Understanding the potential roles and pitfalls of emerging imaging technologies in breast screening requires awareness of key concepts and assumptions (summarised in Box 1). The benefit of early detection of breast cancer has been shown in randomised controlled trials (RCTs) of mammography, with a 25%–35% reduction in breast cancer deaths for women aged 50–69 years who undergo screening.1 Evaluations of population screening in practice, including an Australian program, have similarly demonstrated a benefit in women in this age group.2,3
Women aged 40–49 years may also benefit from regular breast screening, but the reduction in mortality is estimated at 15%–16% — lower than the benefit found for those aged 50–69 years — and of borderline statistical significance.4 In addition, women older than 70 years may benefit from breast screening, but the benefit diminishes with increasing age because of the competing causes of death, and is estimated to be about half the benefit in women aged 50–69.5 Considerations when advising older women about breast screening include a potential for benefit in those with a high risk of breast cancer death but low risk of death from other conditions, and in those perceived to be in good health.5
In Australia, all women aged 40 years and older are eligible to participate in the BreastScreen program, but active population recruitment is targeted at those aged 50–69 years. Screening is aimed at asymptomatic women. Women with symptoms or clinical findings should be investigated with triple testing — the combination of clinical, imaging and needle-biopsy evaluation.6
Mammography does not detect all breast cancers. A proportion are not detected through screening and emerge clinically — interval cancers7 (Box 2). Meta-analysis of the proportional incidence of interval breast cancers (the incidence of interval cancers as a proportion of the expected underlying incidence of breast cancer) shows rates between 19% and 27%.8 Adjunct imaging tests have therefore been used to maximise detection of breast cancer, and to potentially reduce the occurrence of interval cancers and extend the benefit of early detection. Studies of adjunct imaging in breast screening have generally focused on women at increased risk of breast cancer or those in whom mammography might be less sensitive.9
While the benefit of population breast screening is well established, a potential downside that has emerged in epidemiological studies is overdetection — the detection of cancer that would not have presented clinically during a woman’s lifetime (and therefore would not be diagnosed in the absence of screening).10 Overdetection is difficult to quantify validly, and most reported estimates are affected by methodological bias.10 Thus, we have not reported quantitative data, but believe overdetection represents a modest proportion of screen-detected breast cancers. Overdetection does not negate the benefit of breast screening. However, clinicians should be aware of it and might inform women of the possibility to assist informed participation in screening.
Overdetection is also relevant to adjunct screening: in the absence of RCTs, it is difficult to know how many of the additional cancers detected with an adjunct test represent early detection (likely to confer benefit) and how many represent overdetection (which will not confer benefit).10,11
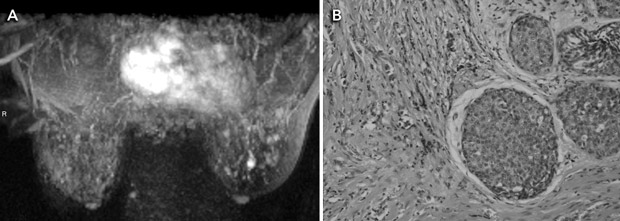
MRI is a highly sensitive test for detecting breast cancer. It is not recommended for screening women at average risk because it is associated with substantial numbers of false-positive results and cost. It has been proposed as adjunct screening for women with a genetic predisposition to breast cancer (Box 3). Mammography has lower sensitivity in this group, detecting fewer than half the breast cancers diagnosed.12
Women with inherited mutations of the breast cancer genes BRCA1 and BRCA2 represent a small proportion of those who develop breast cancer, but have a high lifetime risk of developing this disease.12 High-risk populations include women with known or suspected gene mutations, women with first-degree relatives who are known mutation carriers, and those with a family history suggesting inherited mutations. Diagnosis of breast cancer before the age of 40 years, several affected relatives, bilateral breast cancer, and the occurrence of breast and ovarian cancer in the same family or individual are features associated with inherited breast cancer. Current guidelines define high-risk categories and recommend annual surveillance commencing at age 35 years, or earlier if the youngest affected family member was diagnosed before 40 years of age.13,14
A meta-analysis of five studies of MRI as an adjunct to conventional imaging in high-risk women has provided convincing evidence that MRI detects additional cancers, with an incremental sensitivity of 58% (95% CI, 47%–70%) compared with mammography alone.15-20 Incremental sensitivity was lower when MRI was added to mammography combined with ultrasound examination (44%; 95% CI, 27%–61%) or to mammography combined with ultrasound plus clinical breast examination (range, 31%–33%).15 As summarised in Box 4, the addition of breast MRI led to the earlier detection of eight to 24 additional cancers per 1000 screens.15-20 Differences in MRI yield across studies reflected differences in comparator screening (mammography with or without ultrasound and clinical examination) and variations in disease prevalence. MRI provided the highest cancer yield when used in patients at highest risk.
The effect of detecting MRI-positive (mammogram-negative) cancers on breast cancer mortality, treatment morbidity or the incidence of interval cancers among high-risk (predominantly young) women has not yet been measured. Reductions in breast cancer mortality observed in trials of mammography screening among older, average-risk women have been attributed to the detection of breast cancer at an earlier stage, when the disease is more amenable to treatment. It is not yet clear whether earlier detection of cancers by MRI will bring the same benefits to high-risk women, who may represent a different prognostic group. Accuracy studies comparing tumour size and lymph-node spread between the extra cases detected by MRI and cases detected by conventional imaging alone have not provided consistent evidence that MRI produces a significant stage shift in the detection of breast cancer.15
MRI also increases the likelihood of recall for investigation of false-positive findings, although the evidence is not consistent across studies. Two studies observed that the addition of MRI produces at least a threefold increase in risk of undergoing percutaneous needle biopsy (PNB) for benign findings.17,18 One of these studies also reported a twofold increase in risk of surgical biopsy for benign findings.17 Another study did not find a significant difference in the rate of PNB when MRI was added to mammography, but did not report data on surgical biopsy.16
Australia’s Medical Services Advisory Committee assessed the evidence for breast MRI in 2007;21 subsequently, the Australian Minister for Health and Ageing recommended interim public funding for breast MRI in asymptomatic high-risk women when used as part of organised surveillance. A Medicare Benefits Schedule item number for breast MRI in women in specific high-risk groups was announced in February 2009. This recommendation will be reviewed in the future, as more evidence emerges, including longer follow-up data from existing studies. Similar policy recommendations have been made by the United Kingdom’s National Institute for Health and Clinical Excellence14 and the American Cancer Society.22
Breast ultrasound examination is not currently endorsed as a population-screening test, but has been applied in clinical practice for ad-hoc screening of women with dense breast tissue on mammography. Breast tissue density refers to the radiodense fibroglandular composition of the breast. High density of breast tissue is a significant and independent risk factor for breast cancer,23,24 and also reduces the sensitivity of mammography, as glandular tissue masks cancer on the mammogram. It is therefore not surprising that high density of breast tissue is associated with greater likelihood of an interval cancer in mammography screening,25 as women with dense breast tissue are both more likely to develop breast cancer and more likely to have cancer missed on mammography.
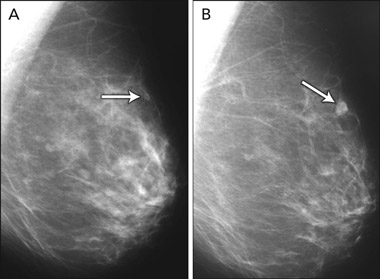
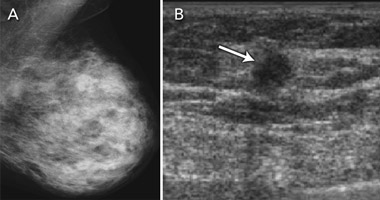
The accuracy of breast ultrasound examination is unaffected by breast tissue density. Ultrasonography has therefore been evaluated and applied in practice to “screen” women with dense breast tissue26-30 (Box 5). Evidence on its capacity to detect breast cancer in asymptomatic women with “negative” mammograms is summarised in Box 6. These studies provide data on additional (incremental) ultrasound detection — both true-positive and false-positive findings. The studies consistently show that ultrasonography is able to detect a substantial number of cancers in women with mammography-negative dense breast tissue, with additional cancers detected in 0.27%–0.46% of women screened with ultrasound.26-30 The detection capability of ultrasonography in this setting depends partly on how well mammography performs in the population being screened — “negative” mammograms may in fact show a cancer that has not been recognised by the screen-reader. This was demonstrated in a 2008 study that included “blinded” evaluation of the mammograms for ultrasound-only-detected cancers: about a quarter of ultrasound-detected cancers were perceived on the mammogram by an experienced radiologist.29 Another study found that the highest proportion of ultrasound-only-detected cancers (0.46%) occurred in women screened on the basis of dense breasts plus additional risk factors, with more than half of the screened women having a personal history of breast cancer.30 The false-positive rate for ultrasonography is considerable and varies between studies. False-positive results (in terms of both additional investigation and unnecessary surgical intervention) are the main potential limitation of ultrasonography in adjunct population screening.
The data in Box 6 also indicate that ultrasonography was able to detect breast cancers that were not identified in dense breasts on mammography screening at an early stage, generally at a comparable or earlier stage than cancers detected on mammography. This suggests that ultrasonography as an adjunct screening test may provide further benefit in screening women with dense breast tissue. It would thus be valuable to conduct large-scale RCTs of ultrasonography as an adjunct to mammography in screening women with dense breasts, with 2–3-year follow-up, to measure its impact on interval cancer rates. Demonstrating a reduction in interval cancers in women who have adjunct screening (relative to women who have mammography screening only) provides a surrogate indicator of longer-term benefit.11
Computer-aided detection (CAD) is an adjunct to mammography interpretation and not a standalone test. It does not have a defined role in screening in Australia at present, but may become more relevant in future. CAD is a complementary tool to mammography, which prompts the reader to review lesions on the mammogram and to consider recalling the patient. Overall, the evidence indicates that CAD does not perform as well as double (human) reading in the context of organised breast screening programs where double reading is the standard of care.31 However, published studies have shown that CAD improves the sensitivity of a single reader, with an incremental cancer detection rate (from adding CAD to a single read) ranging from 1% to 19%.32 The limitation of CAD is that it also significantly increases the recall rate (ie, decreases the reader’s specificity), causing additional recall in about 6%–35% of women.32 Although CAD identifies and prompts readers to “missed” cancers, the high number of false-positive prompts (1.5–4 false prompts per screening examination) can have an adverse effect on clinical decision making and has impeded CAD’s application in screening practice.32
Nevertheless, a recently reported RCT,33 the increasing implementation of digital mammography in Australia (which facilitates use of CAD), and ongoing refinement of CAD algorithms are expected to lead to a role for CAD in screening. An RCT based in the UK (CADET-II) recently demonstrated equivalence between double human reading and single reading with CAD (slightly higher recall using the latter).33 In this trial, mammography readers were highly experienced and were required to regularly interpret a large number of mammograms.33 The use of CAD by less experienced screen-readers may not achieve the same outcomes — it will be important to consider the data from this trial and to evaluate CAD in the context of Australian breast screening services.
1 Key concepts in breast cancer screening
Mammography is the only breast screening test for which benefit (mortality reduction) has been shown in randomised controlled trials (RCTs).
The benefit of early detection of breast cancer (based on mammography screening) has been shown in women with population (average) risk aged 50–69 years; women aged 40–49 years and those aged ≥ 70 years may also benefit from screening.
New breast imaging tests have been examined in studies that compare the test’s ability to detect cancer relative to mammography (as an additional test or adjunct screening, or as a replacement to mammography) in special populations selected on the basis of their risk or breast tissue density.
It is assumed that if a new test finds additional breast cancers not detected by mammography, then this will provide further benefit (ie, may further reduce breast cancer deaths), but this remains uncertain and has not been examined in RCTs.
It is assumed that early detection in women at particularly elevated risk of breast cancer, including women younger than 40 years (in whom prospective RCTs have never been conducted), confers benefit, but there are no data to support or disprove this.
4 Evidence on magnetic resonance imaging (MRI) as an adjunct to mammography (with or without ultrasonography) in screening young women at high risk of breast cancer
6 Evidence on ultrasonography (US) in screening asymptomatic women with mammography-negative dense breast tissue
- Nehmat Houssami1
- Sarah J Lord1,2
- Stefano Ciatto3
- 1 Screening and Test Evaluation Program, School of Public Health, Faculty of Medicine, University of Sydney, Sydney, NSW.
- 2 Clinical Trials Centre, Faculty of Medicine, University of Sydney, Sydney, NSW.
- 3 Istituto per lo Studio e la Prevenzione Oncologica, Istituto Scientifico della Regione Toscana, Florence, Italy.
We thank Dr Ruth M L Warren (Department of Radiology, University of Cambridge, United Kingdom) for the MRI images.
None identified.
- 1. International Agency for Research on Cancer. IARC handbooks of cancer prevention: breast cancer screening. Lyon: IARC, 2002.
- 2. Gabe R, Duffy SW. Evaluation of service screening mammography in practice: the impact on breast cancer mortality. Ann Oncol 2005; 16 Suppl 2: ii153-ii162.
- 3. Roder DM, Houssami N, Farshid G, et al. Population screening and intensity of screening are associated with reduced breast cancer mortality: evidence of efficacy of mammography screening in Australia. Breast Cancer Res Treat 2008; 108: 409-416.
- 4. Moss SM, Cuckle H, Evans A, et al. Effect of mammographic screening from age 40 years on breast cancer mortality at 10 years’ follow-up: a randomised controlled trial. Lancet 2006; 368: 2053-2060.
- 5. Barratt AL, Irwig LM, Glasziou PP, et al. Benefits, harms and costs of screening mammography in women 70 years and over: a systematic review. Med J Aust 2002; 176: 266-271. <MJA full text>
- 6. Irwig L, Macaskill P, Houssami N. Evidence relevant to the investigation of breast symptoms: the triple test. Breast 2002; 11: 215-220.
- 7. Houssami N, Irwig L, Ciatto S. Radiological surveillance of interval breast cancers in screening programmes. Lancet Oncol 2006; 7: 259-265.
- 8. Taylor R, Supramaniam R, Rickard M, et al. Interval breast cancers in New South Wales, Australia, and comparisons with trials and other mammographic screening programmes. J Med Screen 2002; 9: 20-25.
- 9. Irwig L, Houssami N, van Vliet C. New technologies in screening for breast cancer: a systematic review of their accuracy. Br J Cancer 2004; 90: 2118-2122.
- 10. Biesheuvel C, Barratt A, Howard K, et al. Effects of study methods and biases on estimates of invasive breast cancer overdetection with mammography screening: a systematic review. Lancet Oncol 2007; 8: 1129-1138.
- 11. Irwig L, Houssami N, Armstrong B, Glasziou P. Evaluating new screening tests for breast cancer [editorial]. BMJ 2006; 332: 678-679.
- 12. Houssami N, Cuzick J, Dixon JM. The prevention, detection, and management of breast cancer. Med J Aust 2006; 184: 230-234. <MJA full text>
- 13. National Breast Cancer Centre. Advice about familial aspects of breast cancer and epithelial ovarian cancer. Sydney: NBCC, 2006. http://www.nbocc.org.au/bestpractice/resources/BOG182_adviceaboutfamiliala.pdf (accessed Sep 2008).
- 14. National Collaborating Centre for Primary Care. Familial breast cancer: the classification and care of women at risk of familial breast cancer in primary, secondary and tertiary care (partial update of Clinical Guideline 14). London: National Institute for Health and Clinical Excellence, 2006. http://www.nice.org.uk/Guidance/CG41 (accessed Sep 2008).
- 15. Lord SJ, Lei WA, Craft P, et al. A systematic review of the effectiveness of magnetic resonance imaging (MRI) as an addition to mammography and ultrasound in screening young women at high risk of breast cancer. Eur J Cancer 2007; 43: 1905-1917.
- 16. Kuhl CK, Schrading S, Leutner CC, et al. Mammography, breast ultrasound, and magnetic resonance imaging for surveillance of women at high familial risk for breast cancer. J Clin Oncol 2005; 23: 8469-8476.
- 17. Leach MO, Boggis CR, Dixon AK, et al; MARIBS study group. Screening with magnetic resonance imaging and mammography of a UK population at high familial risk of breast cancer: a prospective multicentre cohort study (MARIBS). Lancet 2005; 365: 1769-1778.
- 18. Lehman CD, Blume JD, Weatherall P, et al. Screening women at high risk for breast cancer with mammography and magnetic resonance imaging. Cancer 2005; 103: 1898-1905.
- 19. Sardanelli F, Podo F, D’Agnolo G, et al. Multicenter comparative multimodality surveillance of women at genetic-familial high risk for breast cancer (HIBCRIT study): interim results. Radiology 2007; 242: 698-715.
- 20. Warner E, Plewes DB, Hill KA, et al. Surveillance of BRCA1 and BRCA2 mutation carriers with magnetic resonance imaging, ultrasound, mammography, and clinical breast examination. JAMA 2004; 292: 1317-1325.
- 21. The Medical Services Advisory Committee. Breast magnetic resonance imaging. MSAC application 1098. Canberra: Commonwealth of Australia, 2007.
- 22. Saslow D, Boetes C, Burke W, et al. American Cancer Society guidelines for breast screening with MRI as an adjunct to mammography. CA: Cancer J Clin 2007; 57: 75-89.
- 23. McCormack VA, dos Santos Silva I. Breast density and parenchymal patterns as markers of breast cancer risk: a meta-analysis. Cancer Epidemiol Biomarkers Prev 2006; 15: 1159-1169.
- 24. Kerlikowske K, Ichikawa L, Miglioretti DL, et al. Longitudinal measurement of clinical mammographic breast density to improve estimation of breast cancer risk. J Natl Cancer Inst 2007; 99: 386-395.
- 25. Ciatto S, Visioli C, Paci E, Zappa M. Breast density as a determinant of interval cancer at mammographic screening. Br J Cancer 2004; 90: 393-396.
- 26. Buchberger W, Niehoff A, Obrist P, et al. Clinically and mammographically occult breast lesions: detection and classification with high resolution sonography. Semin Ultrasound CT MR 2000; 21: 325-336.
- 27. Kaplan SS. Clinical utility of bilateral whole breast US in the evaluation of women with dense breast tissue. Radiology 2001; 221: 641-649.
- 28. Kolb TM, Lichy J, Newhouse JH. Comparison of the performance of screening mammography, physical examination and breast US and evaluation of factors that influence them; an analysis of 27,825patient evaluations. Radiology 2002; 225: 165-175.
- 29. Corsetti V, Houssami N, Ferrari A, et al. Breast screening with ultrasound in women with mammography-negative dense breasts: evidence on incremental cancer detection and false positives, and associated cost. Eur J Cancer 2008; 44: 539-544.
- 30. Berg WA, Blume JD, Cormack JB, et al. Combined screening with ultrasound and mammography vs mammography alone in women at elevated risk of breast cancer. JAMA 2008; 299: 2151-2163.
- 31. Taylor P, Potts HWW. Computer aids and human second reading as interventions in screening mammography: two systematic reviews to compare effects on cancer detection and recall rate. Eur J Cancer 2008; 44: 798-807.
- 32. Houssami N, Given-Wilson R, Ciatto S. Overview of the evidence on computer-aided detection (CAD) in mammography screening. J Med Imaging Radiat Oncol 2009. In press.
- 33. Gilbert FJ, Astley SM, Gillan MGC, et al (for the CADET II Group). Single reading with computer-aided detection for screening mammography. N Engl J Med 2008; 359: 1675-1684.





Abstract
Early detection of breast cancer has been shown to reduce breast cancer deaths in randomised controlled trials (RCTs) of mammography in women aged 50–69 years, with weaker evidence of benefit in those aged 40–49 or 70 years and older.
Magnetic resonance imaging (MRI) and ultrasonography have been evaluated in breast cancer screening, relative to, or in addition to, mammography, in selected populations; neither test has been examined in an RCT, and thus evidence of associated screening benefit is uncertain.
MRI is more sensitive than mammography in screening women with suspected or proven inherited mutations of the breast cancer genes. The addition of MRI in screening this population detects 8–24 additional cancers per 1000 screens, but also significantly increases a woman’s risk of being recalled for investigation or surgical biopsy for false-positive findings. In Australia, Medicare funding for MRI screening of women in specific risk groups was announced in February 2009.
Ultrasonography can detect cancers not identified on mammography in asymptomatic women with dense breast tissue. Incremental ultrasound cancer detection is reported in 0.27%–0.46% of women with mammography-negative dense breasts; evidence varies on its association with false-positive findings.
Computer-aided detection (CAD) is a complementary tool to mammography, prompting the reader to consider lesions on the mammogram that may represent cancer. Emerging evidence and improved CAD technology are likely to help define its role in breast screening.