Smoking cessation interventions, involving pharmacological, behavioural and alternative therapies have been extensively studied overseas and in mainstream communities in Australia. While evidence from these studies can influence the design of quit-smoking interventions for Aboriginal people and Torres Strait Islanders, it has some limitations.1 Many of the intervention studies enrol predominantly moderately to highly nicotine-dependent smokers and are conducted in communities where smoking is no longer the norm.2,3 Consequently, pharmacological support has assumed an increasingly dominant role in mainstream quit programs.
One of the factors in determining whether an individual stops smoking is physical nicotine addiction. There are some data from the Indigenous community that describe motivation to quit and the contextual factors in the environment that will affect the habitual and psychological aspects of smoking, but no data on physical addiction to nicotine.1,4 The Fagerström Test for Nicotine Dependence (FTND) is a validated tool that uses six short questions to assess a person’s physical nicotine dependence.5 Its brevity and reliability in predicting relapse6 has led to it being widely used in the evaluation of smoking cessation interventions, and its use is recommended in Australian protocols.7 However, there is little evidence from within the Indigenous community to inform its use.
The Mums and Babies program, involving antenatal care delivered by a mutidisciplinary team, which commenced on 1 January 2000 in Townsville, Queensland, has been described previously.8 From early 2005, all women attending the Mums and Babies program who reported tobacco use have been assessed using the FTND to guide brief interventions for tobacco use. In addition, from 1 November 2005, women undergoing baseline assessment for the Tilly’s Tracks project9 provided a comprehensive smoking history, including a 24-hour retrospective diary detailing their personal smoking status, their partner’s smoking habits, and exposure to environmental tobacco smoke in the home. This information was collected at interview by trained Aboriginal health workers, and was added to the routine antenatal care activities included in a first visit by the clinical team. Women also provided urine samples for cotinine testing.
Cotinine analysis was performed on urine samples of the women who were involved in the Tilly’s Tracks project. Analysis was by enzyme-linked immunosorbent assay (ELISA) (Cozart Microplate EIA, Cozart, Abingdon, Oxfordshire, UK), as described previously.9 A standard curve was generated for each analysis run, and if the cotinine levels for both samples in a duplicate pair were above the standard range, that sample was allocated the maximum level from the standard curve for the run.
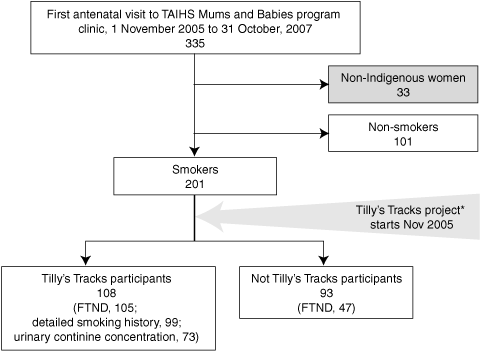
Box 1 shows the derivation of the study groups. Of the 302 pregnant Indigenous women, 71.9% identified as Aboriginal, 17.2% as Torres Strait Islanders and 10.9% as both, and 201 (66.6%) self-reported current tobacco use at their first antenatal care visit.
The mean age of the 302 women was 25.6 years (SD, 5.9), with no difference between smokers and non-smokers. Of the women aged less than 20 years, 79.6% reported current tobacco use (Box 2). The only characteristic that differed between women who did or did not complete an FTND was alcohol use, with those not completing the FTND being more likely to report risky levels of drinking. There were no significant differences in demographic characteristics between the women in the Tilly’s Tracks project who did and did not provide a urinary sample for cotinine estimation.
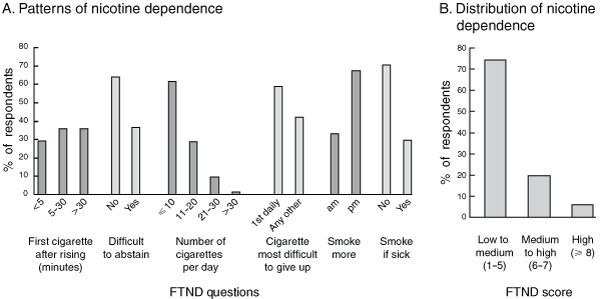
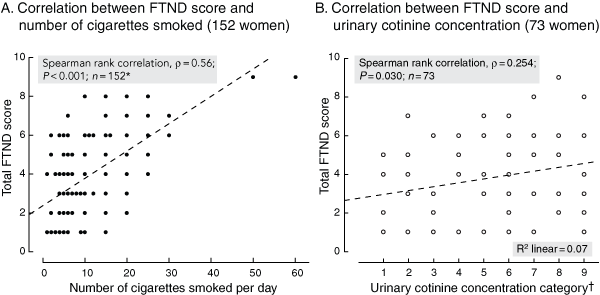
Of the 201 smokers, 152 (75.6%) completed an FTND. The distribution of FTND responses and scores is shown in Box 3. The median FTND score was 4 (IQR, 2–9); 40.1% of these pregnant smokers scored 3 or less on the FTND. The FTND questions that correlated best with the total FTND score were Question 1 (How soon after you get out of bed do you have a cigarette?) and Question 4 (the number of cigarettes smoked each day). There was a significant positive correlation between FTND score and number of cigarettes smoked each day, with the number of cigarettes smoked explaining 30.3% of the variation in the FTND (Box 4, A).
A first-visit urinary cotinine sample was collected from 73 of the 108 women in the Tilly’s Tracks project (67.6%), with a median concentration of 3056.6 ng/mL (IQR, 1068.9–5858.7 ng/mL). There was a significant positive correlation between FTND score and urinary cotinine concentrations (Box 4, B).
Most smoking cessation guidelines suggest that an assessment of nicotine dependence should be made to guide individual quit attempts.7,11,12 Use of the FTND in this study was incorporated into the workflow of a busy Indigenous maternal and child health clinic. Feedback from health workers and other staff was positive. They found the tool simple to incorporate into daily clinical work, easily understood and useful as an “ice breaker”. The study shows significant correlations between the number of cigarettes smoked and nicotine dependence scores. The correlations of both the number of cigarettes smoked and the time until the first cigarette of the day — the two FTND questions that comprise the Heavy Smoking Index (HSI)11 — with the FTND score are to be expected, but many women had low total FTND scores, and the other FTND questions could potentially have been significant contributors to the total FTND score. With international evidence suggesting that relying solely on the number of cigarettes smoked as an indicator of nicotine dependence will underestimate the number of patients with a high level of nicotine addiction,13 the FTND or HSI may be useful in primary care clinics.
The level of nicotine dependence in the Indigenous community in Australia has not been previously reported. Among the group of Indigenous women in our study, 40% scored 3 or lower on the FTND, indicating low levels of physical nicotine dependence. This distribution of scores is consistent with findings of other studies in pregnant women.14,15 It is possible that our cohort of Indigenous women under-scored on the FTND, as has been suggested in African American teenagers.16 However, the credibility of these demonstrated lower levels of nicotine dependence may be supported by the current Indigenous tobacco narrative, which includes suggestions that Indigenous people cope without constant access to cigarettes,1 and an apparent failure of nicotine replacement therapy (NRT) in the Northern Territory.17 The NT trial showed that NRT conferred little advantage over a brief intervention alone, which was partly blamed on inadequate resources and a low-intensity intervention. However, the degree of nicotine dependence was not reported in that study, and it is possible that the nicotine dependence of the participants may have been lower than was assumed,4 which might have contributed to the lack of efficacy of NRT.
Low levels of nicotine dependence among Indigenous pregnant women has significant implications for the implementation of quit-smoking programs. Evidence suggests that NRT is not of value in light smokers or those with low nicotine dependence,18,19 which is reflected in a number of smoking cessation guidelines.7,12 This reinforces the need to assess nicotine dependence. While pharmacological therapies are important, they are expensive, have side effects, and their use must be tailored to individual needs. The FTND scores in this study suggest cessation programs need to consider the smoking behaviours of each Indigenous woman to develop individual quit plans with strategies to counter the habitual aspects of their smoking.
Our study had some limitations. The cohort comprised pregnant women, and the overall demographic characteristics of our cohort were typical of Indigenous pregnant women,8 so our sample was probably representative of that group. However, our findings may not be applicable to the non-pregnant Indigenous community. The FTND has previously been found to be psychometrically sound and to correlate strongly with cotinine levels;5,20 the significant but weaker association found in this group of women may be explained by a number of factors. Nicotine metabolism in pregnancy is accelerated, resulting in reduced cotinine levels,21 and cigarette consumption tends to vary throughout pregnancy.22 In addition, recording cotinine levels using a maximum cut-off level is likely to have underestimated the correlation coefficient for FTND and cotinine level. While cotinine levels are method-dependent,20,23 those reported for these pregnant women are comparable to levels found in other reports.22,24 To further explore these issues in the Indigenous Australian community would require more rigorous investigation.
2 Comparison of demographic and other characteristics for smokers and non-smokers among 302 pregnant Australian Indigenous women attending the Mums and Babies program at the Townsville Aboriginal and Islander Health Service between 1 November 2005 and 31 October 2007
3 Patterns and distribution of nicotine dependence as assessed using the Fagerström Test of Nicotine Dependence (FTND) at the first antenatal visit for 152 women attending the Mums and Babies program clinic at Townsville Aboriginal and Islander Health Service between November 1 2005 and October 31 2007
4 Correlations between Fagerström Test for Nicotine Dependence (FTND) score and (A) the reported number of cigarettes smoked (152 women), and (B) the urinary cotinine concentration measured (73 women) at the first antenatal visit to the Mums and Babies program clinic at the Townsville Aboriginal and Islander Health Service between 1 November 2005 and 31 October 2007
 | |||||||||||||||
- Kathryn S Panaretto1
- Melvina R Mitchell2
- Lynette Anderson1
- Conor Gilligan3
- Petra Buettner4
- Sarah L Larkins4
- Sandra Eades5
- 1 Queensland Aboriginal and Islander Health Council, Queensland Aboriginal and Islander Health Population Health Hub, Townsville, QLD.
- 2 Community Child Health Unit, Queensland Health, Townsville, QLD.
- 3 School of Medicine and Public Health, University of Newcastle, Newcastle, NSW.
- 4 James Cook University, Townsville, QLD.
- 5 Baker Institute, Melbourne, VIC.
We thank the Board of TAIHS for their support of the project. We also thank Kaye Thompson for her work in the Tilly’s Tracks project. This study was supported in part by National Health and Medical Research Council Project Grant No. 510776.
None identified.
- 1. Ivers RG. An evidence-based approach to planning tobacco interventions for Aboriginal people. Drug Alcohol Rev 2004; 23: 5-9.
- 2. Baker A, Ivers RG, Bowman J, et al. Where there’s smoke, there’s fire: high prevalence of smoking among some sub-populations and recommendations for intervention. Drug Alcohol Rev 2006; 25: 85-96.
- 3. Shiffman S. Reflections on smoking relapse research. Drug Alcohol Rev 2006; 25: 15-20.
- 4. Johnston V, Thomas D. Smoking behaviours in a remote Australian Indigenous community: the influence of family and other factors. Soc Sci Med 2008; 67: 1708-1716.
- 5. Heatherton TF, Kozlowski LT, Frecker RC, Fagerström KO. The Fagerström Test for Nicotine Dependence: a revision of the Fagerström Tolerance Questionnaire. Br J Addict 1991; 86: 1119-1127.
- 6. Piper M, McCarthy D, Baker T. Assessing tobacco dependence: a guide to measure evaluation and selection. Nicotine Tob Res 2006; 8: 339-351.
- 7. Royal Australian College of General Practitioners. Smoking cessation guidelines for Australian general practice: Practice handbook 2004. Melbourne: RACGP, 2005.
- 8. Panaretto KS, Lee HM, Mitchell MR, et al. Impact of a collaborative shared antenatal care program for urban Indigenous women: prospective cohort study. Med J Aust 2005; 182: 514-519. <MJA full text>
- 9. Gilligan C, Sanson-Fisher R, Eades S, et al. Assessing the accuracy of self-reported smoking status among pregnant Aboriginal and Torres Strait Islander women using cotinine biochemical validation. Drug Alcohol Rev 2009. In press.
- 10. Ministerial Council on Drugs Strategy. National Drug Strategy. Aboriginal and Torres Strait Islander peoples complementary action plan 2003–2006. Background paper. Canberra: National Drug Strategy, 2003. (accessed Oct 2009). <MJA full text>
- 11. Royal Australian College of General Practitioners. SNAP: a population health guide to behavioural risk factors in general practice. Melbourne: RACGP, 2004.
- 12. van Schayck OC, Pinnock H, Ostrem A, et al. International Primary Care Respiratory Group consensus statement. Tackling the smoking epidemic – practical guidance for primary care. Prim Care Respir J 2008; 17: 185-193.
- 13. de Leon J, Diaz FJ, Becoña E, et al. Exploring brief measures of nicotine dependence for epidemiological surveys. Addict Behav 2003; 28: 1481-1486.
- 14. Ward K, Vander Weg M, Sell M, et al. Characteristics and correlates of quitting smoking among black and white low-income pregnant smokers. Am J Health Behav 2006; 30: 651-662.
- 15. Händel G, Hannöver W, Röske K, et al. Intention to change smoking in pregnant and postpartum women according to number of pregnancies. Eur J Public Health 2009; 19: 218-221.
- 16. Schroeder JR, Moolchan ET. Ethnic differences among adolescents seeking smoking cessation treatment: a structural analysis of responses on the Fagerström Test for Nicotine Dependence. Nicotine Tob Res 2007; 9: 137-145.
- 17. Ivers RG, Farrington M, Burns CB, et al. A study of the use of free nicotine patches by Indigenous people. Aust N Z J Public Health 2003; 27: 486-490.
- 18. Tang J, Law M, Wald N. How effective is nicotine replacement therapy in helping people to stop smoking? BMJ 1994; 308: 21-26.
- 19. Peters MJ, Morgan LC. The pharmacotherapy of smoking cessation. Med J Aust 2002; 176: 486-490. <MJA full text>
- 20. Haufroid V, Lison D. Urinary cotinine as a tobacco-smoke exposure index: a minireview. Int Arch Occup Environ Health 1998; 71: 162-168.
- 21. Dempsey D, Jacob Pr, Benowitz N. Accelerated metabolism of nicotine and cotinine in pregnant smokers. J Pharmacol Exp Ther 2002; 301: 594-598.
- 22. Pickett K, Rathouz P, Kasza K, et al. Self-reported smoking, cotinine levels, and patterns of smoking in pregnancy. Paediatr Perinat Epidemiol 2005; 19: 368-376.
- 23. Gorber S, Schofield-Hurwitz S, Hardt J, et al. The accuracy of self-reported smoking: a systematic review of the relationship between self-reported and cotinine-assessed smoking status. Nicotine Tob Res 2009; 11: 12-24.
- 24. Lawrence T, Aveyard P, Croghan E. What happens to women’s self-reported cigarette consumption and urinary cotinine levels in pregnancy? Addiction 2003; 98: 1315-1320.





Abstract
Objectives: To examine patterns of nicotine dependence, the value of the Fagerström Test for Nicotine Dependence (FTND) and its correlation with self-reported tobacco use and urinary cotinine concentrations among pregnant Indigenous women in Townsville.
Design, participants and setting: Cross-sectional study of 201 consecutive women who self-reported tobacco use at their first antenatal visit to Townsville Aboriginal and Islander Health Service (TAIHS) between 1 November 2005 and 31 October 2007. All smokers were to be assessed by FTND, and 108 women participating in the Tilly’s Tracks project (a randomised trial of an intervention to reduce smoking in pregnant Aboriginal and Torres Strait Islander women) were to have a comprehensive smoking history taken and urinary cotinine samples collected.
Main outcome measures: Self-reported smoking status, FTND scores and urinary cotinine concentrations.
Results: Of 302 Indigenous women presenting to TAIHS, 201 (66.6%) identified as current tobacco users at their first antenatal visit; this proportion rose to 79.6% in women aged < 20 years. An FTND was completed for 152 women (75.6%), with a median score of 4, and 40.1% scoring 3 or less, indicating low levels of nicotine dependence. There were significant correlations between the FTND and number of cigarettes smoked (ρ = 0.56; P < 0.001) and urinary cotinine concentrations (ρ = 0.25; P = 0.030). Of those who provided comprehensive smoking histories, the median age of starting smoking was 15 years, with a median of two previous quit attempts; 71.4% reported partners who smoked and 27.3% reported smoking occurred inside the house.
Conclusion: The use of the FTND in Indigenous pregnant women may assess physical nicotine dependence, thus providing information that will help in preparing quit-smoking plans, including tailoring of pharmacological support to individual need. Quit-smoking programs that better address the behavioural and psychological aspects of smoking within the Indigenous community in Australia are needed.