Clinical practice guidelines for the management of acute coronary syndromes, including myocardial infarction, have been published by the National Heart Foundation (NHF) of Australia and the Cardiac Society of Australia and New Zealand (CSANZ).1 These recommendations expand on previous guidelines,2 incorporate a systematic review of available evidence, and aim to assist health professionals with the best practice management of cardiac patients.
Adherence to guidelines-based care is associated with improved patient outcomes.3-6 However, overseas audits suggest only a proportion of patients are being treated according to best practice.4-7 In Australia, there is limited information on the measurement and publication of quality indicators.
The Heart Protection Partnership (HPP) project was created to audit adherence to evidence-based guidelines in acute care facilities across Australia. Its purpose was to provide a “snapshot” of the quality of care, as assessed by adherence to the NHF/CSANZ guidelines.1 The program then aimed to provide feedback to health care providers across Australia about the level of care rendered to real-world patients, through evaluation of actual performance versus optimal care standards. Through identification of treatment gaps and baseline indicator feedback, the intention was that individual centres could then implement locally adapted interventions for improving compliance.
The HPP Steering Committee (a multistate, multidisciplinary panel incorporating cardiologists, interventional cardiologists, general physicians and representatives of the NHF) developed audit criteria based on NHF/CSANZ guidelines and definitions.1 Once a hospital had agreed to enrol patients in the audit, a Care Coordinator (research assistant) was assigned to facilitate the audit and follow-up. Box 1 lists the participating centres and principal investigators. At each centre, up to 100 consecutive patients admitted with chest pain to a monitored bed were asked to participate; their written consent was obtained before their enrolment. Patients were free to withdraw at any time.
Primary outcome measures were the proportion of patients whose care met quality of care standards for diagnostic and risk-stratification procedures and management according to the NHF/CSANZ guidelines.1 Compliance with guidelines was assessed by the Care Coordinator according to whether an action, such as medication prescribing, was indicated, and adjusted for stated contraindication. This was determined in conjunction with the principal investigator at the centre, with clarification from the treating physician as required.
Between January 2003 and August 2005, 2380 patients were recruited from 27 hospitals across five states in Australia. Thirteen hospitals had both angiographic and PCI facilities at the time of the audit. Patient data are summarised in Box 2. Interventional hospitals had more men (69% v 65%; P = 0.057), fewer Indigenous patients (4% v 13%; P < 0.001), more smokers (28% v 23%; P = 0.058), and more patients with hyperlipidaemia (50% v 41%, P < 0.001) or known ischaemic heart disease (25% v 17%; P < 0.001). A greater proportion of patients at interventional centres had myocardial infarction as the primary discharge diagnosis (52% v 38%); atypical chest pain was a more common finding at non-interventional centres (12% v 8%; P < 0.006). The total in-hospital major adverse cardiovascular event rate was 3.9%, with no significant difference between interventional (4.4%) and non-interventional centres (3.2%; P = 0.12).
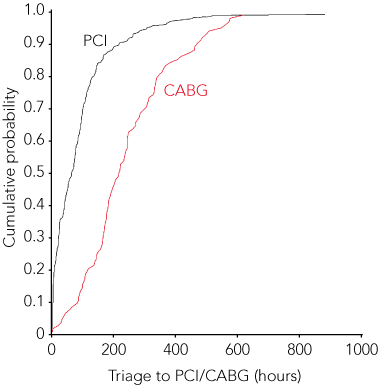
At interventional centres, the revascularisation rates were 24.4% (95% CI, 22.09%–26.92%) for PCI and coronary artery bypass graft surgery (CABG). In the group under-going angiography, 31% was for single vessel disease, 52% for multivessel disease and 16% for no significant coronary disease. The median time from admission to PCI was 63.1 h, and the median time from admission to CABG was 9.1 days (P < 0.001). Box 3 shows cumulative probability curves by time from triage to PCI and CABG.
Box 4 shows results of a logistic regression model for referral for angiography.
Box 5 shows use of medications adjusted for stated contraindication, for all centres during hospital admission, including emergency department, coronary care unit and ward.
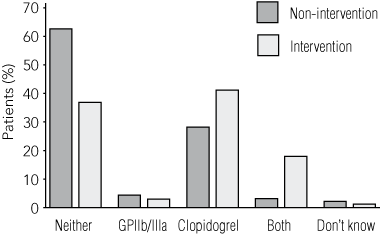
The use of clopidogrel, a glycoprotein IIb/IIIa (GPIIb/IIIa) inhibitor, or both across all centres is shown in Box 6.
Many national projects, such as those in the United States3-5,7-11 and Europe6,12,13 have emphasised the importance of systematically measuring performance and outcomes to improve total quality of care. Our study was the first of its type in Australia to prospectively audit the care of consecutive patients presenting with undifferentiated chest pain to monitored beds across the nation. It was conducted during 2003–2005, allowing a reasonable amount of time for dissemination and uptake of the NHF/CSANZ guidelines published in 2000.2 The audit was timed to occur just before the update of the guidelines in 2006.
Our study showed wide variations in adherence to evidence-based guidelines in Australian acute care facilities for patients presenting with undifferentiated chest pain, about 71% of whom had a discharge diagnosis of an acute coronary syndrome. Substantial gaps in use of guidelines-based treatment paths and medications were evident at all centres. A similar audit of acute coronary syndrome patients,14 conducted after ours, reaffirms our finding. However, we more particularly found adherence was significantly lower in non-interventional centres than in interventional centres.
In our study, prescribing of medical therapy according to recommendations varied significantly. The rates of medication prescribing are similar to those reported in other Australian-based studies, as well as international audits such as GRACE. For example, the rate of aspirin prescribing in our study was 91%, compared with 90% in a Queensland study,15 92.9% in a similar audit,14 and 93% in the GRACE study.16 Similarly, the respective rates for ACE inhibitor prescribing were 58%, 56%, 48.5% and 73%. Notably, the largest discrepancies in medical therapy, both between settings and in terms of deviation from the guidelines, arose in the use of acute treatments, such as GPIIb/IIIa antagonists and early use of clopidogrel. The use of these agents was low, particularly in non-interventional centres. A study of the early use of GPIIb/IIIa antagonists in the US8 found a similarly low rate of 25%, and other audits conducted in Australia found a rate of 5%.15 It is not apparent why use of these agents is so low — possible explanations include access to these treatments, training and education in their use, and cost. This is an area where further investigation is suggested.
A previous study found little overall difference in quality of care, with regard to use of medical therapies, between hospital types in Queensland.15 However, that study compared tertiary versus non-tertiary hospitals, and did not include some of the largest cardiac centres in Queensland. A further study did find a link between the quality of care and funding initiatives directed towards the implementation of “multiple systematic interventions”.17 In another study, variability in care of patients with acute coronary syndrome depended on whether they experienced STEMI, non-STEMI or unstable angina.14 The CRUSADE initiative in the US demonstrated marked variation in the use of recommended medical therapies between leading (most adherent) and lagging (least adherent) hospitals.5 This variation was most evident with therapies considered recent innovations or more aggressive. If our results are compared with results from these leading and lagging centres, based on acute medication use, it appears that Australian practice varies widely between that of leading and lagging centres in the US, depending on the treatment. For example, overall use of GPIIb/IIIa inhibitors in Australian centres is lower than in the most lagging US hospitals, whereas use of any heparin was similar to the most leading US hospitals.9-11
Referral rates for angiography at centres with PCI capability were similar to rates described in GRACE and other registries.18
However, we found a significantly reduced rate of referral for investigation and further evaluation at non-interventional centres than at interventional centres. These findings have been noted in previous audits in Queensland and rural New South Wales. The Queensland study found lower rates of referral for coronary angiography for patients with acute coronary syndromes admitted to non-tertiary centres without interventional facilities (55% v 85%).15 A study in NSW found patients admitted to metropolitan hospitals were more likely to be referred for angiography than patients managed in non-metropolitan hospitals.19
In New Zealand, one study showed a significantly reduced rate of referral for investigation and further evaluation at community hospitals compared with tertiary hospitals with interventional facilities.20 Another New Zealand study showed reduced rates of adherence to medical therapy, referral for angiography and revascularisation in centres without cardiologists.21 The New Zealand Audit Group concluded that patients admitted to hospitals without interventional facilities in general received fewer investigations and less revascularisation than patients admitted to interventional centres.22
The difference in referral rates we observed cannot be attributed to the difference in patient demographics alone. Logistic regression analysis showed that the odds of being referred for angiography are 7.4 times higher at an interventional centre than at a non-interventional centre when adjusted for age, sex, diagnosis and presence of risk factors. We also found men were more likely than women to be referred for angiography. Patients with unstable syndromes were more likely to be referred for angiography than those with simple angina. Patients with hyperlipidaemia were also more likely to be referred for angiography. There was a lower likelihood to refer patients with renal impairment for angiography. With increasing age, the odds of being referred for angiography also decreased. These factors are known to bias physicians in referring patients for angiography.23-25
1 Participating sites and investigators
Coffs Harbour Hospital: J Waites
Lismore Base Hospital: M Tscalis
Nepean Hospital: D Fitzpatrick
Port Macquarie Base Hospital: K Alford
Cairns Base Hospital: P Boyd, C Lim
Gladstone Hospital: S Anandaraja
Mackay Base Hospital: S De Silva, B Weich
Prince Charles Hospital: D Walters
Rockhampton Hospital: M Schoeman
Royal Brisbane Hospital: J Atherton
Townsville General Hospital: S David
Townsville Mater Hospital: W Thoreau
Royal Hobart Hospital: P Roberts-Thompson
Monash Medical Centre: J Boxall
Northern Hospital: D Eccleston
Royal Melbourne Hospital: D Eccleston
4 Logistic regression model for referral for angiography
Interventional centre (reference = non-interventional centre) |
|||||||||||||||
Received 6 December 2006, accepted 26 November 2007
- Darren L Walters1,2
- Constantine N Aroney2,3
- Derek P Chew4,5
- Linden Bungey6
- Steven G Coverdale7
- Roger Allan8
- David Brieger9
- 1 Department of Cardiology, Prince Charles Hospital, Brisbane, QLD.
- 2 University of Queensland, Brisbane, QLD.
- 3 Holy Spirit Northside Hospital, Brisbane, QLD.
- 4 Flinders University, Adelaide, SA.
- 5 Flinders Medical Centre, Adelaide, SA.
- 6 Merck Sharp & Dohme Australia, Brisbane, QLD.
- 7 Nambour General Hospital, Nambour, QLD.
- 8 South East Health, Prince of Wales Hospital, Sydney, NSW.
- 9 Department of Cardiology, Concord Repatriation General Hospital, Sydney, NSW.
The authors would like to thank Dr Annie Solterbeck for her assistance with the statistical analysis of the data.
This study was funded by an unrestricted educational grant from Merck Sharp & Dohme Australia (MSD). Some of the authors served as consultants on an advisory board for MSD. MSD was not directly involved in the preparation of the manuscript, and approval was not sought from MSD for any of the material presented.
- 1. Aroney CN, Aylward P, Kelly A, et al on behalf of the Acute Coronary Syndrome Guidelines Working Group. Guidelines for the management of acute coronary syndromes 2006. Med J Aust 2006; 184 (8 Suppl): S1-S30. <MJA full text>
- 2. Aroney C, Boyden AN, Jellinek MV, et al on behalf of the Unstable Angina Working Group. Management of unstable angina guidelines — 2000. Med J Aust 2000; 173 (8 Suppl): S65-S88.
- 3. Gulati M, Patel S, Jaffe AS, et al Impact of contemporary guideline compliance on risk stratification models for acute coronary syndromes in the registry of acute coronary syndromes. Am J Cardiol 2004; 94: 873-878.
- 4. Eagle KA, Montoye CK, Riba AL, et al. Guideline-based standardized care is associated with substantially lower mortality in Medicare patients with acute myocardial infarction: the American College of Cardiology’s Guidelines Applied in Practice (GAP) Projects in Michigan. J Am Coll Cardiol 2005; 46: 1242-1248.
- 5. Peterson ED, Roe MT, Mulgund J, et al. Association between hospital process performance and outcomes among patients with acute coronary syndromes. JAMA 2006; 295: 1912-1920.
- 6. Vikman S, Airaksinen KEJ, Tierala I, et al. Improved adherence to practice guidelines yields better outcome in high-risk patients with acute coronary syndrome without ST-elevation: findings from nationwide FINACS studies. J Intern Med 2004; 256: 316-323.
- 7. Roe MT, Parsons LS, Pollack CV, et al. Quality of care by classification of myocardial infarction: treatment patterns for ST-segment elevation vs non-ST-segment elevation myocardial infarction. Arch Intern Med 2005; 165: 1630-1636.
- 8. Peterson ED, Pollack CV Jr, Roe MT, et al for the National Registry of Myocardial Infarction (NRMI) 4 investigators. Early use of glycoprotein IIb/IIIa inhibitors in non-ST-elevation acute myocardial infarction: observations from the National Registry of Myocardial Infarction 4. J Am Coll Cardiol 2003; 42: 45-53.
- 9. Bhatt DL, Roe MT, Peterson ED, et al on behalf of the CRUSADE Investigators. Utilization of early invasive management strategies for high-risk patients with non-ST-segment elevation acute coronary syndromes: results from the CRUSADE Quality Improvement Initiative. JAMA 2004; 292: 2096-2104.
- 10. Ohman EM, Roe MT, Smith SC, et al for the CRUSADE investigators. Care of non-ST-segment elevation patients: insights from the CRUSADE national quality improvement initiative. Am Heart J 2004; 148 (5 Suppl 1): S34-S39.
- 11. Ryan JW, Peterson ED, Chen AY, et al on behalf of the CRUSADE investigators. Optimal timing of intervention in non-ST-segment elevation acute coronary syndromes: insights from the CRUSADE (Can Rapid risk stratification of Unstable angina patients Suppress ADverse outcomes with Early implementation of the ACC/AHA guidelines) Registry. Circulation 2005; 112: 3049-3057.
- 12. Dorsch MF, Lawrance RA, Sapsford RJ, et al for the EMMACE (Evaluation of Methods and Management of Acute Coronary Events) Study Group. A simple benchmark for evaluating quality of care of patients following acute myocardial infarction. Heart 2001; 86: 150-154.
- 13. Schiele F, Meneveau N, France Seronde M, et al on behalf of the Réseau de Cardiologie de Franche Comté group. Compliance with guidelines and 1-year mortality in patients with acute myocardial infarction: a prospective study. Eur Heart J 2005; 26: 873-880.
- 14. Chew DP, Amerena J, Coverdale S, et al. Current management of acute coronary syndromes in Australia: observations from the acute coronary syndromes prospective audit. Intern Med J 2007; 37: 741-748.
- 15. Scott IA, Duke AB, Darwin IC, et al for the CHI Cardiac Collaborative. Variations in indicated care of patients with acute coronary syndromes in Queensland hospitals. Med J Aust 2005; 182: 325-330. <MJA full text>
- 16. Granger CB, Steg PG, Peterson E, et al; GRACE Investigators. Medication performance measures and mortality following acute coronary syndromes. Am J Med 2005; 118: 858-865.
- 17. Scott IA, Darwin IC, Harvey KH, et al. Multisite, quality-improvement collaboration to optimise cardiac care in Queensland public hospitals. Med J Aust 2004; 180: 392-397. <MJA full text>
- 18. Fox KA, Anderson FA, Dabbous OH, et al. Intervention in acute coronary syndromes: do patients undergo intervention on the basis of their risk characteristics? The Global Registry of Acute Coronary Events (GRACE). Heart 2007; 93: 177-182
- 19. Heller RF, O’Connell RL, D’Este C, et al. Differences in cardiac procedures among patients in metropolitan and non-metropolitan hospitals in New South Wales after acute myocardial infarction and angina. Aust J Rural Health 2000; 8: 310-317.
- 20. Tang EW, Wong C-K, Herbison P. Community hospital versus tertiary hospital comparison in the treatment and outcome of patients with acute coronary syndrome: a New Zealand experience. N Z Med J 2006; 119: U2078.
- 21. Conaglen P, Sebastian C, Jayaraman C, et al. Management of unstable angina and non-ST-elevation myocardial infarction: do cardiologists do it better? A comparison of secondary and tertiary centre management in New Zealand. N Z Med J 2004; 117: U890.
- 22. Ellis C, Devlin G, Matsis P, et al; New Zealand Acute Coronary Syndromes [NZACS] Audit Group. Acute coronary syndrome patients in New Zealand receive less invasive management when admitted to hospitals without invasive facilities. N Z Med J 2004; 117: U954.
- 23. Daly C, Clemens F, Lopez Sendon JL, et al; Euro Heart Survey Investigators. Gender differences in the management and clinical outcome of stable angina. Circulation 2006; 113: 490-498.
- 24. Shaw LJ, Miller DD, Romeis JC, et al. Prognostic value of noninvasive risk stratification in younger and older patients referred for evaluation of suspected coronary artery disease. J Am Geriatr Soc 1996; 44: 1190-1197.
- 25. Charytan DM, Setoguchi S, Solomon DH, et al. Clinical presentation of myocardial infarction contributes to lower use of coronary angiography in patients with chronic kidney disease. Kidney Int 2007; 71: 938-945.





Abstract
Objective: To evaluate the use of clinical practice guidelines for the management of acute coronary syndromes published by the National Heart Foundation (NHF) of Australia and the Cardiac Society of Australia and New Zealand (CSANZ) in patients presenting with chest pain.
Design: Cross-sectional study of consecutive patients admitted with chest pain.
Setting: Prospective case note review was undertaken in 2380 patients admitted to 27 hospitals across five states in Australia between January 2003 and August 2005. Patients were divided into two groups: those who presented to centres with angiography and percutaneous intervention facilities (n = 1260) and those treated at centres without these facilities (n = 1120).
Main outcome measures: The proportion of patients whose care met quality of care standards for diagnostic and risk-stratification procedures and management according to NHF/CSANZ treatment guidelines.
Results: Significant delays were identified in performing electrocardiography, administering thrombolysis, transferring high-risk patients to tertiary centres, and performing revascularisation. Medical therapy was underused, especially glycoprotein IIb/IIIa antagonists in patients with high-risk acute coronary syndromes. Patients treated at centres without interventional facilities were less likely to receive guidelines-based medical therapy and referral for coronary angiography (20.11%) than patients treated at centres with interventional facilities (66.43%; P < 0.001).
Conclusion: There are deficits in the implementation and adherence to evidence-based guidelines for managing chest pain in hospitals across Australia, and significant differences between hospitals with and without interventional facilities.