|||||||||||
Nitrate |
44 (23.2%) |
198 (35.1%) |
0.003 |
27% |
|||||||||||
Specialist management |
|
|
|
|
|||||||||||
Referral to Cardiology Department |
38 (20.1%) |
164 (29.0%) |
0.018 |
— |
|||||||||||
HFPSF = heart failure with preserved systolic function. LVSD = left ventricular systolic dysfunction. ns = not significant. * Significance level, P ≤ 0.05. † The only β-blocker on the hospital formulary with trial efficacy for chronic heart failure. ‡ Excludes aldosterone receptor antagonists. |
|||||||||||||||
3 Comparison of survival among patients with left ventricular systolic dysfunction (LVSD) and heart failure with preserved systolic function (HFPSF)
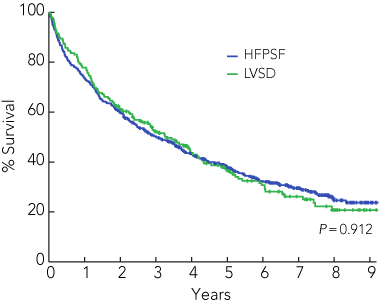
Year* |
0 |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
|||||
Surviving patients |
|||||||||||||||
LVSD |
564 |
415 |
330 |
270 |
216 |
174 |
135 |
88 |
31 |
3 |
|||||
HFPSF |
189 |
147 |
114 |
85 |
62 |
48 |
36 |
23 |
12 |
2 |
|||||
* Time from the first matching echocardiogram until the last date of follow-up. |
|||||||||||||||
4 Multivariate analysis of characteristics of heart failure with preserved systolic function, after controlling for sex
Variable |
Hazard ratio (95% CI) |
P* |
|||||||||||||
Age |
1.034 (1.02–1.04) |
< 0.001 |
|||||||||||||
“Other” relative as next of kin† |
1.502 (1.04–2.16) |
0.028 |
|||||||||||||
Hypertension |
0.769 (0.62–0.92) |
0.004 |
|||||||||||||
Dementia |
2.627 (1.34–5.15) |
0.005 |
|||||||||||||
Renal failure |
1.689 (1.38–2.06) |
< 0.001 |
|||||||||||||
Cerebrovascular disease |
1.686 (1.15–2.46) |
0.007 |
|||||||||||||
Peripheral vascular disease |
1.461 (1.11–1.92) |
0.007 |
|||||||||||||
Chronic obstructive pulmonary disease |
1.378 (1.03–1.72) |
0.005 |
|||||||||||||
Left ventricular size |
1.084 (1.02–1.15) |
0.009 |
|||||||||||||
* Significance level, P ≤ 0.05. † Not spouse or child. |
|||||||||||||||
Received 11 December 2008, accepted 3 August 2009
- Dennis T Wong1
- Robyn A Clark2
- Benjamin K Dundon1
- Andrew Philpott1
- Payman Molaee1
- Sepehr Shakib1
- 1 Royal Adelaide Hospital, Adelaide, SA.
- 2 Sansom Institute for Health Research, University of South Australia, Adelaide, SA.
Sepehr Shakib and Robyn Clark are alumni of the National Institute of Clinical Studies. Benjamin Dundon was supported by a Cardiac Society of Australia and New Zealand post-graduate research scholarship (2007) and a National Health and Medical Research Council (NHMRC)/National Heart Foundation of Australia (NHFA) co-funded post-graduate research scholarship 2008–2009. Payman Molaee was supported by an NHMRC/NHFA co-funded post-graduate research scholarship 2008–2009.
None identified.
- 1. Hogg K, Swedberg K, McMurray J. Heart failure with preserved left ventricular systolic function: epidemiology, clinical characteristics and prognosis. J Am Coll Cardiol 2004; 43: 317-327.
- 2. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2008: the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2008 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association of the ESC (HFA) and endorsed by the European Society of Intensive Care Medicine (ESICM). Eur Heart J 2008; 29: 2388-2442.
- 3. The Heart Failure Society of America (HFSA). 2006 Comprehensive heart failure practice guidelines. J Card Fail 2006; 12: e1-e122.
- 4. Krum H, Jelinek MV, Stewart S, et al. Guidelines for the prevention, detection and management of people with chronic heart failure in Australia. Med J Aust 2006; 185: 549-556. <MJA full text>
- 5. Auregemma GP, Zile MR, Gaasch WH. Contractile behaviour of the left ventricle in diastolic heart failure with emphasis on regional systolic function. Circulation 2006; 113: 296-304.
- 6. Dauterman KW, Massie BM, Gheorghiade M. Heart failure associated with preserved systolic function: a common and costly clinical entity. Am Heart J 1998; 135 (6 Pt 2 Suppl): S310-S319.
- 7. McMurray J, Carson P, Komajda M, et al. Heart failure with preserved ejection fraction: clinical characteristics of 4133 patients enrolled in the I-PRESERVE trial. Eur J Heart Fail 2008; 10: 149-156.
- 8. Bhatia RS, Tu JV, Lee DS, et al. Outcome of heart failure with preserved ejection fraction in a population-based study. N Engl J Med 2006; 355: 260-269.
- 9. Gottdiener JS, McClelland RL, Marshall R, et al. Outcome of congestive heart failure in elderly persons: influence of left ventricular systolic function. The Cardiovascular Health Study. Ann Intern Med 2002; 137: 631-639.
- 10. Ho KK, Pinsky JL, Kannel WB, et al. The epidemiology of heart failure. The Framingham Study. J Am Coll Cardiol 1993; 22 (4 Suppl A): 6A-13A.
- 11. Jiamsripong P, Honda T, Reuss CS, et al. Three methods for evaluation of left atrial volume. Eur J Echocardiogr 2008; 9: 351–355.
- 12. Diller PM, Smucker DR, David B, Graham RJ. Congestive heart failure due to diastolic or systolic dysfunction. Frequency and patient characteristics in an ambulatory setting. Arch Fam Med 1999; 8: 414-420.
- 13. World Medical Association. World Medical Association Declaration of Helsinki. Ethical principles for medical research involving human subjects. http://www.wma.net/en/30publications/10policies/b3/index.html (accessed May 2008).
- 14. Broadhead WE, Kaplan BH, James SA, et al. The epidemiologic evidence for a relationship between social support and health. Am J Epidemiol 1983; 117: 521-537.
- 15. Moody HR. Family support for the elderly: the international experience. Ethics in an aging society. Baltimore: Oxford University Press, 1992.
- 16. McClellan WM, Flanders WD, Langston RD, et al. Anaemia and renal insufficiency are independent risk factors for death among patients with congestive heart failure admitted to community hospital: a population-based study. J Am Soc Nephrol 2002; 13: 1928-1936.
- 17. Aronow WS, Kronzon I. Effect of enalapril on congestive heart failure treated with diuretics in elderly patients with prior myocardial infarction and normal left ventricular ejection fraction. Am J Cardiol 1993; 71: 602-604.
- 18. Bonow RO, Udelson JE. Left ventricular diastolic dysfunction as a cause of congestive heart failure. Mechanisms and management. Ann Intern Med 1992; 117: 502-510.
- 19. Brutsaert DL, Sys SU, Gillebert TC, et al. Diastolic failure: pathophysiology and therapeutic implications. J Am Coll Cardiol 1993; 22: 318-325.
- 20. Ahmed A, Rich MW, Love TE, et al. Digoxin and reduction in mortality and hospitalisation in heart failure: a comprehensive post hoc analysis of the DIG trial. Eur Heart J 2006; 27: 178-186.
- 21. Senni M, Tribouilloy CM, Rodeheffer RJ, et al. Congestive heart failure in the community: trends in incidence and survival in a 10 year period. Arch Intern Med 1999; 159: 29-34.
- 22. McDermott MM, Feinglass J, Lee PI, et al. Hospitalised congestive heart failure patients with preserved vs abnormal left ventricular systolic function: clinical characteristics and drug therapy. Am J Med 1995; 134: 728-736.
- 23. McDermott MM, Feinglass J, Lee PI, et al. Systolic function, readmission rates and survival among consecutively hospitalized patients with congestive heart failure. Am Heart J 1997; 134: 728-736.
- 24. Stewart S, MacIntyre K, Hole DA, et al. More malignant than cancer? Five year survival following first year admission for heart failure in Scotland. Eur J Heart Fail 2001; 3: 331-322.
- 25. Maisel AS, Koon J, Krishnaswamy P, et al. Utility of B-natriuretic peptide as a rapid, point-of-care test for screening patients undergoing echocardiography to determine left ventricular dysfunction. Am Heart J 2001; 141: 367-374.
- 26. Krishnaswamy P, Lubien E, Clopton P, et al. Utility of B-natriuretic peptide levels in identifying patients with left ventricular systolic or diastolic dysfunction. Am J Med 2001; 111: 274-279.
- 27. Lubien E, DeMaria A, Krishnaswamy P, et al. Utility of B-natriuretic peptide in detecting diastolic dysfunction: comparison with Doppler velocity recordings. Circulation 2002; 1005: 595-601.
- 28. Tschope C, Kasner M, Westermann D, et al. The role of NT-proBNP in the diagnostics of isolated diastolic dysfunction: correlation with echocardiographic and invasive measurements. Eur Heart J 2005; 26: 2277-2284.





Abstract
Objective: To determine whether heart failure with preserved systolic function (HFPSF) has different natural history from left ventricular systolic dysfunction (LVSD).
Design and setting: A retrospective analysis of 10 years of data (for patients admitted between 1 July 1994 and 30 June 2004, and with a study census date of 30 June 2005) routinely collected as part of clinical practice in a large tertiary referral hospital.
Main outcome measures: Sociodemographic characteristics, diagnostic features, comorbid conditions, pharmacotherapies, readmission rates and survival.
Results: Of the 2961 patients admitted with chronic heart failure, 753 had echocardiograms available for this analysis. Of these, 189 (25%) had normal left ventricular size and systolic function. In comparison to patients with LVSD, those with HFPSF were more often female (62.4% v 38.5%; P = 0.001), had less social support, and were more likely to live in nursing homes (17.9% v 7.6%; P < 0.001), and had a greater prevalence of renal impairment (86.7% v 6.2%; P = 0.004), anaemia (34.3% v 6.3%; P = 0.013) and atrial fibrillation (51.3% v 47.1%; P = 0.008), but significantly less ischaemic heart disease (53.4% v 81.2%; P = 0.001). Patients with HFPSF were less likely to be prescribed an angiotensin-converting enzyme inhibitor (61.9% v 72.5%; P = 0.008); carvedilol was used more frequently in LVSD (1.5% v 8.8%; P < 0.001). Readmission rates were higher in the HFPSF group (median, 2 v 1.5 admissions; P = 0.032), particularly for malignancy (4.2% v 1.8%; P < 0.001) and anaemia (3.9% v 2.3%; P < 0.001). Both groups had the same poor survival rate (P = 0.912).
Conclusions: Patients with HFPSF were predominantly older women with less social support and higher readmission rates for associated comorbid illnesses. We therefore propose that reduced survival in HFPSF may relate more to comorbid conditions than suboptimal cardiac management.