Clinical record
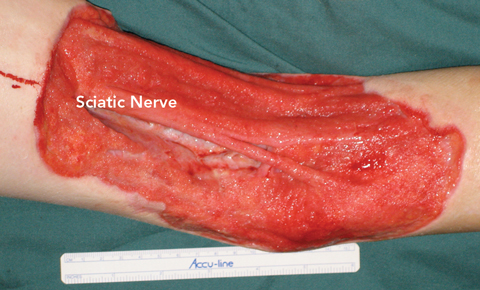
A previously healthy 11-year-old boy presented to the Royal Children’s Hospital (RCH) with a 20 × 12 cm necrotising wound of the left popliteal fossa (Figure A). A further inflammatory lesion was evident on the anteromedial left thigh, extending into the groin. The boy was febrile and cachectic, weighing 30 kg.
When the patient failed to improve after 5 days of intravenous antibiotic treatment, clinical suspicion of an atypical disease was heightened. Multidisciplinary discussions led to a specific request to the microbiology department to attempt to identify fungal disease. Further tissue cultures under specific conditions (see below) showed growth of the fungus Saksenaea vasiformis. Histopathological analysis of resected tissue revealed extensive subcutaneous fat necrosis with calcification and many non-septate hyphae with irregular diameters and branching — features consistent with zygomycosis (mucormycosis).1 These features were not seen on Gram stain.
Antifungal therapy was initiated with amphotericin B 45 mg given via central venous line over 24 hours initially and then intravenous liposomal amphotericin B 150 mg daily. Major predisposing factors to zygomycosis were excluded: specifically, hyperglycaemia, ketoacidosis and immunosuppressive disorders.1 Notable findings included a raised IgE level. Although this can be associated with hyper-IgE syndrome, the patient had no other signs of this disease.
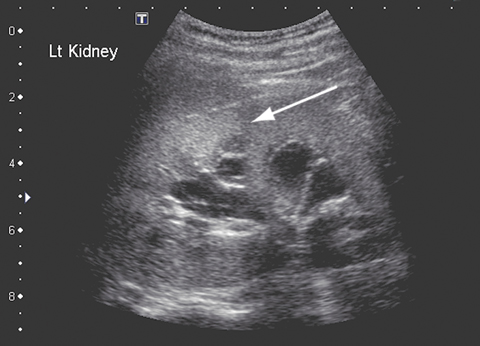
After 5 weeks of treatment with amphotericin B, the patient became unwell, with a high fever and moderate renal impairment. His creatinine level had increased from 0.06 to 0.17 mmol/L (RR, 0.03–0.08 mmol/L); his urea level was 18.7 mmol/L (RR, 2.1–6.5 mmol/L), and the glomerular filtration rate was 29 mL/min (RR, 100–140 mL/min). A renal ultrasound scan revealed parenchymal fungal spores (Figure B). Amphotericin B treatment was ceased and twice daily oral posaconazole 400 mg commenced. Renal function normalised within about 7 days. Follow-up imaging 6 weeks later revealed resolution of renal fungal disease.
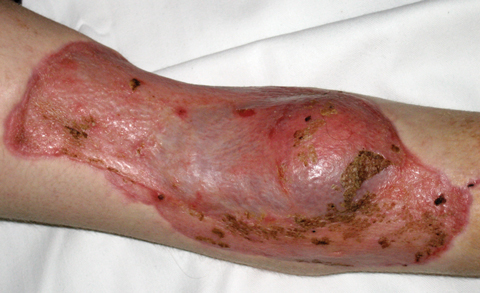
After 6 weeks of antifungal treatment and no growth evident on serial tissue cultures, the wounds were surgically closed. The popliteal fossa wound was treated with a pedicled medial gastrocnemius muscle flap and skin graft to cover the exposed neurovascular structures. The groin wound was closed with a sartorius muscle turnover flap and skin graft. Both the wound and donor sites fully healed without complication (Figure C).
 |
|
A: Necrotising wound of the left popliteal fossa on presentation. |
 |
 |
Since it was first isolated in 1953,2 Saksenaea vasiformis has been reported to cause human infection in about 30 cases. In only two cases has disseminated disease been reported, both occurring in immunocompetent patients and having fatal outcomes.3,4 S. vasiformis belongs to the class Zygomycetes and order Mucorales. The usual manifestation of disease is reported to be skin and soft-tissue infections, most commonly occurring secondary to trauma,1 but can also follow haematogenous dissemination.
Lessons from practice
Multidisciplinary care is fundamental in dealing with complex or unusual conditions.
Always consider atypical pathogens, particularly fungi, when a condition fails to respond to the perceived ideal treatment.
If considering a fungal pathogen, consult microbiological opinion to aid identification of the species.
Posaconazole can be used safely and successfully in children to treat disseminated fungal disease.
A multidisciplinary team approach is essential for a successful outcome. Exclusion of underlying immunodeficiency is paramount, as its presence often leads to a fatal outcome.1
Surgical treatment combined with appropriate antifungal therapy is the mainstay of treatment. In the case we describe, the patient required serial debridements and the use of vacuum dressings until microbiological evidence of eradication of fungi was achieved. Vacuum dressings were a good alternative to more labour-intensive and painful forms of dressing. The dressings were changed in theatre every 3–5 days. Negative pressure dressings have been shown to improve wound healing by increasing tissue oxygenation via a reduction of interstitial fluid5,6 and reducing bacterial counts in the wound.7
Diagnosis and identification of the fungus is important. Zygomycosis must be suspected in any wound that is worsening despite appropriate treatment. Tissue culture, rather than routine wound swabs, provides the most appropriate specimen. It is important to inform the microbiology laboratory of clinical suspicion of zygomycosis. The tissue should be cultured intact to preserve the viability of the irregularly septate zygomycete hyphae. This also ensures that cultures are incubated for the extended period necessary for fungal isolation. S. vasiformis is a fast-growing organism and can usually be isolated from culture within 72 hours.1,8 Once isolated, full identification for fungi may require extended time and specialist media in a reference laboratory.
Posaconazole has been shown to be effective against zygomycosis when other therapy has been ineffective or complications have occurred due to the therapy.9 As posaconazole is not generally used in children under the age of 13 years, its safety profile in children is unknown. The drug is available as an oral suspension and is generally administered at a dose of 800 mg daily in divided doses. The patient described here received 400 mg twice daily without complication. The duration of therapy is dictated by the presence of disease. Minor common side effects include nausea and headache. Serious side effects are rare, but hepatic impairment may occur, so liver function should be monitored every 2–3 weeks during therapy. Posaconazole interacts with several immunosuppressive agents and has few contraindications.
- 1. Holland J. Emerging zygomycoses of humans: Saksenaea vasiformis and Apophysomyces elegans. Curr Top Med Mycol 1997; 8: 27-34.
- 2. Saksena SB. A new genus of the Mucorales. Mycologia 1953; 45: 426-436.
- 3. Torell J, Cooper BH, Helgeson NG. Disseminated Saksenaea vasiformis infection. Am J Clin Pathol 1981; 76: 116-121.
- 4. Hay RJ, Campbell CK, Marshall WM, et al. Disseminated zygomycosis (mucormycosis) caused by Saksenaea vasiformis. J Infect 1983; 7: 162-165.
- 5. Bucalo B, Eaglstein WH, Falanga V. Inhibition of cell proliferation by chronic wound fluid. Wound Repair Regen 1993; 1: 181-186.
- 6. Argenta LC, Morykwas MJ. Vacuum-assisted closure: a new method for wound control and treatment: clinical experience. Ann Plast Surg 1997; 38: 563-576.
- 7. Morykwas MJ, Argenta LC, Shelton-Brown EI, McGuirt W. Vacuum-assisted closure: a new method for wound control and treatment: animal studies and basic foundation. Ann Plast Surg 1997; 38: 553-562.
- 8. Padmaja IJ, Ramani TV, Kalyani S. Cutaneous zygomycosis: necrotising fascitis due to Saksenaea vasiformis. Indian J Med Microbiol 2006; 24: 58-60.
- 9. MIMS online drug information for posaconazole. http://www.mims.com.au (accessed Sep 2008).





None identified.