In Western countries, about 60% of people over the age of 60 years will develop colonic diverticula. The increasing incidence in developed countries is directly related to reduced fibre intake following the introduction of steel rolling mills. Men and women are affected equally. However, only 10%–25% of those affected will have symptoms, with 15% of symptomatic patients suffering significant complications. In patients requiring urgent surgical intervention, overall mortality is between 12% and 36%.1
The sigmoid colon is the most common site for formation of diverticula, as it has the smallest diameter in the colon and a different motility pattern and is less compliant than the ascending colon.2 Intraluminal pressures in the colon have been shown to be nine times higher in patients with diverticular disease than in control patients. Right-sided colonic diverticula, though rare, occur more commonly among Asian people, perhaps due to a genetic predisposition.
Low dietary fibre intake reduces stool bulk and prolongs transit time, resulting in increased intraluminal pressure.2 Recent observations suggest diverticular disease may be related to long-term alterations in diet and to the patient’s microecology. A low-fibre diet alters the host’s microflora, resulting in changes to the colonic immune system.3 Slower transit time leads to increased exposure to intraluminal antigens.
A proportion of patients suffer disabling recurrent abdominal pain and altered bowel habit similar to irritable bowel syndrome. They may have changes in gut motility and sensitivity as a result of mucosal inflammation. It has been suggested that diverticular disease may be a form of chronic inflammatory bowel disease and that its recurrent nature may be controlled by altering the inflammatory response.3
Non-steroidal anti-inflammatory drugs have been implicated as a possible factor contributing to bleeding and perforation from diverticular disease.1 These drugs reduce gut prostaglandin synthesis, which affects the maintenance of the mucosal barrier and results in mucosal damage, increased colonic permeability and bacterial translocation.
Diverticulosis usually causes no symptoms. Its acute presentation is due to an obstructing faecalith, which results in micro-perforation of the diverticulum and secondary infection of the colon. The inflammation is extracolonic, with minimal intraluminal evidence of diverticulitis at colonoscopy during an acute attack. The extent of perforation will determine the presentation, which can range in severity from diverticulitis to faecal peritonitis (Box 1). This can be rapidly assessed with computed tomography (CT) scanning, which is the imaging modality of choice.5 It is very sensitive and has superseded contrast enemas, which carry a risk of perforation and barium leaks.
Most symptomatic patients (85%) can be managed non-operatively. About a third of patients will have a recurrent attack.6 Uncomplicated diverticulitis can be treated with bowel rest, broad-spectrum antibiotics and slow progression to a high-fibre diet. With appropriate antibiotic therapy, small abscesses will regress, possibly due to internal drainage. Abscesses less than 2 cm in diameter can be safely managed in an outpatient setting with oral antibiotics and sports drink supplementation.7
Pericolic abscesses larger than 5 cm in diameter can be drained percutaneously with CT guidance.5 This permits rapid control of sepsis, with success rates of up to 80%, and allows for subsequent single-stage resection. Following drainage, some patients may have a persistent symptomatic fistula caused by a faecalith obstructing the diverticular neck. Drainage alone is insufficient to control sepsis. If faecal drainage continues, surgical intervention is usually required.
Mayo first introduced surgical resection for diverticulitis in 1907. Proximal transverse colostomy without resection of the diseased colon was a common method in the early 1900s.8 In 1942, Smithwick recommended a three-stage approach, as he found that the inflammatory process did not resolve without resection. This involved a proximal colostomy initially, followed by sigmoid resection, and finally, closure of the diverting stoma.9 The three-stage resection later fell out of favour, as mortality in patients with peritonitis was high if the diseased sigmoid was left behind.10 A two-stage procedure subsequently evolved. In the 1950s, the Hartmann procedure, involving sigmoid colon resection and end colostomy, was frequently performed for complicated diverticular disease. Operative mortality was almost halved.10
A trend towards single-stage resection was advocated in the early 1980s. The use of a one-stage procedure had earlier been limited by concerns about anastomotic leakage in the presence of peritoneal contamination, colonic inflammation and unprepared bowel. The risk of anastomotic leakage after resection for diverticular disease is a concern for several reasons, which include the presence of inflammation, shortening of the colon due to inflammatory fibrosis, muscular abnormalities, and the presence of diverticula at the site of anastomosis. In recent years, a number of authors have reported successes in treatment with primary anastomosis without colonic lavage.11-13 Good anastomotic integrity is influenced by a good blood supply and tension-free technique.11 A single-stage resection benefits the patient by avoiding a second operation for bowel restoration and its associated complications. The offset is a longer operative time and increased risk of anastomotic leak.
Guidelines issued by the American Society of Colon and Rectal Surgeons now recommend elective colectomy (based on individual circumstances) after a single attack of acute diverticulitis or after non-operative management of complicated diverticulitis.6 Previously, elective resection had been recommended after two episodes of uncomplicated diverticulitis. This was based on historical data suggesting that recurrent attacks were less likely to respond to non-operative therapy: between 1950 and 1961, it was observed in 521 cases of diverticulitis that 70% responded during the first episode but only 6% after the third episode.14
However, recent studies have not found any evidence that patients having subsequent uncomplicated attacks are less likely to respond to non-operative therapy.4,15,16 This has reduced support for routine elective resection. Advances in intensive care, antibiotics and application of CT-guided drainage procedures may account for the successful treatment of most patients in recent years. In a review of 10 studies on the natural history of diverticular disease, Janes and colleagues concluded that the probability of readmission with each subsequent attack diminishes and that there is limited evidence to suggest that patients are more likely to suffer complications.4 After recovering from an episode of diverticulitis, the risk of an individual requiring an urgent Hartmann procedure is 1 in 2000 patient-years of follow-up.4
Recent evidence has also shown that prophylactic resections are of little benefit in preventing subsequent complications.2 Expectant management, rather than elective resection, was associated with fewer deaths, 2% fewer colostomies and greater cost-effectiveness.
Contrary to previous opinion, diverticular disease may not be more aggressive in the young, as bad experiences usually result from late presentations or missed diagnoses.4 Younger patients have a longer life span and may have a higher cumulative risk for recurrent diverticulitis, but the number of attacks is not necessarily an over-riding factor in deciding whether surgery is warranted.6
The presence of specific comorbid conditions such as immunosuppression may justify resection after one episode of diverticulitis, as patients with such conditions are more prone to septic complications.17 They also suffer higher postoperative morbidity and mortality owing to delayed diagnosis and treatment. Patients awaiting organ transplantation also warrant similar consideration.
Complicated diverticular disease is defined as diverticulitis associated with abscess, fistula, bleeding, obstruction, phlegmon or perforation.4 Many patients presenting with complicated diverticulitis have no history of diverticular disease. In a cohort study of 330 patients with complicated diverticulitis, 54.6% had presented with this condition as the first manifestation of their disease.18 In another cross-sectional study, 78% of patients with perforated diverticulitis had no history of diverticulitis.19 The indications for emergency surgery include purulent or faecal peritonitis, uncontrolled sepsis, obstruction, fistula and the inability to exclude carcinoma.
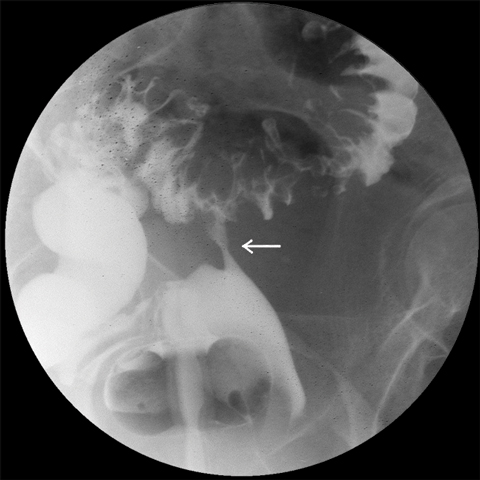
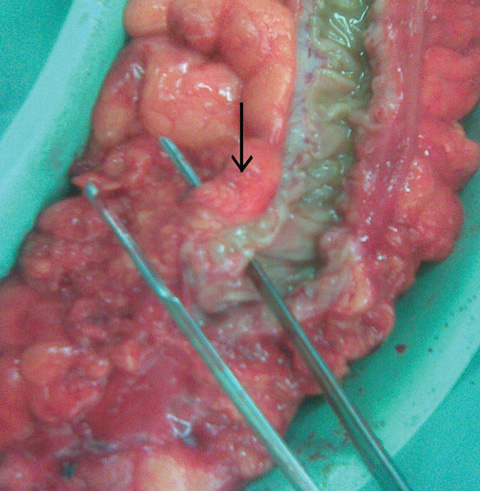
Colovesical fistulas are the most common fistulas associated with diverticular disease. They are more common in men, due to the barrier effect of the uterus in women, but colovesical fistulas can occur in women who have undergone hysterectomies. A one-stage resection with excision of the fistula from the adjacent organ is usually sufficient (Box 2, Box 3).
Right-sided diverticular disease tends to present more often with massive bleeding than left-sided disease. In a study of an Asian population, intervention for bleeding occurred more often for right-sided disease (41%) than for left-sided disease (8%).20 A thinner right colonic wall may make vessels more vulnerable to injury. Surgical intervention for diverticular bleeding should be reserved for cases in which medical, endoscopic or angiographic techniques have failed. If a definite site of colonic bleeding is not identified preoperatively, a subtotal colectomy with ileorectal anastomosis is warranted, as the right colon can be a common site of bleeding.
The Hartmann procedure still remains a safe option for managing patients who have complicated diverticular disease or are in poor condition. However, this procedure carries a significant risk of morbidity (23%–95%) and mortality (7%–28%).21 The presence of premorbid conditions, such as old age, collagen disease and cardiorespiratory disease, significantly increases the risk of postsurgical complications.18 The optimal timing for reversal of the Hartmann procedure is unclear. One study with a cut-off period of 3 months reported no difference in morbidity with early or delayed reversal.22 An earlier study reported better results after early closure (at 1 month) than after later closure (at 4 months). They noted more dense adhesions and less accessible rectal stumps in the delayed-reversal group.23 Sometimes the reversal may never eventuate, owing to poor medical health. About a third of patients retain their stoma for life.
A single-stage operation is advantageous because of the higher cumulative morbidity and mortality associated with multiple-stage resections. Primary anastomosis performed with or without bowel preparation has been shown to be safe in an emergency setting. Intraoperative colonic lavage before anastomosis may be unnecessary. It prolongs operative time and potentially increases the risk of faecal contamination.11 Schilling and colleagues reported no anastomotic leakages in their management of 13 patients with perforated sigmoid diverticulitis.13 Colonic lavage was not performed during the procedures. Morbidity and mortality in this group were similar to morbidity and mortality in another group who underwent a two-stage procedure.
In a systematic review of 98 published studies, Salem and Flum reported that mortality and morbidity in patients with diverticular peritonitis who underwent a primary anastomosis were no higher than in patients who underwent a Hartmann procedure.24 In 50 studies reporting 569 cases of primary anastomosis, they found that mortality was 9.9%, with an anastomotic leak rate of 13.9%. These rates were lower in patients who additionally had either a protective stoma or an intraoperative colonic lavage. In comparison, among 1051 patients who underwent a Hartmann procedure, mortality was 19.6%, with 10.3% having stoma complications and 4.3% having anastomotic leaks. The authors concluded that primary anastomosis is a safe alternative in selected patients.
Another recent systematic review by Constantinides and colleagues found no significant differences between mortality after primary resection with anastomosis and mortality after the Hartmann procedure in trials matched for degree of severity of peritonitis.25 The authors believed that the retrospective nature of these studies allowed for considerable selection bias, which limited their conclusions. They did not identify any beneficial effect of intracolonic lavage or faecal diversion in reducing postoperative mortality. Neither was there any significant difference in operative time between the two operations. It is possible that if surgeons are performing many primary anastomoses safely, they may be operating too often on patients with mild pathology that would have settled without operative intervention.
Several groups have found that laparoscopic drainage of a diverticular abscess can be effective and may obviate subsequent resection. Mutter and colleagues advocated an alternative two-stage laparoscopic approach to treating generalised peritonitis secondary to perforated diverticulitis: a limited primary laparoscopic lavage and drainage were performed to control intra-abdominal sepsis;26 any adhesions were untouched, to avoid disturbing a sealed perforation; and a passive drain was left exteriorising through the left iliac fossa. This converted a generalised purulent peritonitis into a localised diverticulitis, which could then be managed with antibiotic therapy. Once the inflammation had settled, a delayed laparoscopic resection could be performed in a non-emergency setting.
A similar approach using fibrin glue has been proposed. Faranda and colleagues treated 18 patients with laparoscopic peritoneal lavage, then applied fibrin glue over the infected sigmoid colon.27 Four patients required suturing of a perforation and six patients required an omentoplasty. A drain was inserted and antibiotic treatment was continued postoperatively. Thirteen patients later underwent an elective laparoscopic colectomy. The initial perforation was not found in all pathological specimens, and no patients developed an intraperitoneal abscess.
Such techniques are advantageous in that a laparotomy and stoma can be avoided.
- Kevin Ooi1
- Shing W Wong2
- Department of Surgery, Prince of Wales Hospital and University of New South Wales, Sydney, NSW.
None identified.
- 1. Morris CR, Harvey IM, Stebbings WSL, et al. Epidemiology of perforated colonic diverticular disease. Postgrad Med J 2002; 78: 654-658.
- 2. Janes SJ, Meagher A, Frizelle FA. Management of diverticulitis. BMJ 2006; 332: 271-275.
- 3. Floch MH, White J. Diverticulitis: new concepts and new therapies. J Clin Gastroenterol 2005; 39: 355-356.
- 4. Janes S, Meagher A, Frizelle FA. Elective surgery after acute diverticulitis. Br J Surg 2005; 92: 133-142.
- 5. Kaiser AM, Jiang JK, Lake JP, et al. The management of complicated diverticulitis and the role of computer tomography. Am J Gastroenterol 2005; 100: 910-917.
- 6. Rafferty J, Shellito P, Hyman NH, Buie WD; Standards Committee of American Society of Colon and Rectal Surgeons. Practice parameters for sigmoid diverticulitis. Dis Colon Rectum 2006; 49: 939-944.
- 7. Mizuki A, Nagata H, Tatemichi M, et al. The outpatient management of patients with acute mild-to-moderate colonic diverticulitis. Aliment Pharmacol Ther 2005; 21: 889-897.
- 8. Vargas HD, Ramirez RT, Hoffman GC, et al. Defining the role of laparoscopic-assisted sigmoid colectomy for diverticulitis. Dis Colon Rectum 2000; 43: 1726-1731.
- 9. Pearce NW, Scott SD, Karran SJ. Timing and method of reversal of Hartmann’s procedure. Br J Surg 1992; 79: 839-841.
- 10. Krukowski ZH, Matheson NA. Emergency surgery for diverticular disease complicated by generalized and faecal peritonitis: a review. Br J Surg 1984; 71: 921-927.
- 11. Zorcolo L, Covotta L, Carlomagno N, Bartolo DCC. Safety of primary anastomosis in emergency colorectal surgery. Colorectal Dis 2003; 5: 262-269.
- 12. Gooszen AW, Tollenaar AE, Geelkerken RH, et al. Prospective study of primary anastomosis following sigmoid resection for suspected acute complicated diverticular disease. Br J Surg 2001; 88: 693-697.
- 13. Schilling MK, Maurer CA, Kollmar O, Büchler MW. Primary vs. secondary anastomosis after sigmoid colon resection for perforated diverticulitis (Hinchey Stage III and IV): a prospective outcome and cost analysis. Dis Colon Rectum 2001; 44: 699-703.
- 14. Parks TG. Natural history of diverticular disease of the colon: a review of 521 cases. BMJ 1969; 639: 642.
- 15. Salem TA, Molloy RG, O’Dwyer PJ. Prospective study on the management of patients with complicated diverticular disease. Colorectal Dis 2006; 8: 173-176.
- 16. Chapman JR, Dozois EJ, Wolff BG, et al. Diverticulitis: a progressive disease? Do multiple recurrences predict less favourable outcomes? Ann Surg 2006; 243: 876-880.
- 17. Tyau ES, Prystowsky JB, Joehl RJ, Nahrwold DL. Acute diverticulitis: a complicated problem in the immunocompromised patient. Arch Surg 1991; 126: 855-859.
- 18. Chapman J, Davies M, Wolff B, et al. Complicated diverticulitis: is it time to rethink the rules? Ann Surg 2005; 242: 576-581.
- 19. Hart AR, Kennedy JH, Stebbings WS, et al. How frequently do large bowel diverticula perforate? An incidence and cross-sectional study. Eur J Gastroenterol Hepatol 2000; 12: 661-666.
- 20. Wong SK, Ho YH, Leong AP, Seow-Choen F. Clinical behaviour of complicated right-sided and left-sided diverticulosis. Dis Colon Rectum 1997; 40: 344-348.
- 21. Belmont C, Klas JV, Perez J, et al. The Hartmann procedure. Arch Surg 1996; 131: 612-617.
- 22. Chua CL. Surgical considerations in the Hartmann’s procedure. Aust N Z J Surg 1996; 66: 676-679.
- 23. Roe AM, Prabhu S, Ali A, et al. Reversal of Hartmann’s procedure: timing and operative technique. Br J Surg 1991; 78: 1167-1170.
- 24. Salem L, Flum DR. Primary anastomosis or Hartmann’s procedure for patients with diverticular peritonitis? A systematic review. Dis Colon Rectum 2004; 47: 1953-1964.
- 25. Constantinides VA, Tekkis PP, Athanasiou T, et al. Primary resection with anastomosis vs Hartmann procedure in nonelective surgery for acute colonic diverticulitis: a systematic review. Dis Colon Rectum 2006; 49: 966-981.
- 26. Mutter D, Bouras G, Forgione A, et al. Two-stage totally minimally invasive approach for acute complicated diverticulitis. Colorectal Dis 2006; 8: 501-505.
- 27. Faranda C, Barrat C, Catheline JM, Champault G. Two-stage laparoscopic management of generalized peritonitis due to perforated sigmoid diverticula: eighteen cases. Surg Laparosc Endosc Percutan Tech 2000; 10: 135-138.





Abstract
There are controversies surrounding the indications, time and place for elective surgery, and role of multistage operations in the treatment of complicated diverticular disease.
Most patients with uncomplicated diverticulitis can be managed non-operatively.
Previous indications for elective surgery after two attacks of diverticulitis have been questioned. Evidence that patients are less likely to respond to medical therapy in subsequent attacks of diverticulitis is lacking. Decisions should be based on individual circumstances.
The Hartmann procedure remains a safe option for patients with free perforation and generalised peritonitis.
In experienced hands, a one-stage procedure can be as successful as a two-stage procedure in an emergency setting in selected patients.
When possible, an operation should be converted from an emergency to a semi-elective one using techniques such as radiologically or laparoscopically guided drainage of collections.