Methicillin-resistant Staphylococcus aureus (MRSA) bacteraemia is a frequent and dangerous complication of modern health care. At the very minimum, an episode of MRSA bacteraemia increases length of stay in hospital and has substantial financial costs, but some series have suggested that MRSA bacteraemia is associated with a mortality rate of 20%–25%.1 In Western Australia, all MRSA infections have been notifiable since 1985.2 In the United Kingdom, mandatory reporting of MRSA bacteraemia by National Health Service Trust hospitals commenced in May 2001.3
What is not always appreciated is that methicillin-susceptible S. aureus (MSSA) bacteraemia is also a serious health care-associated infection. In 2005, a report estimated a median rate of hospital-onset S. aureus bacteraemia (SAB) across 17 Australian hospitals of 0.7 cases per 1000 admissions.4 In 2006, there was a call for SAB, whether due to methicillin-resistant or methicillin-susceptible strains, to be used as a quality indicator for hospital infection control programs.5 Accordingly, a report from the Australian Commission on Safety and Quality in Health Care in 2008 identified health care-associated SAB as a key outcome measure for the National Hand Hygiene Initiative.6 While WA has been the leader with respect to MRSA notification, Tasmania recently went further and has made all cases of SAB notifiable, regardless of whether they are health care-related.7
Since 2001, Austin Health has had a strong focus on control of MRSA through the introduction of an institution-wide program to provide and promote alcohol-chlorhexidine hand hygiene for all health care workers, and alcohol wipes for all shared equipment.8 The rate of MRSA infections has fallen progressively over 7 years at Austin Health, and episodes of MRSA bacteraemia have become statistically less useful as a performance indicator. However, we, like others, observed patients with MSSA bacteraemia that was likely to be hospital-acquired.4,5 To address this problem, we introduced the “AuSABs” (Austin-associated S. aureus bacteraemias) program, whereby all episodes of SAB, not just those caused by MRSA, were made quality indicators. In this report, we evaluate the effectiveness and practicality of our AuSABs program.
An episode of SAB, including its association with a vascular device, was defined according to published criteria (Box 1).9,10 As part of our program, we introduced the term “AuSAB” (meaning SAB associated with health care at Austin Health), instead of “health care-associated SAB”, to encourage a sense of ownership and responsibility for these adverse events at Austin Health. Where possible, the source of bacteraemia was determined by identification of a primary focus, which was confirmed where possible by isolating S. aureus with the same antibiogram from that site.
Blood cultures were ordered as clinically indicated, and MRSA and MSSA isolates were identified by standard techniques.11 Isolates from episodes of AuSAB were examined for clonality by means of pulsed-field gel electrophoresis (PFGE) of SmaI digests of chromosomal DNA. The resulting PFGE pattern was analysed using GelCompar II, version 3.5 (Applied Maths NV, Sint-Martens-Latem, Belgium), as described previously.12 PFGE digestion patterns were considered clonal if GelCompar estimated a similarity of ≥ 80%.12
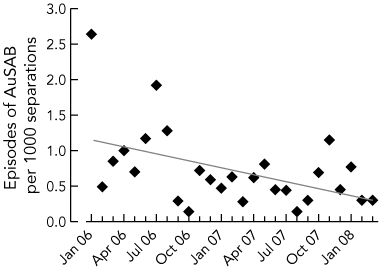
Rates of AuSABs per 1000 separations in the initial 9 months and subsequent 18 months were compared by two-tailed χ2 tests (Stata statistical software, release 5.0; StataCorp, College Station, Tex, USA). To calculate “cases prevented”, a linear regression line was fitted to the entire 27-month dataset using GraphPad Prism 4 software (GraphPad Software, San Diego, Calif, USA), which also calculated the slope of the regression line (Box 2) and upper and lower 95% confidence limits of the slope. Using Excel (Microsoft, Redmond, Wash, USA), the area under the observed regression curve (AUC) was compared with the AUC obtained assuming the initial rate of 1.1 per 1000 separations had remained unchanged.
Over the 27 months of the program from 1 January 2006 to 31 March 2008, there were 182 486 separations (67% same-day patients or “non-inpatients”), accounting for an equivalent of 673 626 total occupied bed-days. During the same period, we investigated 247 episodes of SAB, of which 131 (53.0%) were classified as AuSABs (Box 3) by the definition in Box 1. Sixteen of the 131 patients with an AuSAB (12.2%) died, and it was considered that the AuSAB contributed to death in nine of the 131 cases (6.9%).
The overall AuSAB rate was 0.7 per 1000 separations (1.94 per 10 000 occupied bed-days) over 27 months. However, the rate was 1.1 per 1000 separations in the first 9 months, during which the program was being developed and implemented, and fell to 0.51 per 1000 separations for the subsequent 18 months (P < 0.001; Box 2). If the rate of 1.1 per 1000 separations had continued for the entire 27-month period, we estimate that there would have been 80 more patients with SAB (95% CI, 20–140) than we actually observed (an additional three cases per month, on average)
As shown in Box 3, MSSA was isolated in 90 (68.7%) and MRSA was isolated in 41 (31.3%) of the 131 AuSABs. All but one of the AuSAB isolates were typed by PFGE. The 41 MRSA isolates could be clustered by PFGE into eight groups, with one main group comprising 27 isolates and seven smaller groups with four or fewer isolates. In contrast, the 90 AuSAB MSSA isolates clustered into 33 different groups, 25 of which were comprised of only one or two isolates. Of the other eight MSSA PFGE groups, the two largest had 18 and 11 isolates, respectively, which were identified from AuSAB episodes throughout the 27-month period, and did not seem to predominate in any particular month or location.
During the 27-month program, there were six occasions when the monthly AuSAB rate exceeded 1.0 per 1000 separations (Box 2). On one occasion (November 2007), the cases could not be linked to a particular area, and were a mixture of line-associated and non-line-associated infections. The other five occasions when the monthly rate was high were within the 9-month roll-out period and, of the 68 AuSABs that occurred, 33 (49%) were line-associated. Of these 33, 17 were associated with “clinical area A” and nine with “clinical area B”. Although it is difficult to establish with certainty, the infections seemed to be linked to issues with line insertion in clinical area A and accessing lines in clinical area B.
Episodes of SAB are an unambiguous marker of invasive infection, and more than half of the episodes we observed were health care-related. By monitoring all health care-associated SABs and not just MRSA, as with our previous strategy,8 we detected 90 additional health care-associated SABs (of a total 131 [68.7%]) that were caused by MSSA. We also detected 35 health care-associated SABs in non-inpatients (26.7%), which may have been missed by a ward-based surveillance program, and even more may have been missed if we had aimed the surveillance program at high-risk wards, such as the intensive care unit, only. Our program showed that vascular access device-related bacteraemias accounted for 49.6% of AuSABs, and many of these were associated with system errors that were previously undetected, and were potentially preventable. Others have also suggested there is great potential within hospitals to reduce or nearly eliminate nosocomial infections, in particular catheter-related bacteraemias.13-23 Furthermore, the multifaceted interventions used in our program, including staff education, nurse empowerment, intervention bundles and checklists, are relatively simple to implement in busy hospitals, and have proved to be sustainable.23
This quality improvement program was simple to perform and required relatively little time to maintain, at an estimated annual cost of $7000–$9000 (a proportion of 0.1 of an infection control clinical nurse consultant salary). Our findings mirror results obtained with a similar program at Canberra Hospital.14 Compared with the small cost of establishing the program, the economic costs of health care-associated SABs are huge. Estimates vary, but in the United States, the cost of an episode of SAB ranges from US$20 000 to US$50 000.1,15,16 Financial data for Australia are scarce, but assuming a cost of A$22 000 per episode,5 the total AuSAB cost at our institution for 27 months was almost $3 million. So, the annual cost of running the program ($7000–$9000) was less than half the cost of one episode of SAB. By preventing an estimated 80 AuSAB infections, the program may have saved our institution $1.75 million, and prevented 5–6 deaths over 27 months.
The AuSAB program is now core business for our infection control team, with investigation and feedback integrated into the team’s daily activities. Results are reported to the hospital executive, and are used as a performance indicator by the Infection Control Committee. The data generated from our program are also useful for comparison with other Australian hospitals, and the AuSAB rate has fallen below the previously published national median of 0.7 per 1000 admissions.4 At Austin Health, our overall performance target is now less than 0.5 AuSABs per 1000 separations, but our target for those that are line-related should be close to zero.23 The introduction of institution-wide SAB surveillance and feedback is a first step towards this goal.
1 Definitions of Staphylococcus aureus bacteraemia (SAB) used in this study, derived from published criteria9,10
2 Rates of Staphylococcus aureus bacteraemia associated with health care at Austin Health (AuSAB), with fitted computer-generated linear regression curve
- Claire Dendle1,2
- Rhea D Martin1
- Donna R Cameron1
- Elizabeth A Grabsch1
- Barrie C Mayall1
- M Lindsay Grayson1,2
- Paul D R Johnson1
- 1 Austin Health, Melbourne, VIC.
- 2 University of Melbourne, Melbourne, VIC.
We thank the staff of the Austin Health Microbiology Laboratory for isolating, identifying and reporting SABs; Shirley Xie and Wei Gao from the Microbiology Department for technical assistance with PFGE; Sandi Gamon and the staff of the Radiology Department for interventions in the Radiology Department; and the medical and nursing teams of Austin Health who willingly contributed to the review of each AuSAB.
None identified.
- 1. Cosgrove SE, Qi Y, Kaye KS, et al. The impact of methicillin resistance in Staphylococcus aureus bacteraemia on patient outcomes: mortality, length of stay, and hospital charges. Infect Control Hosp Epidemiol 2005; 26: 166-174.
- 2. Torvaldsen S, Roberts C, Riley TV. The continuing evolution of methicillin-resistant Staphylococcus aureus in Western Australia. Infect Control Hosp Epidemiol 1999; 20: 133-135.
- 3. Millar M, Coast J, Ashcroft R. Are methicillin-resistant Staphylococcus aureus bloodstream infection targets fair to those with other types of healthcare-associated infection or cost-effective? J Hosp Infect 2008; 69: 1-5.
- 4. Collignon P, Nimmo GR, Gottlieb T, Gosbell IB; Australian Group on Antimicrobial Resistance. Staphylococcus aureus bacteraemia, Australia. Emerg Infect Dis 2005; 11: 554-561.
- 5. Collignon PJ, Wilkinson IJ, Gilbert GL, et al. Health-care associated Staphylococcus aureus bloodstream infections: a clinical quality indicator for all hospitals. Med J Aust 2006; 184: 404-406. <MJA full text>
- 6. Cruickshank M, Ferguson J, editors. Reducing harm to patients from health care associated infection: the role of surveillance. Sydney: Australian Commission on Safety and Quality in Health Care, 2008. http://www.health.gov.au/internet/safety/publishing.nsf/Content/prog-HAI_Surveillance#report (accessed Aug 2009).
- 7. Department of Human Services, Tasmania. Guidelines for notification of notifiable diseases, human pathogenic organisms and contaminants. Public Health Act 1997. Issued 17 December 2008. http://www.dhhs.tas.gov.au/__data/assets/pdf_file/0019/31681/Notifiable_Diseases_Guidelines_Final1.pdf (accessed Aug 2009).
- 8. Johnson PD, Martin R, Burrell LJ, et al. Efficacy of an alcohol/chlorhexidine hand hygiene program in a hospital with high rates of nosocomial methicillin-resistant Staphylococcus aureus (MRSA) infection. Med J Aust 2005; 183: 509-514. <MJA full text>
- 9. Australian Infection Control Association. Blood stream infection (BSI). http://www.health.qld.gov. au/chrisp/icare/aica_bsi_def_v1.pdf (accessed Aug 2009).
- 10. O’Grady NP, Alexander M, Dellinger EP, et al. Guidelines for the prevention of intravascular catheter-related infections. Centers for Disease Control and Prevention. MMWR Recomm Rep 2002; 51 (RR-10): 1-29.
- 11. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing. Fifteenth informational supplement (M100-S15 Vol. 25 No. 1). Wayne, Pa: CLSI, 2005.
- 12. Ballard SA, Pertile KK, Lim M, et al. Molecular characterization of vanB elements in naturally occurring gut anaerobes. Antimicrob Agents Chemother 2005; 49: 1688-1694.
- 13. Horan TC, Gaynes RP. Surveillance of nosocomial infections. In: Mayhall CG, editor. Hospital epidemiology and infection control. 3rd ed. Philadelphia: Lippincott Williams & Wilkins, 2004: 1659-1702.
- 14. Dreimanis D, Beckingham W, Collignon P, Roberts J. Staphylococcus aureus bacteraemia surveillance: a relatively easy to collect but accurate clinical indicator on serious health-care associated infections and antibiotic resistance. Aust Infect Control 2005; 10: 127-130.
- 15. Noskin GA, Rubin RJ, Schentag JJ, et al. The burden of Staphylococcus aureus infections on hospitals in the United States. Arch Intern Med 2005; 165: 1756-1761.
- 16. Rubin RJ, Harrington CA, Poon A, et al. The economic impact of Staphylococcus aureus infection in New York City hospitals. Emerg Infect Dis 1999; 5: 9-17.
- 17. Haley RW, Culver DH, White JW. The efficacy of infection surveillance and control programs in preventing nosocomial infections in US hospitals. Am J Epidemiol 1985; 121: 182-205.
- 18. Harbarth S, Sax H, Gastmeier P. The preventable proportion of nosocomial infections: an overview of published reports. J Hosp Infect 2003; 54: 258-266.
- 19. Eggimann P, Harbarth S, Constantin MN, et al. Impact of a prevention strategy targeted at vascular-access care on the incidence of infections acquired in intensive care. Lancet 2000; 355: 1864-1868.
- 20. Makasi H, Watanabe H, Degawa S, et al. Significant reduction of methicillin-resistant Staphylococcus aureus bacteremia in geriatric wards after introduction of infection control measures against nosocomial infections. Intern Med 2001; 40: 214-220.
- 21. Huang SS, Yokoe DS, Hinrichsen VL, et al. Impact of routine intensive care unit surveillance cultures and resultant barrier precautions in hospital-wide methicillin-resistant Staphylococcus aureus bacteraemia. Clin Infect Dis 2006; 43: 971-978.
- 22. Tomford JW, Hershey CO, McLaren CE, et al. Intravenous therapy team and peripheral venous catheter-associated complications. A prospective controlled study. Arch Intern Med 1984; 144: 1191-1194.
- 23. Berenholtz SM, Pronovost PJ, Lipsett PA, et al. Eliminating catheter-related bloodstream infections in the intensive care unit. Crit Care Med 2004; 32: 2014-2020.





Abstract
Objective: To evaluate the practicality and effectiveness of a new program that made health care-associated Staphylococcus aureus bacteraemia (SAB) a quality indicator at Austin Health.
Design and setting: Roll-out of the program over 9 months and review over 27 months from January 2006. Every episode of SAB at Austin Health was promptly reviewed, and classified as community- or health care-associated and as inpatient- or non-inpatient-related. Feedback was provided to treating clinicians for every SAB episode considered potentially preventable, and education-based interventions were introduced where appropriate.
Main outcome measure: Episodes of SAB associated with health care at Austin Health per 1000 separations (hospital discharges) per month.
Results: We identified 131 episodes of health care-associated SAB, of which 90 (68.7%) were caused by methicillin-susceptible S. aureus, 96 (73.3%) occurred in inpatients, and 65 (49.6%) were associated with a vascular access device. The health care-associated SAB rate was 1.1 per 1000 separations in the first 9 months, and fell by 55% to 0.51 per 1000 separations in the subsequent 18 months. We estimated that there were 80 fewer SAB episodes (95% CI, 20–140) than expected had the initial rate remained unchanged, a notional saving of $1.75 million to Austin Health over 27 months. About 16 hours per month of clinical nurse consultant time was required to maintain the program, representing a 0.1 equivalent full-time position, or a cost of $7000–$9000 per year.
Conclusion: Introducing a structured program to investigate all health care-associated SABs, rather than only infections with methicillin-resistant S. aureus, revealed a large under-recognised burden of potentially preventable infections. The program was simple and low-cost, and the rate of health care-associated SAB has fallen significantly since its introduction.