In the United States, the United Kingdom and Australia, people over the age of 50 years now constitute the majority of those who die from asthma.1-3 Links between asthma and an increased risk of mortality among older people have now been established,4 prompting calls for the application of appropriate prevention and education strategies for older patients with asthma.5
Deficits in physicians’ provision of care and their understanding of asthma guidelines have been previously identified.6,7 Furthermore, recent research has revealed differences in the health beliefs and behaviours of older people with asthma that may render currently promoted asthma education strategies less effective in older age groups.8 In response to this, we designed a multifaceted educational intervention to improve the content and style of general practice consultations for older people with asthma, using previously validated best-practice models to change physician behaviour.9-11 The intervention was also based on a needs analysis of the priorities of older people with asthma.8
The GP intervention comprised (in the following order):
a 2-hour group educational session involving theoretical and practical knowledge of asthma;
participation in a videorecorded simulated patient consultation; and
a 1-hour academic detailing visit at the GP’s usual practice location 1–4 weeks after the educational session. This session was used to provide feedback to GPs and was individually tailored to individual GP needs.
GP outcomes were assessed by the use of eight simulated patients recruited from the Monash Centre for Medical and Health Sciences Education, who were provided with asthma medication devices and information on asthma, and who rehearsed a script based on patients’ experience. Simulated patients were all non-physicians who had prior experience of portraying a specific patient case in a consistent, standardised fashion and in evaluating health professional encounters.12 All were blinded to the intervention status of the GPs. Simulated patients used a previously validated evaluation tool13 to evaluate their satisfaction with the consultation and its style according to rapport, how organised the GP was, and whether the GP had addressed all of their concerns.
All consultations with simulated patients were videorecorded on an unattended camera, and the research officer and an academic detailer assessed the content of consultations according to a checklist developed from National Asthma Council Australia guidelines14 and the content of the intervention.
Primary endpoints for patient outcomes were patients’ lung function15 and scores on the Asthma Control Questionnaire,16 an asthma knowledge questionnaire,17 an asthma-related quality-of-life questionnaire,18 a questionnaire assessing adherence to asthma preventer medication,19 and the European Community Respiratory Health Survey.20
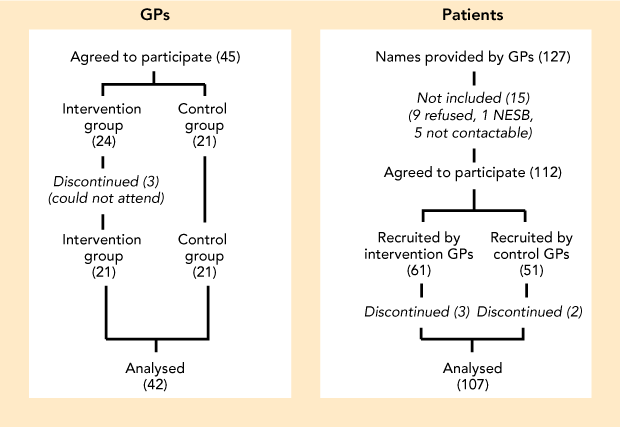
Box 1 shows that, of 45 GPs who enrolled in the educational program, 42 completed all components of the intervention and evaluation (21 in the intervention group and 21 in the control group). Participating GPs provided the names of 127 patients, 107 of whom completed participation in the study.
There was no significant difference between the demographic variables of GPs in the control and intervention groups (Box 2).
There was little difference between patients recruited by GPs in the intervention and control groups at baseline (Box 3). The only significant difference was that those recruited by GPs in the control group had a higher proportion of current smokers who had smoked for more than 10 years (16% v 2%; P = 0.02). Both groups of patients showed adequate asthma control and asthma-related quality of life (Box 4).
Several features of our intervention differed from previously published interventions aimed at developing best practice in asthma care. Specifically, our intervention targeted older people with asthma; was robust in design, evaluating both patient and GP outcomes; and was based on qualitative needs analyses performed on data obtained from both patients8 and health care providers.7 Critically, our intervention was based on best-practice models of implementing change in health professional behaviour,9-11 which it proved to do.
Previously published interventions to improve the delivery of asthma care have focused on the development of patient-centred care and the provision of asthma action plans.21,22 These have been shown to increase GPs’ confidence, reduce emergency department attendances and improve asthma-related quality of life. Yet, these previous trials have predominantly been performed in children.21,23 Older people with asthma have generally been excluded from studies such as these.24 Our study was based on our previous research, which identified unique issues in the delivery of care to older people with asthma.8
There are several possible reasons for the failure of our intervention to significantly influence patient outcomes. Although GPs who enrolled into our study reflected the demographic trends of GPs in the general population,25 most were either Fellows of the RACGP or had undertaken postgraduate training. Enrolment into the educational program itself suggests an existing interest in providing quality asthma care. Their patients’ lung function, asthma control, asthma-related quality of life and adherence to taking asthma medication at baseline were mostly adequate,26 with little room for improvement in these measures.
3 Demographic characteristics of the patients, comparing those recruited by general practitioners in the intervention group and control group
- Dianne P Goeman1
- Lena A Sanci2
- Simon L Scharf3
- Michael Bailey4
- Robyn E O’Hehir1,4
- Christine R Jenkins5
- Jo A Douglass1
- 1 Department of Allergy, Immunology and Respiratory Medicine (AIRmed), Cooperative Research Centre for Asthma and Airways, Alfred Hospital and Monash University, Melbourne, VIC.
- 2 Department of General Practice, University of Melbourne, Melbourne, VIC.
- 3 Aged Care Services, Caufield General Medical Centre, Melbourne, VIC.
- 4 Monash University, Melbourne, VIC.
- 5 Woolcock Institute of Medical Research, Sydney, NSW.
We thank the participating doctors and their patients, Ms Adrienne James, Manager of the Alfred Hospital’s Lung Health Promotion Centre for providing the GP academic detailing sessions, Dr Bella Blaher for assistance with videorecording, Ms Edwina Yu for assistance with videorecording and data collection, Ms Debbie Kiegaldie from the Monash Centre for Medical and Health Sciences Education, and our simulated patients.
None identified.
- 1. Campbell MJ, Cogman GR, Holgate SL, et al. Age specific trends in asthma mortality in England and Wales, 1983–95: results of an observational study. BMJ 1997; 314: 1439-1441.
- 2. Moorman JE, Rudd RA, Johnson CA. National Surveillance for Asthma — United States, 1980–2004. Centers for Disease Control and Prevention. MMWR Surveillance Summaries, 19 Oct 2007. 56 (SS08). http://www.cdc.gov/mmwr/preview/mmwrhtml/ss5608a1.htm (accessed Jun 2009).
- 3. Australian Bureau of Statistics. Causes of death, Australia, 2006. Canberra: ABS, 2008. (ABS Cat. No. 3303.0.) http://www.abs.gov.au/AUSSTATS/abs@.nsf/DetailsPage/3303.02006? OpenDocument (accessed Jun 2009).
- 4. Dantzer C, Tessier JF, Nejjari C, et al. Mortality of elderly subjects with self-reported asthma in a French cohort, 1991–1996. Eur J Epidemiol 2001; 17: 57-63.
- 5. National Asthma Council Australia. 2008 April 10 — Asthma deaths spike [media release]. http://www.nationalasthma.org.au/content/view/470/969/ (accessed Jun 2009).
- 6. Doerschug KC, Peterson MW, Dayton CS, Kline JN. Asthma guidelines: an assessment of physician understanding and practice. Am J Respir Crit Care Med 1999; 159: 1735-1741.
- 7. Goeman DP, Hogan CD, Aroni RA, et al. Barriers to delivering asthma care: a qualitative study of general practitioners. Med J Aust 2005; 183: 457-460. <MJA full text>
- 8. Goeman D, O’Hehir R, Jenkins C, et al. “You have to learn to live with it”: a qualitative and quantitative study of older people with asthma. Clin Respir J 2007; 1: 99-105.
- 9. Oxman AD, Thomson MA, Davis DA, et al. Changing physician performance. JAMA 1995; 274: 700-705.
- 10. Norman GR, Neufeld V, Walsh A, et al. Measuring physicians’ performances by using simulated patients. J Med Educ 1985; 60: 925-934.
- 11. Sanci L, Coffey C, Veit F, et al. Evaluation of the effectiveness of an educational intervention for general practitioners in adolescent health care: randomised controlled trial. BMJ 2000; 320: 224-230.
- 12. Colliver JA, Swartz MH. Assessing clinical performance with standardised patients. JAMA 1997; 278: 790-791.
- 13. Sanci LA, Day NA, Coffey CMM, et al. Simulations in evaluation of training: a medical example using standardised patients. Eval Program Plann 2002; 25: 35-46.
- 14. National Asthma Council Australia. Asthma management handbook 2002. Melbourne: NAC, 2002.
- 15. ATS/ERS guidelines for spirometry testing. Eur Respir J 2005; 26: 319-338.
- 16. Juniper E, O’Byrne P, Ferrie P, et al. Measuring asthma control. Am J Respir Crit Care Med 2000; 162: 1330-1334.
- 17. Allen RM, Jones MP. The validity and reliability of an asthma knowledge questionnaire used in the evaluation of a group asthma education self-management program for adults with asthma. J Asthma 1998; 35: 537-545.
- 18. Marks GB, Dunn SM, Woolcock AJ. A scale of measurement of quality of life in adults with asthma. J Clin Epidemiol 1992; 45: 461-472.
- 19. Brooks CM, Richards JM, Kohler CL, et al. Assessing adherence to asthma medication and inhaler regimens: a psychometric analysis of adult self-report scales. Med Care 1994; 32: 298-307.
- 20. Burney P, Luczynska C, Chinn S, et al. The European Community Respiratory Health Survey. Eur Respir J 1994; 169: 954-960.
- 21. Liaw ST, Sulaiman ND, Barton CA, et al. An interactive workshop plus locally adapted guidelines can improve asthma knowledge and management. BMC Fam Pract 2008; 9: 22.
- 22. Lefevre F, Piper M, Weiss K, et al. Do written action plans improve patient outcomes in asthma? An evidence-based analysis. J Fam Pract 2002; 51: 842-848.
- 23. Clark NM, Gong M, Schork M, et al. Impact of education of physicians on patient outcomes. Pediatrics 1998; 101: 831-836.
- 24. McMurdo MET, Witham MD, Gillespie ND. Including older people in clinical research. BMJ 2005; 331: 1036-1037.
- 25. Charles J, Britt H, Valenti L. The evolution of the general practice workforce in Australia, 1991–2003. Med J Aust 2004; 181: 85-90. <MJA full text>
- 26. Juniper EF, Bousquet J, Abetz L, Bateman ED; Goal Committee. Identifying “well controlled” and “not well controlled” asthma using the Asthma Control Questionnaire. Respir Med 2006; 100: 616-621.





Abstract
Objective: To evaluate the effectiveness of a multifaceted educational intervention for general practitioners to improve the outcomes of older people with asthma.
Design: Cluster randomised controlled trial.
Participants and setting: 42 GPs recruited from metropolitan Melbourne between 1 August 2006 and 31 July 2007, randomly assigned to an intervention or control group, and 107 patients with asthma, aged 55 years or older (consecutive patients recruited by the GPs).
Main outcome measures: Evaluation by means of a videorecorded consultation with a simulated patient for GPs; and for patients, asthma control and quality of life, lung function and action plan ownership at baseline and at 4 months.
Results: GPs in the intervention group scored significantly higher than those in the control group for the content and style of their consultation with simulated patients. At 4 months’ follow-up, there was no significant difference between patient groups in the asthma control scores, asthma-related quality of life or lung function.
Conclusion: This trial showed an improvement in GPs’ performance in delivering asthma care to older people. Despite this, there was no significant improvement in patient outcomes.
Trial registration: Australian New Zealand Clinical Trials Registry ACTRN12607000634471.