Escherichia coli is one of the commonest causes of bacteraemia around the world, accounting for 15%–40% of all significant bacteraemia isolates,1-10 with the majority of infections having their onset in the community.3-5 Before the antibiotic era, E. coli was an infrequent bloodstream isolate. Very few cases were reported up until 1929,11 and reports from Boston in 1935 showed that bacteraemia was much more commonly due to organisms other than E. coli (0.7 episodes per 1000 hospital admissions). After the introduction of antibiotics there was a marked increase in the number of cases of E. coli bacteraemia.12-14 Since the 1980s, the reported incidence of E. coli bacteraemia has ranged from 1.8 to 5.5 episodes per 1000 hospital admissions.3,6-8,15 Rising antibiotic resistance now threatens to make treatment of these infections increasingly difficult.
Canberra is also the tertiary referral centre for a large rural region (58 700 km2) in the south-east of NSW known as the Australian Capital Region (ACR) (http://www.business.nsw.gov.au/region/profiles/capital+Region.htm). In 2002, the total population of this region, including Canberra, was just over 529 000.
Place of acquisition. Place of acquisition was classified as either community-associated or health care-associated (inpatient or non-inpatient), according to national definitions.16 Health care-associated infections were those acquired during hospitalisation or as a direct complication of an indwelling device, medical procedure or instrumentation, or associated with drug-induced neutropenia. Inpatient health care-associated infections were episodes that occurred more than 48 hours after hospital admission and within 48 hours of discharge. All other health care-associated infections were classified as non-inpatient health care-associated infections. All other episodes were classified as community-associated.
Focus of infection. Focus of infection was classified according to the body system affected and/or primary site of sepsis, as adapted from national definitions.16 The focus was classified as “haematological” if associated with neutropenia without evidence of an alternative focus. Bacteraemia in the neonate, acquired from the mother during delivery, was classified as a “maternal” focus.
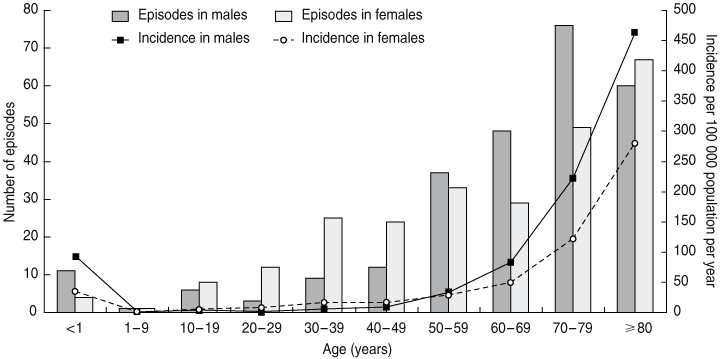
Over the 5-year study period, 585 episodes of E. coli bacteraemia occurred in Canberra (Box 1), of which 76 episodes were excluded from our analysis as they were in non-Canberra residents (most of whom lived in the surrounding ACR). Six additional episodes of E. coli bacteraemia occurred in Canberra residents while attending hospitals within the ACR. Thus, 515 episodes of E. coli bacteraemia were identified in Canberra residents (mean age, 63 years; median age, 68 years). Population incidences, by age and sex, are shown in Box 2. The overall incidence was 28 episodes per 100 000 population per year, with the highest incidence being 463 per 100 000 in men aged 80 years and over.
Sixty-eight per cent of episodes were community-associated, 19% were inpatient health care-associated and 13% were non-inpatient health care-associated. Females were more likely to have community-associated bacteraemia, while males had higher rates of health care-associated bacteraemia (Box 3).
Genitourinary and gastrointestinal tract foci accounted for three-quarters of the episodes (Box 4) and were mostly community-associated. The majority of patients with genitourinary infections were female (60%), while gastrointestinal infections predominated in males (62%).
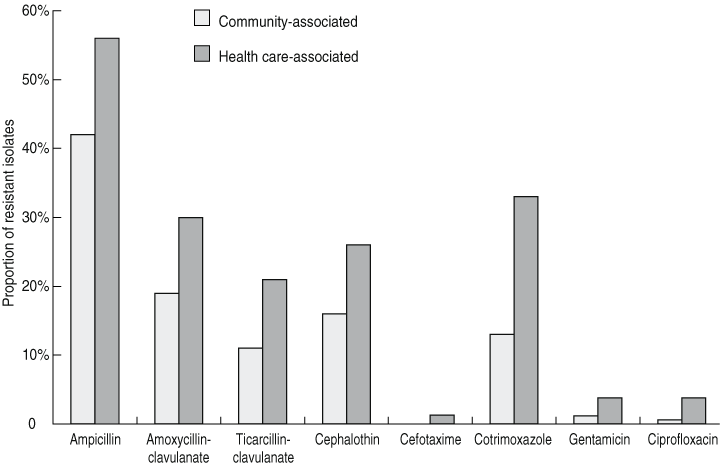
Antibiotic resistance was most common to ampicillin (47%), amoxycillin-clavulanate (23%), cotrimoxazole (20%), cephalothin (19%) and ticarcillin-clavulanate (14%). In contrast, few isolates were resistant to gentamicin (2.1%), ciprofloxacin (1.8%) and cefotaxime (0.4%). Resistance was significantly higher in health care-associated isolates than in community-associated isolates (Box 5).
Few studies have examined the population incidence of E. coli bacteraemia. In a Copenhagen study,15 the highest incidence was 464 episodes per 100 000 population per year in men aged 80–90 years. The percentage of health care-associated episodes in that study (55%) was much higher than the percentage in our study (32%), suggesting that not all community-associated episodes were captured in the Danish study.
In a recent, large, retrospective cohort study of people aged ≥ 65 years in the state of Washington, USA, the rate of community-onset E. coli bacteraemia was 150 per 100 000 person-years.17 In men aged over 85 years, the rate was 462 per 100 000 person-years. As the study excluded health care-associated infections, the overall true incidence of E. coli bacteraemia may have been 30%–50% higher.
In a rural hospital in Kenya,18 the incidence of E. coli bacteraemia in children under 1 year old was high, at 204 per 100 000 children per year, but considered to be an underestimate because of the relatively high number of children who died before hospitalisation.
Since 2001, the European Antimicrobial Resistance Surveillance System has reported data on episodes of invasive E. coli isolates (from the bloodstream and cerebrospinal fluid).1 However, population and hospital coverage within countries is variable, with resultant large differences in calculated incidence rates between countries. In countries that appear to have good population coverage, we calculate that the incidences of invasive E. coli infections per 100 000 population in 2004 were as follows: Austria, 32; Ireland, 32; Finland, 39; Sweden, 39; and Iceland, 40.
The incidence in our study was the same as that in the Copenhagen study (28 episodes per 100 000 population per year).15 Our results are likely to slightly underestimate the incidence, as we were unable to capture Canberra residents who became bacteraemic outside Canberra. Over the study period, an average of 4480 Canberra residents per year were admitted to Australian hospitals outside Canberra.19-23 Based on our incidence of 1.4 episodes per 1000 admissions, an additional 32 episodes of E. coli bacteraemia may have occurred in Canberra residents treated elsewhere over the 5-year study period, bringing the total number of episodes to 547 and the population incidence to 30 episodes per 100 000 population per year. However, “leakage” of cases or under-diagnosis was more likely to have occurred in other studies with less well defined or inclusive populations1,15,17,18 than in our study, which involved an entire geographically isolated region.
Earlier studies of E. coli bacteraemia showed a predominance of hospital-associated infections (55%–72%).6,11,14 But most recent studies have shown that community-associated infections are more common, with hospital-associated infections now accounting for only 14%–30% of episodes.3-5 In our study, 40% of infections in males and 24% of infections in females were health care-associated and therefore potentially preventable. A third of these arose from the genitourinary tract, predominantly resulting from prostate biopsies or urinary catheterisation. Changes in practice relating to prostate biopsies, prompt definitive prostate surgery for obstruction in older men, and timely removal of urinary catheters could help reduce such episodes.
Before the advent of antibiotics, the case-fatality rate among people with E. coli bacteraemia ranged from 15% to 40%.11,12 Despite the introduction of antibiotics, the case-fatality rate from the 1950s to the early 1990s showed no significant improvement.4,6-8,13-15 More recently, reported 7-day case-fatality rates have ranged from 5.9% to 9% — however, rates continue to rise to 16% at 1 month.9,10,17 Our 7-day case-fatality rate was 5%. Extended follow-up (from September 2000 to August 2001) of a subset of 46 patients aged over 16 years showed that case-fatality rates increased to 18% at 1 month and 31% at 6 months.
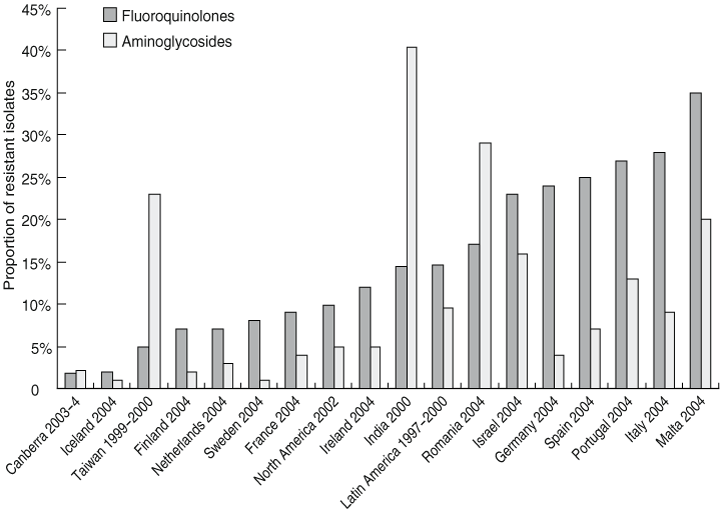
The relatively low antibiotic resistance rates in E. coli blood isolates examined in our study were similar to rates reported in other recent Australian data.24 Resistance rates were significantly higher in health care-associated infections, presumably reflecting more frequent exposure of patients to antibiotics in health care environments. Resistance rates, particularly to aminoglycosides and fluoroquinolones, were much lower in our study than rates reported in many other countries (Box 6).
1 Total episodes of Escherichia coli bacteraemia occurring in Canberra, by patients’ residential status, 2000–2004
ACR = Australian Capital Region. ACT = Australian Capital Territory. NSW = New South Wales. |
|||||||||||||||
Received 7 May 2007, accepted 10 September 2007
- Karina J Kennedy1
- Jan L Roberts2
- Peter J Collignon3
- Microbiology and Infectious Diseases, The Canberra Hospital, Canberra, ACT.
We would like to thank Angelique Clyde-Smith of ACT Pathology; Reimke Kampen, Dianne Dreimanis, Wendy Beckingham, Greg Morley and Philippa Keating from the Bloodstream Infection Surveillance Project; and Paul Whiting and Aline Nguyen from Capital Pathology for assistance in data collection. We also thank Anne Gardner for contributing the 1- and 6-month mortality data.
None identified.
- 1. European Antimicrobial Resistance Surveillance System. Annual report 2004. Bilthoven, The Netherlands: EARSS, 2005. http://www.rivm.nl/earss/result/Monitoring_reports/Annual_reports.jsp (accessed Aug 2006).
- 2. Sader HS, Jones RN, Andrade-Baiocchi S, Biedenbach DJ; SENTRY Participants Group (Latin America). Four-year evaluation of frequency of occurrence and antimicrobial susceptibility patterns of bacteria from bloodstream infections in Latin American medical centers. Diagn Microbiol Infect Dis 2002; 44: 273-280.
- 3. McGregor AR, Collignon PJ. Bacteraemia and fungaemia in an Australian general hospital —associations and outcomes. Med J Aust 1993; 158: 671-674.
- 4. Gosbell IB, Newton PJ, Sullivan EA. Survey of blood cultures from five community hospitals in south-western Sydney, Australia, 1993–1994. Aust N Z J Med 1999; 29: 684-692.
- 5. Javaloyas MD, Garcia-Somoza D, Gudiol F. Epidemiology and prognosis of bacteremia: a 10-y study in a community hospital. Scand J Infect Dis 2002; 34: 436-441.
- 6. Gransden WR, Eykyn SJ, Phillips I, Rowe B. Bacteremia due to Escherichia coli: a study of 861 episodes. Rev Infect Dis 1990; 12: 1008-1018.
- 7. Geerdes HF, Ziegler D, Lode H, et al. Septicemia in 980 patients at a university hospital in Berlin: prospective studies during 4 selected years between 1979 and 1989. Clin Infect Dis 1992; 15: 991-1002.
- 8. Vazquez F, Mendoza MC, Viejo G, Mendez FJ. Survey of Escherichia coli septicemia over a six-year period. Eur J Clin Microbiol Infect Dis 1992; 11: 110-117.
- 9. Lark RL, Saint S, Chenoweth C, et al. Four-year prospective evaluation of community-acquired bacteremia: epidemiology, microbiology, and patient outcome. Diagn Microbiol Infect Dis 2001; 41: 15-22.
- 10. Pedersen G, Schønheyder HC, Sørensen HT. Source of infection and other factors associated with case fatality in community-acquired bacteremia — a Danish population-based cohort study from 1992 to 1997. Clin Microbiol Infect 2003; 9: 793-802.
- 11. Felty AR, Keefer CS. Bacillus coli sepsis. A clinical study of twenty-eight cases of blood stream infection by the colon bacillus. JAMA 1924; 82: 1430-1433.
- 12. Finland M, Jones WF, Barnes MW. Occurrence of serious bacterial infections since introduction of antibacterial agents. JAMA 1959; 170: 2188-2197.
- 13. McCabe WR, Jackson GG. Gram-negative bacteremia. I. Etiology and ecology. Arch Intern Med 1962; 110: 847-855.
- 14. DuPont HL, Spink WW. Infections due to gram-negative organisms: an analysis of 860 patients with bacteremia at the University of Minnesota Medical Center, 1958–1966. Medicine 1969; 48: 307-330.
- 15. Olesen B, Kolmos HJ, Orskov F, et al. Bacteraemia due to Escherichia coli in a Danish university hospital, 1986–1990. Scand J Infect Dis 1995; 27: 253-257.
- 16. Australian Commission on Safety and Quality in Health Care. Blood stream infection (BSI) definition. http://www.safetyandquality.gov.au/internet/safety/publishing.nsf/Content/former-pubs-archive-hcai-definitions (accessed Sep 2006).
- 17. Jackson LA, Benson P, Neuzil KM, et al. Burden of community-onset Escherichia coli bacteremia in seniors. J Infect Dis 2005; 191: 1523-1529.
- 18. Berkley JA, Lowe BS, Mwangi I, et al. Bacteremia among children admitted to a rural hospital in Kenya. N Engl J Med 2005; 352: 39-47.
- 19. Australian Institute of Health and Welfare. Australian hospital statistics 2000–01. Canberra: AIHW, 2002. (AIHW Cat. No. HSE 20; Health Services Series No. 19.) http://www.aihw. gov.au/publications/index.cfm/series/41/start Row/11 (accessed Jan 2008).
- 20. Australian Institute of Health and Welfare. Australian hospital statistics 2001–02. Canberra: AIHW, 2003. (AIHW Cat. No. HSE 25; Health Services Series No. 20.) http://www.aihw.gov.au/publications/index.cfm/series/41/startRow/11 (accessed Jan 2008).
- 21. Australian Institute of Health and Welfare. Australian hospital statistics 2002–03. Canberra: AIHW, 2004. (AIHW Cat. No. HSE 32; Health Services Series No. 22.) http://www.aihw.gov.au/publications/index.cfm/series/41 (accessed Jan 2008).
- 22. Australian Institute of Health and Welfare. Australian hospital statistics 2003–04. Canberra: AIHW, 2005. (AIHW Cat. No. HSE 37; Health Services Series No. 23.) http://www.aihw.gov.au/publications/index.cfm/series/41 (accessed Jan 2008).
- 23. Australian Institute of Health and Welfare. Australian hospital statistics 2000–01. Canberra: AIHW, 2006. (AIHW Cat. No. HSE 41; Health Services Series No. 26.) http://www.aihw.gov.au/publications/index.cfm/series/41 (accessed Jan 2008).
- 24. Turnidge J, Bell J, Pearson J, Franklin C, on behalf of the Australian Group on Antimicrobial Resistance. Gram-negative survey. 2004 antimicrobial susceptibility report. Canberra: AGAR, 2005. http://antimicrobial-resistance.com (accessed Aug 2006). (Follow links to AMR surveillance, GNB 2004 susceptibility report.)
- 25. Kumar S, Rizvi M, Vidhani S, Sharma VK. Changing face of septicaemia and increasing drug resistance in blood isolates. Indian J Pathol Microbiol 2004; 47: 441-446.
- 26. Biedenbach DJ, Moet GJ, Jones RN. Occurrence and antimicrobial resistance pattern comparisons among bloodstream infection isolates from the SENTRY antimicrobial surveillance program (1997–2002). Diagn Microbiol Infect Dis 2004; 50: 59-69.
- 27. Lau SM, Peng MY, Chang FY. Resistance rates to commonly used antimicrobials among pathogens of both bacteremic and non-bacteremic community-acquired urinary tract infection. J Microbiol Immunol Infect 2004; 37: 185-191.





Abstract
Objective: To determine the population incidence and clinical features of Escherichia coli bacteraemia in Canberra, Australia.
Design, setting and participants: Canberra (including the nearby local government areas of Queanbeyan and Yarrowlumla) has a geographically isolated population of about 366 000 people. Its six hospitals also provide tertiary medical services for the surrounding region. Confining our analysis (by residential postcodes) to Canberra residents only, we used microbiology laboratory records and population statistics to calculate the population incidence of E. coli bacteraemia from January 2000 to December 2004. Clinical data were also collected prospectively on episodes occurring within three of the hospitals.
Main outcome measures: Population incidence of E. coli bacteraemia; place of acquisition of infection; focus of infection within body; recovery, new morbidity or death at 7 days.
Results: During the 5-year period, 515 episodes of E. coli bacteraemia occurred in Canberra residents, an incidence of 28 per 100 000 population per year. The highest rate was in men aged ≥ 80 years (463 per 100 000). Overall, E. coli bacteraemia occurred in equal numbers in males and females, but incidence was higher in males aged < 1 year and ≥ 60 years. Most episodes occurred in people aged ≥ 60 years (316/511 [62%]) and most were community-associated (347/511 [68%]). Half the infections (257/511) had a genitourinary focus and 28% (141/511) a gastrointestinal focus. The 7-day case-fatality rate was 5%. Prostate biopsies and urinary catheters were notable preventable foci of health care-associated bacteraemia. Resistance of isolates to gentamicin (2.1%), ciprofloxacin (1.8%) and cefotaxime (0.4%) was low.
Conclusions: E. coli is the most common cause of bacteraemia in Canberra, and incidence increases with age. Most cases have a community onset, but many episodes are related to health care procedures. Ongoing surveillance is important for identifying risk factors that may be modified to reduce disease.