A 51-year-old man with no known history of asbestos exposure presented with hydropneumothorax. Soluble mesothelin-related protein testing and combined positron emission tomography and computed tomography were used to diagnose malignant pleural mesothelioma. One year after radical surgery and radiotherapy, there was no clinical recurrence.
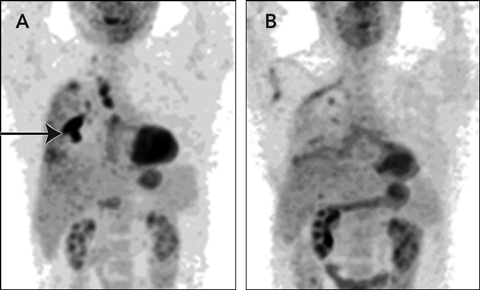
The patient remained well on follow-up. His lung function improved and, despite dyspnoea during exercise, he walked 20 km per week. Serial chest radiographs showed significant pleural thickening but no other abnormality. However, as the cause of the pleural effusion remained undiagnosed, the serum soluble mesothelin-related protein (SMRP) level was measured. At 2.8 nM, it was higher than the reference range (< 2.5 nM),1 suggesting malignant pleural mesothelioma (MPM). The biopsy specimens collected at presentation were reviewed, but no evidence of malignancy was found. A combined positron emission tomography/computed tomography (PET/CT) scan revealed intense tracer uptake (Box, A), suggesting MPM involving the pleura at the right lower lobe and apex, with minimal uptake in the paratracheal and subcarinal lymph nodes. Results of all other investigations (eg, full blood count, liver function tests, and measurement of creatinine and blood glucose levels) were normal.
The patient recovered well and returned to part-time work. Some residual neuropathic pain arising from the thoracotomy scar was controlled with medication. One year after surgery, a repeat PET/CT scan showed no residual tumour or uptake of tracer in the lymph nodes (Box, B), and the SMRP level was within the reference range. He remained well 19 months after surgery.
Until recently, MPM was a relatively rare disease, with an incidence of 1–2 cases per million per year in the general population. However, its incidence is increasing, and Australia now has the highest reported incidence in the world.2,3 The increasing incidence is explained by high asbestos use from the 1940s to the 1980s and the long latency period of this tumour.3,4 In Australia, about 90% of men with MPM have a history of occupational asbestos exposure.2 Patients usually present with pleural effusion and breathlessness, but symptoms are non-specific in many cases — particularly in the early stages of disease.
MPM is difficult to diagnose in its early stages and, to date, no treatments have been shown to increase life expectancy. However, several new drugs have recently become available, and trials evaluating whether early treatment (including surgery combined with radiotherapy and chemotherapy) improves survival are underway. Ideally, early detection would allow intervention to control or eradicate the neoplasm. There is also considerable interest in the use of biomarkers to facilitate early detection of several malignancies, including MPM. SMRP is increasingly being used to detect MPM after clinical presentation5,6 and has recently been approved in the United States for diagnosis and monitoring of MPM.
Pleural effusions are commonly seen in clinical practice. However, despite advances in cytological techniques and application of Light’s criteria for distinguishing between exudative and transudative pleural effusions,7 diagnosis of pleural malignancy is often delayed. Closed needle biopsy is seldom used in Australia, as its sensitivity in malignant pleural effusions (particularly MPM) is low. For patients with a negative result on cytological analysis of pleural fluid, a repeat closed needle biopsy returns a positive result in only 7%8 and could seed tumour along the biopsy track. To improve sensitivity, the pleural biopsy specimen needs to be large, hence video-assisted thoracoscopic surgery (VATS) and open pleural biopsies are now the procedures of choice in suspected MPM. They also have the advantage of enabling concurrent pleurodesis. With large tissue samples, open biopsy has a sensitivity of 97% and specificity of 56% for identifying epithelial MPM.9 VATS is associated with some risks, but overall these are low. The overall incidence of postoperative complications is 10.9%, including prolonged air leak (6.7%) and recurrent pleural effusion (0.7%).10 Early pleurodesis has been shown to significantly improve quality of life in patients with MPM, and is currently recommended in the British Thoracic Society’s guidelines on the management of malignant mesothelioma.11
Mesothelin is a 40 kDa protein that is important in cell adhesion. It is a differentiation antigen, and is present on normal mesothelial cells of the pleura, peritoneum and pericardium.5 SMRPs are thought to be either cleaved peptide fragments of mesothelin, or abnormal variants of mesothelin that are unable to bind to membranes and are overexpressed in several human tumours, including MPM.
Several studies have examined SMRP testing for diagnosis and monitoring of MPM.5,6,12 A recent study showed that blood SMRP level had a specificity of 95% and sensitivity of 83% in identifying patients with MPM.6 However, an elevated SMRP level can also occur in metastatic disease, especially ovarian cancer, pancreatic cancer and adenocarcinomas, and its use for screening in the absence of symptoms is unproven.12
PET/CT scanning is a new technique that has been shown to be useful in patients with MPM for predicting survival and evaluating response to chemotherapy.13,14 However, few data are available on its use as a diagnostic tool. In 28 patients with suspected MPM, PET/CT imaging was compared with VATS and surgical biopsies. Fluorodeoxyglucose-PET imaging was sensitive (91%) and highly specific for malignancy (100%), although the activity of some epithelial MPMs was close to the threshold of abnormality.15
- 1. Creaney J, van Bruggen I, Hof M, et al. Combined CA 125 and mesothelin levels for the diagnosis of malignant mesothelioma. Chest 2007; 132: 1239-1246.
- 2. Leigh J, Driscoll T. Malignant mesothelioma in Australia, 1945-2002. Int J Occup Environ Health 2003; 9: 206-217.
- 3. Hyland R, Ware S, Johnson A, Yates DH. Malignant mesothelioma in women in New South Wales. Scand J Work Environ Health 2007; 33: 286-292.
- 4. Yates DH, Corrin B, Stidolph PN, Browne K. Malignant mesothelioma in south east England: clinicopathological experience of 272 cases. Thorax 1997; 52: 507-512.
- 5. Maeda M, Hino O. Molecular tumor markers for asbestos-related mesothelioma: serum diagnostic markers. Pathol Int 2006; 56: 649-654.
- 6. Robinson BW, Creaney J, Lake R, et al. Soluble mesothelin-related protein — a blood test for mesothelioma. Lung Cancer 2005; 49 Suppl 1: S109-S111.
- 7. Light RW, MacGregor MI, Luchsinger PC, Ball WC. Pleural effusions: the diagnostic separation of transudates and exudates. Ann Intern Med 1972; 77: 507-513.
- 8. Prakash UB, Reiman HM. Comparison of needle biopsy with cytological analysis for the evaluation of pleural effusion: analysis of 414 cases. Mayo Clin Proc 1985; 60: 158-164.
- 9. Bueno R, Reblando J, Glickman J, et al. Pleural biopsy: a reliable method for determining the diagnosis but not subtype in mesothelioma. Ann Thorac Surg 2004; 78: 1774-1776.
- 10. Jancovic R, Lang-Lazdunski L, Pons F, et al. Complications of video-assisted thoracic surgery: a five-year experience. Ann Thorac Surg 1996; 61: 533-537.
- 11. British Thoracic Society Standards of Care Committee. BTS statement on malignant mesothelioma in the UK, 2007. Thorax 2007; 62 Suppl 2: ii1-ii19.
- 12. Park EK, Sandrini A, Yates DH, et al. Soluble mesothelin-related protein (SMRP) in an asbestos-exposed population: the dust diseases board cohort study. Am J Respir Crit Care Med 2008; 178: 832-837.
- 13. Flores R, Akhurst T, Gonen M, et al. Positron emission tomography predicts survival in malignant pleural mesothelioma. J Thorac Cardiovasc Surg 2006; 132: 763-768.
- 14. Ceresoli GL, Chiti A, Zucali PA, et al. Early response evaluation in malignant pleural mesothelioma by positron emission tomography with [18F]fluorodeoxyglucose. J Clin Oncol 2006; 24: 4587-4593.
- 15. Bénard F, Sterman D, Smith RJ, et al. Metabolic imaging of malignant pleural mesothelioma with fluorodeoxyglucose positron emission tomography. Chest 1998; 114: 713-722.





Deborah Yates has received several research grants from the Dust Diseases Board Research Grants Scheme in the area of asbestos-related disease.