Pelvic osteomyelitis is a rare occurrence, with reported rates of 1%–11% of all cases of acute haematogenous osteomyelitis (AHO) occurring between the ages of 7 and 14 years.1 In a review of the medical records of 220 children with AHO, 19 children had pelvic osteomyelitis.2 A review of cases of AHO at our institution showed infection of the pelvis in 8% of 102 patients with AHO and a male : female ratio of 2.1 : 1.3
Pelvic osteomyelitis has been described in three entities — gluteal, lumbar and abdominal.4 Although these designations can be useful for determining the spread of infection, they have no prognostic value.4,5
Acute pelvic osteomyelitis is often not initially recognised. It can be difficult to detect due to its variable clinical signs and differential diagnoses.1 In one study, the time span between initial symptoms and the diagnosis ranged from 1 to 8 days.2 Difficulty diagnosing the condition may result in a delay of appropriate treatment.1,6,7
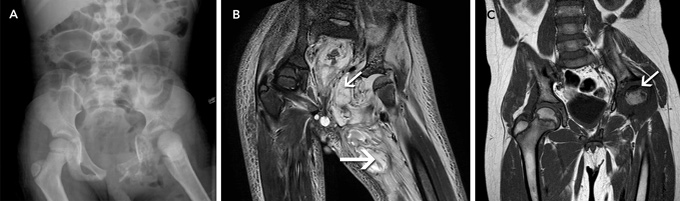
In our patient, plain radiographs (which are usually normal for the first 7–10 days8) and magnetic resonance imaging (MRI) scans showed extensive bony and soft tissue destruction of the left hemipelvis, hip and thigh. Is it plausible that these changes occurred within 2 weeks, considering the patient’s history? As we were unable to find supporting reports in the literature, this matter was discussed within various hospital departments. Concerns were raised that the time frame was too short to cause this level of destruction. It was proposed that the mixed flora that were cultured could cause a high level of destruction within 14 days. Although plausible, there was no direct evidence to support this theory.
When during the course of the infection did the bowel perforation occur? In the literature, we found two case reports of a bowel perforation associated with pelvic osteomyelitis. Both occurred in adult patients, and bony infection was secondary to Crohn disease in both cases.9,10 On arrival at our hospital, our patient’s radiographs showed dilated loops of bowel and no signs of free gas. The MRI scan showed oedema and signs of inflammation of the soft tissue at the perineum, surrounding the rectum, urethra, bladder and sigmoid colon. Free fluid was seen within the pelvis; however, there was no obvious pelvic collection. Enteric flora were cultured from the initial intra-operative swabs. These factors indicate that the bowel perforation was probably present on arrival, possibly as a sealed perforation, which was only detected 5 days after the initial debridement. Although we were unable to ascertain the cause of the bowel perforation, we believe that its association with pelvic osteomyelitis was significant.
We recommend that sophisticated imaging studies be performed at an early stage. MRI is the most sensitive test for osteomyelitis of the pelvis and spine, with a reported sensitivity of 97%–100% and specificity of 73%–92%.11
Staphylococcus aureus is the most common causative organism in AHO, accounting for 76% of cases, with an increased emergence of community-acquired methicillin-resistant Staphylococcus aureus (MRSA) accounting for 9% of these.3 Aggressive community-acquired MRSA, which was isolated from the initial swabs, was thought to be the causative organism of this patient’s pelvic osteomyelitis. We believe that the late diagnosis and the bowel perforation contributed to the extraordinary tissue destruction described in this article. We believe that the extent of the patient’s recovery, including the partial repair of bony destruction, was remarkable.
Most cases of pelvic osteomyelitis reported in the literature were treated adequately with antibiotics alone; surgical intervention was rarely required.1,5 Complications, including recurrence and permanent sequelae, were uncommon, with rates reported between zero and 7%.1,5 Increasing rates of community-acquired MRSA require clinicians to consider whether a change to more efficacious antibiotic therapy is necessary if the patient is not responding to flucloxacillin alone.
Unusual circumstances that delay the diagnosis can always occur, particularly in rural communities, where access to sophisticated imaging is limited.
Lessons from practice
Negligible accidents can form the starting point of life-threatening osteomyelitis, particularly in the pelvic region. To avoid major complications, it is important to repeat physical examinations within a short time period (ie, daily).
If any doubt arises regarding a correct diagnosis, check for inflammatory parameters and refer the patient to a major hospital for sophisticated imaging and commencement of appropriate treatment.
Consider magnetic resonance imaging early, as this is the most sensitive method for detecting (pelvic) osteomyelitis.
- 1. Highland TR, LaMont RL. Osteomyelitis of the pelvis in children. J Bone Joint Surg Am 1983; 65: 230-234.
- 2. Weber-Chrysochoou C, Corti N, Goetschel P, et al. Pelvic osteomyelitis: a diagnostic challenge in children. J Pediatr Surg 2007; 42: 553-557.
- 3. Goergens ED, McEvoy A, Watson M, Barrett IR. Acute osteomyelitis and septic arthritis in children. J Paediatr Child Health 2005; 41: 59-62.
- 4. Beaupré A, Carroll N. The three syndromes of iliac osteomyelitis in children. J Bone Joint Surg Am 1979; 61: 1087-1092.
- 5. Davidson D, Letts M, Khoshhal K. Pelvic osteomyelitis in children: a comparison of decades from 1980–89 with 1990–2001. J Pediatr Orthop 2003; 23: 514-521.
- 6. Zvulunov A, Gal N, Segev Z. Acute hematogenous osteomyelitis of the pelvis in childhood: diagnostic clues and pitfalls. Pediatr Emerg Care 2003; 19: 29-31.
- 7. Rand N, Mosheiff R, Matan Y, et al. Osteomyelitis of the pelvis. J Bone Joint Surg Br 1993; 75: 731-733.
- 8. Capitanio MA. Early roentgen observations in acute osteomyelitis. Am J Roentgenol Radium Ther Nucl Med 1970; 108: 488-496.
- 9. Armstrong N, Schurr M, Helgerson R, Harms B. Fungal sacral osteomyelitis as the initial presentation of Crohn’s disease of the small bowel, report of a case. Dis Colon Rectum 1998; 41: 1581-1584.
- 10. Chandler JT, Riddle CD Jr. Osteomyelitis associated with Crohn’s disease. A case report and literature review. Orthopedics 1989; 12: 285-288.
- 11. Mazur JM, Ross G, Cummings J, et al. Usefulness of magnetic resonance imaging for the diagnosis of acute musculoskeletal infections in children. J Pediatr Orthop 1995; 15: 144-147.





None identified.