Rates of smoking during pregnancy in Australia in 2003 were highest among teenage mothers, with 42.1% reporting smoking compared with 17.3% for all mothers.1 The high prevalence of smoking among teenage mothers is consistent with international studies (eg, 46.2% in Liverpool, United Kingdom, and 47.0% in Nova Scotia, Canada),2,3 which also reflect overall higher rates of other perinatal risk behaviours such as substance misuse, poor nutritional intake and low attendance at antenatal classes.4 Despite various initiatives in the past decade and trials reporting efficacy of smoking interventions in pregnant women, rates of smoking in teenagers remain high and affect the health of their unborn babies.5,6
Cigarette smoking during pregnancy is one of the few modifiable risk factors for low birthweight (LBW), preterm birth, placental complications and perinatal mortality.7-9 Maternal smoking status has been shown to be a more important determinant of baby birthweight and growth than maternal height, weight, number of previous pregnancies and their outcomes, or sex of the baby.10 LBW is not only significant as a key indicator of a newborn’s overall health, but also, according to the fetal origins hypothesis,11 affects childhood development and predisposes children to chronic disease in later life. Moreover, intrauterine exposure to smoking has been linked with health problems such as sudden infant death syndrome,9 childhood obesity12 and diabetes.13
Smoking during the third trimester of pregnancy is an important risk factor for LBW, as this is a period of rapid fetal weight gain.14 A randomised controlled trial has shown that women who quit smoking in the first 3–4 months of pregnancy give birth to infants of similar birthweight to infants of women who have never smoked.15 Thus it is important that prenatal smoking cessation programs persist throughout pregnancy and target mothers who continue to smoke.
Young maternal age has also been associated with LBW and poor perinatal outcomes.2,7,16 High rates of smoking among Australian teenagers make it difficult to ascertain whether young maternal age, smoking behaviour or other factors are responsible for the high incidence of LBW in babies of teenage mothers.
A retrospective population-based study was conducted using the National Perinatal Data Collection, a state- and territory-based collection of data on all births of at least 400 g birthweight or at least 20 weeks’ gestation.1 Data on singleton live births occurring between January 2001 and December 2004 were obtained, giving a sample size of 46 938 teenage mothers aged < 20 years and 755 393 mothers aged 20–34 years at the date of delivery. Restricting the sample to births in the five states and territories in which smoking data were available, 55% of teenage mothers (25 828) remained. Of the states and territories with smoking data available, three had information on smoking status and quantity of cigarettes (dose) in the second half of pregnancy (38.9% of the teenage mothers [18 276] resided in those three states). Where information on smoking status was not available (0.55% of all records), data were excluded from the analysis.
A teenage mother was defined as any mother giving birth at an age of less than 20 years. Teenage mothers were compared with mothers aged 20–34 years, who represented the standard cohort of mothers. A smoker was defined as a mother who self-reported smoking at least one cigarette during pregnancy, while a quitter was defined as a mother who reported smoking during pregnancy but not during the second half of pregnancy. LBW was defined as a birthweight of less than 2500 g.1
Data were collected through two questions, which were not standardised nationally. In five states and territories, maternal smoking status (yes/no) was self-reported at the first antenatal visit or at delivery through a question asking whether the mother smoked at all during pregnancy. Three of the states/territories also collected data ascertaining the average number of cigarettes smoked per day in the second half of pregnancy (categorised as none, ≤ 10 per day, > 10 per day, or unknown).1
Data on the geographical location of usual residence were mapped to levels of remoteness using the Australian Bureau of Statistics Australian Standard Geographical Classification remoteness structure.17 The remote and very remote groups were combined into one category.
The Socio-Economic Indexes for Areas (SEIFA) Index of Advantage/Disadvantage18 was used as a measure of the social and economic wellbeing of Australian mothers. Data were stratified into quintiles for analysis, with the lowest quintile representing the most disadvantaged group.
Smoking prevalences in pregnant women, by year and maternal age group, are shown in Box 1. The smoking status of smokers during the second half of pregnancy (in the three states in which data were available) is shown in Box 2.
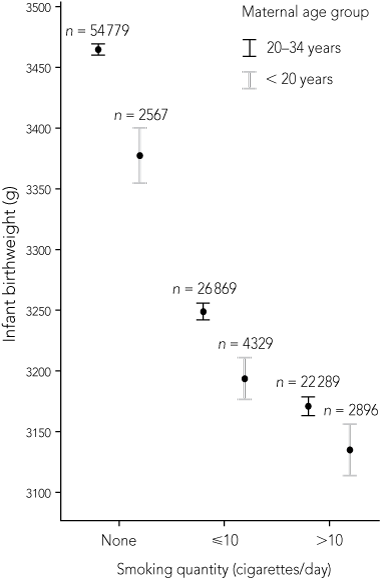
The mean birthweight of babies was also related to smoking quantity, with mothers who smoked more than 10 cigarettes a day having significantly lower birthweight babies. Teenage mothers had poorer birthweight outcomes than older mothers for all categories of smoking quantity (Box 3).
On univariate analysis of mothers aged < 35 years, factors significantly associated with LBW were smoking (OR, 2.53 [95% CI, 2.45–2.60]), Indigenous status (OR, 3.09 [95% CI, 2.96–3.23]), SEIFA category (OR [lowest quintile], 1.76 [95% CI, 1.68–1.84]), remoteness category (OR [most remote category], 2.37 [95% CI, 2.20–2.55]), parity (OR [multiparity, 4–8], 2.22 [95% CI, 2.07–2.37]) and maternal age group (OR, 1.81 [95% CI, 1.73–1.90]). After multiple regression analysis, a significant association remained between LBW and smoking, Indigenous status, SEIFA category and parity, but maternal age group was no longer significant. (Remoteness was not included in the multiple regression model, as it was highly correlated with the SEIFA Index of Advantage/Disadvantage.) Smoking and Indigenous status were the strongest predictors of LBW (Box 4).
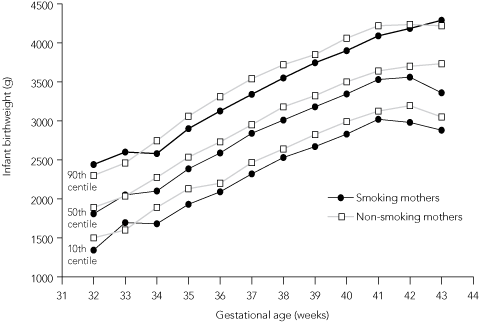
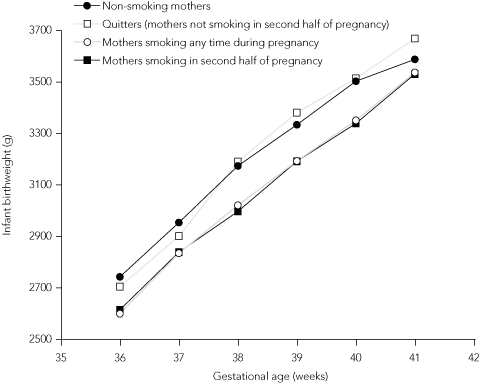
To further investigate the impact of smoking on the birthweight of babies born to teenage mothers, we constructed birthweight-for-gestational-age curves for smokers compared with non-smokers (Box 5). After 33 weeks’ gestation, babies born to teenage smokers had persistently lower average birthweights than babies born to non-smokers. In a further analysis of the same data, the birthweight of babies born to teenage mothers who ceased smoking in the second half of pregnancy were plotted alongside smokers and non-smokers (Box 6). For births at 38–41 weeks’ gestation, teenage mothers who had quit smoking in the second half of pregnancy had babies with mean birthweights that were no different from those of babies born to non-smokers.
Smoking by teenage mothers during pregnancy remains a significant public health problem in Australia, with potential lifelong implications for both mother and baby.11 The significantly higher rates of LBW seen in babies of teenage mothers reflect the high overall level of smoking observed in this age group. As almost half of all teenage mothers and over half of Indigenous teenage mothers are smoking during pregnancy, current smoking reduction strategies appear to be having limited effect. In our study, the quitting rates of one in 15 among teenage mothers and one in 12 among mothers aged 20–34 years indicate how difficult smoking cessation remains for pregnant women. Nevertheless, pregnancy remains a unique opportunity for the health sector to engage this highly vulnerable group in reducing all risk behaviours, including smoking.
Our birthweight-for-gestational-age curve (Box 5) concurs with other studies showing that maternal smoking affects baby birthweight after 33 weeks’ gestation,2,16 although the trend was more marked and consistent in our population. We speculate that the difference can be attributed to the large population size in our study, which minimised information and selection biases.
Reducing the quantity of cigarettes smoked during pregnancy also has an impact on the risk of LBW in infants. A 2005 hospital-based study of 534 teenage mothers giving birth at Liverpool Women’s Hospital, UK, found lower average baby birthweights were associated with increasing quantity of cigarettes smoked.2 Another study showed that reducing cigarette smoking to eight cigarettes a day significantly improved baby birthweight.19 These findings are consistent with our results showing an association between higher cigarette dose and LBW.
Our multivariate analysis showed that maternal smoking and Indigenous status were the strongest predictors of LBW in infants, with maternal age no longer being a significant factor. The clear association observed between smoking and birthweight is consistent with other studies.2,7,8,16
Our study reports baseline data from five states and territories, with the remaining three states not collecting smoking data in the period 2001–2004. In 2005, two of those states began collecting smoking data, but there is still a need for uniform national data. A Smoking in Pregnancy workshop was held in 2007 to initiate a program of data development as part of a federally funded initiative to stop smoking during and after pregnancy.20 The aim of this process is to gain agreement on a method of collecting nationally uniform data to monitor the effects of smoking during pregnancy and to enable accurate monitoring of smoking and quitting rates among women who give birth.
Smoking remains one of the few modifiable risk factors in pregnancy. Teenagers often present later for their first antenatal visit than older mothers, and may not access health services at all.4 Indigenous teenagers are a particularly vulnerable group, with heightened disparities in reproductive health outcomes seen both in Australia and internationally.7,21-23
Access to antenatal care and related health education, including breastfeeding promotion, is often compromised by cultural, linguistic and geographical barriers.24 A recent study investigating factors to consider in smoking interventions for Indigenous mothers emphasised the need for culturally appropriate programs that included subsidised nicotine replacement therapy, support groups and community education about the harmful effects of active and passive smoking. Such programs would ideally be delivered through Aboriginal Medical Services.23
Health care providers, especially general practitioners, are in an ideal position to influence maternal smoking rates. However, studies have demonstrated that doctors only occasionally implement smoking cessation interventions or offer advice on how to quit.25 Our results show that harm minimisation strategies need to target not only smoking cessation but also smoking reduction, particularly in teenage mothers. Primary health care providers should first counsel pregnant women to cease smoking, and, if this is unsuccessful, advise them to continually reduce their cigarette intake.
2 Cessation of smoking in second half of pregnancy, in mothers who reported smoking during pregnancy, 2001–2004*
3 Relationship between infant birthweight and quantity of cigarettes smoked by pregnant women, by maternal age group*
 | |||||||||||||||
4 Factors associated with low birthweight, 2001–2004*
AOR = adjusted odds ratio. OR = odds ratio. SEIFA = Socio-Economic Indexes for Areas.18 |
|||||||||||||||
Received 23 August 2007, accepted 19 November 2007
- Denise L Chan1
- Elizabeth A Sullivan2
- 1 Faculty of Medicine, University of New South Wales, Sydney, NSW.
- 2 Perinatal and Reproductive Epidemiology and Research Unit, School of Women’s and Children’s Health, University of New South Wales, Sydney, NSW.
The Australian Institute of Health and Welfare (AIHW) provides funding for the National Perinatal Data Collection and the AIHW National Perinatal Statistics Unit (NPSU). We acknowledge the work of state- and territory-based midwives and other health workers in putting together state and territory perinatal collections. We are grateful for the assistance provided by Paula Laws, Alex Wang, Narelle Grayson, Jane Walker and other staff at the NPSU and by Associate Professor Deborah Black, School of Public Health and Community Medicine, University of New South Wales, in undertaking this study.
None identified.
- 1. Laws PJ, Grayson N, Sullivan EA. Smoking and pregnancy. Sydney: Australian Institute of Health and Welfare National Perinatal Statistics Unit, 2006. (AIHW Cat. No. PER 33.)
- 2. Delpisheh A, Attia E, Drammond S, Brabin BJ. Adolescent smoking in pregnancy and birth outcomes. Eur J Public Health 2005; 16: 168-172.
- 3. Dodds L. Prevalence of smoking among pregnant women in Nova Scotia from 1988 to 1992. CMAJ 1995; 152: 185-190.
- 4. Klein J, The Committee on Adolescence. Adolescent pregnancy: current trends and issues. Pediatrics 2005; 116: 281-286.
- 5. Lumley J, Oliver SS, Chamberlain C, Oakley L. Interventions for promoting smoking cessation during pregnancy. Cochrane Database Syst Rev 2004; (4): CD001055.
- 6. Panjari M, Bell R, Bishop S, et al. A randomized controlled trial of a smoking cessation intervention during pregnancy. Aust N Z J Obstet Gynaecol 1999; 39: 312-317.
- 7. Chan A, Keane RJ, Robinson JS. The contribution of maternal smoking to preterm birth, small for gestational age and low birthweight among Aboriginal and non-Aboriginal births in South Australia. Med J Aust 2001; 174: 389-393.
- 8. Bai J, Wong F, Gyaneshwar R, Stewart H. Profile of maternal smokers and their pregnancy outcomes in south western Sydney. J Obstet Gynaecol Res 2000; 26: 127-132.
- 9. The health consequences of smoking: a report of the Surgeon General. Atlanta, Ga: US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health, 2004. http://www.cdc.gov/tobacco/data_statistics/sgr/sgr_2004/index.htm (accessed Nov 2007).
- 10. Kramer MS. Determinants of low birth weight: methodological assessment and meta-analysis. Bull World Health Organ 1987; 65: 663-737.
- 11. De Boo HA, Harding JE. The developmental origins of adult disease (Barker) hypothesis. Aust N Z J Obstet Gynaecol 2006; 46: 4-14.
- 12. von Kries R, Toschke A, Koletzko B, Slikker W. Maternal smoking during pregnancy and childhood obesity. Am J Epidemiol 2002; 156: 954-961.
- 13. Montgomery S, Ekbom A. Smoking during pregnancy and diabetes mellitus in a British longitudinal birth cohort. BMJ 2002; 324: 26-27.
- 14. Hill M. Medical consequences of smoking in pregnancy. Sydney: School of Medical Sciences, University of New South Wales, 2005. http://embryology.med.unsw.edu.au/pdf/Smokings1.pdf (accessed Nov 2007).
- 15. Bernstein IM, Mongeon JA, Badger GJ, et al. Maternal smoking and its association with birth weight. Obstet Gynecol 2005; 106: 986-991.
- 16. Dewan N, Brabin B, Wood L, et al. The effects of smoking on birthweight-for-gestational-age curves in teenage and adult primigravidae. Public Health 2003; 117: 31-35.
- 17. Australian Bureau of Statistics. Australian Standard Geographical Classification (ASGC). Canberra: ABS, 2001. http://www.abs.gov.au/Ausstats/abs@.nsf/956c382b0b05ba7d4a 2568010004e173/a3658d8f0ad7a9b6ca 256ad4007f1c42!OpenDocument (accessed Nov 2007).
- 18. Australian Bureau of Statistics. Census of population and housing — SEIFA 2001 product information. Canberra: ABS, 2001. http://www.abs. gov.au/Websitedbs/D3110124.NSFf5c7b8fb 229cf017ca256973001fecec/d8f7e0b7b36c 2023ca256de30081bea2!OpenDocument (accessed Feb 2008).
- 19. England LJ, Kendrick JS, Wilson HG, et al. Effects of smoking reduction during pregnancy on the birth weight of term infants. Am J Epidemiol 2001; 154: 694-701.
- 20. Pyne C. National smoking and pregnancy advisory group [media release]. Canberra: Australian Government Department of Health and Ageing, 2005; 12 Sep. http://www.health.gov. au/internet/ministers/publishing.nsf/content/health-mediarel-yr2005-cp-pyn053.htm/$FILE/pyn053.pdf (accessed Nov 2007).
- 21. Gilbert WM, Jandial D, Field NT, et al. Birth outcomes in teenage pregnancies. J Matern Fetal Neonatal Med 2004; 16: 265-270.
- 22. Anachebe NF, Sutton MY. Racial disparities in reproductive health outcomes. Am J Obstet Gynecol 2003; 188: S37-S42.
- 23. Heath DL, Panaretto KS, Manessis V, et al. Factors to consider in smoking interventions for Indigenous women. Aust J Prim Health 2006; 12: 131-136.
- 24. Gilchrist D, Woods B, Binns CW, et al. Aboriginal mothers, breastfeeding and smoking. Aust N Z J Public Health 2004; 28: 225-228.
- 25. Cooke M, Mattick RP, Walsh RA. Differential uptake of a smoking cessation programme disseminated to doctors and midwives in antenatal clinics. Addiction 2001; 96: 495-505.





Abstract
Objective: To determine the association between smoking in pregnant teenagers and baby birthweight.
Design, setting and participants: A retrospective population-based study of women aged < 20 years who gave birth to liveborn singletons in Australia between January 2001 and December 2004. Data were drawn from the National Perinatal Data Collection.
Main outcome measures: Maternal smoking, birthweight, low birthweight (LBW).
Results: The prevalence of LBW in babies born to teenage smokers was 9.9%, compared with 6.0% in babies born to teenage non-smokers (odds ratio [OR], 1.72 [95% CI, 1.57–1.90]). On average, babies born to teenage smokers were 179.8 g lower in birthweight than babies born to teenage non-smokers (95% CI, 165.5 –194.1 g; t = 24.6, P < 0.001). Smoking, Indigenous status, Socio-Economic Indexes for Areas category and parity were independently associated with LBW (all ORs > 1.3; P < 0.001) after adjusting for maternal age group. Teenagers smoking > 10 cigarettes a day had babies with lower birthweight that those who smoked ≤ 10 cigarettes a day, demonstrating a dose–response relationship. The babies of teenage smokers who stopped smoking before 20 weeks’ gestation had birthweights similar to those of babies born to teenage non-smokers. One in 15 teenage smokers stopped smoking during pregnancy.
Conclusion: Babies whose mothers smoked during pregnancy were more likely to have LBW than babies whose mothers did not smoke. Mothers who continue to smoke in the second half of pregnancy increase their baby’s risk of LBW. There is significant scope to improve the quitting rate, and health professionals need to target smoking cessation at all contacts with pregnant women who continue to smoke.