Chlamydia trachomatis is the most commonly diagnosed bacterial sexually transmissible infection (STI) in Australia. Notification rates have risen dramatically, from 47.4 per 100 000 population in 1997 to 203 per 100 000 in 2005.1 Chlamydial infection causes significant morbidity, particularly in women: up to two-thirds of tubal factor infertility and a third of ectopic pregnancy may be attributable to chlamydia.2 As over 80% of infections in men and women are asymptomatic, screening is the only way to detect cases and reduce the duration of infection and the risk of complications.2,3 Non-invasive testing (using urine samples)4 and single-dose treatment with azithromycin5 now make widespread chlamydia testing feasible.
In its first National Sexually Transmissible Infections Strategy released in 2005,6 the federal government stated that a chlamydia screening pilot program targeting sexually active young people under 25 years of age should be a priority action. It announced in the same year that it would provide $12.5 million over 4 years for increased chlamydia awareness, improved surveillance and a pilot chlamydia testing program.7 It subsequently committed $3.5 million to chlamydia prevention projects targeting high-risk groups8 and called for tenders for the design, modelling and evaluation of a chlamydia pilot program in general practice.9
Opportunistic screening was introduced in Sweden10 and parts of the United States in the 1980s.11 These programs principally targeted young women when they presented to family planning and STI clinics in the US and a variety of health care settings in Sweden.
In Sweden, after opportunistic screening was introduced, the prevalence of chlamydia among those tested (chlamydia positivity) declined by nearly 70% in women and 61% in men between 1985 and 1993.12 Over a similar period, chlamydia positivity fell by 29%–41% in US women.11
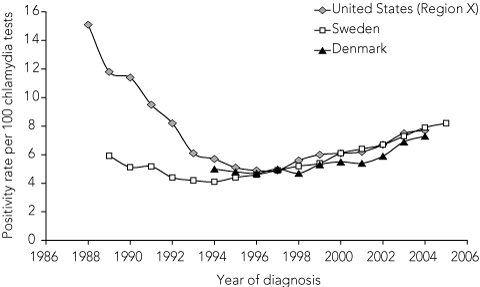
However, since the mid-1990s, this downward trend has reversed, with chlamydia positivity increasing both in Sweden13 and the US14 (Box 1). A similar trend occurred in Denmark, which has had widespread chlamydia testing since the early 1990s although it has no official chlamydia screening program.15
While opportunistic screening has not led to a sustained reduction in chlamydia prevalence in Sweden, Denmark and the US, there is evidence that chlamydia screening can reduce the incidence of complications associated with chlamydia. Two randomised controlled trials (RCTs) have investigated the impact of chlamydia screening on the incidence of pelvic inflammatory disease (PID) among women in a health maintenance organisation in the US16 and among high school students in Denmark.17 Both studies demonstrated about 50% lower PID incidence among those screened. Although potential biases in the study design reduce the validity of these RCT results,18 the data are supported by ecological analyses showing falling hospital admission rates for PID and ectopic pregnancy after chlamydia diagnosis rates fell in Sweden.19
Mathematical modelling studies of the impact of screening on chlamydia prevalence have predicted that chlamydia prevalence should fall if women aged 15–24 years are screened annually, and that screening men as well will lead to a faster and greater reduction in overall prevalence.20,21 Thus, it is unclear why there has been an increase in chlamydia prevalence, despite the screening of 25%–30% of 15–24-year-old women in Sweden each year since 1985.22 The increased use of more sensitive tests only partially explains the increased rates.22 Failure to target men as well as women for screening may be another contributing factor:18 only about 25% of tests conducted in Sweden annually are for men,13 yet chlamydia positivity among young men is more than double that among women of the same age.22
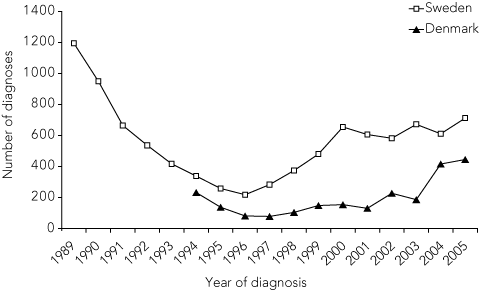
Another possible explanation for the fall and then rise in chlamydia prevalence is that sexual behaviour may have changed. If this were the case, we might expect to see a similar pattern in gonorrhoea diagnoses — and, indeed, the number of gonorrhoea diagnoses did reach a low point in Sweden and Denmark in 1996–1997, with subsequent considerable rises (Box 2). The fall in gonorrhoea incidence in the late 1980s coincided with reduced sexual risk behaviour in response to HIV/AIDS awareness and prevention campaigns, and the subsequent increase in gonorrhoea diagnoses in the late 1990s has been attributed, in part, to increased sexual risk behaviour.23
The National Survey of Sexual Attitudes and Lifestyles, a sexual behaviour survey conducted in the United Kingdom in 1990 and again in 2000,24 showed significant increases in sexual risk behaviour between the two surveys, with the number of heterosexual partners in the preceding 5 years increasing significantly for both sexes. Australian data show that age at first sexual intercourse has fallen, with women aged 16–24 years reporting a median age of 16 at first sexual intercourse compared with 19 for women aged 50–59 years.25 None of the published mathematical models assessing chlamydia screening has taken altered patterns of sexual behaviour into account.20,21 This may help to explain why these models predict a sustained fall in chlamydia prevalence after 10 years’ screening, whereas the results of overseas screening programs in fact show the opposite.
There are several options that could be investigated. Firstly, we could look to the success of the National Cervical Screening Program in Australia for guidance. Pap smear testing was initially offered opportunistically to women when they visited their GP. This was replaced by an organised national cervical screening program after it became clear that opportunistic screening was having limited impact on cervical cancer incidence.26 This included the creation of Pap test registers that coordinated a system of recall to women. This program has been an outstanding success, and Australia now has the lowest cervical cancer mortality rate in the developed world.27 It is possible that a centralised register recalling people to be screened at regular intervals would be an effective component of a chlamydia screening program. As chlamydia is an STI, young adults might be reluctant to have their details recorded on a chlamydia register. However, as chlamydia, unlike cervical cancer, is not life-threatening, it is possible that a chlamydia register could be used simply to remind people that their test was due rather than record test results. Further research should investigate the acceptability and effectiveness of a chlamydia register to recall people for regular testing. In this issue of the Journal, Bowden and colleagues28 report some success with screening for chlamydia at the time of carrying out routine Pap smears (→ Screening for Chlamydia trachomatis at the time of routine Pap smear in general practice: a cluster randomised controlled trial).
Secondly, we can learn from the success Australia has had in increasing immunisation rates. The General Practice Immunisation Incentives Scheme29 provides incentives to GPs to increase childhood immunisation rates in Australia. Since its introduction in 1998, immunisation rates have increased from 73% in 1998 to 90% in 2003.30 Similar incentive schemes have been established to improve the management of diabetes and asthma by GPs.31 It could be argued that immunising children or managing diabetes is very different from recommending an STI test, and that the reality that many young adults attend a GP with a parent or guardian makes it potentially difficult to raise the subject of chlamydia testing. Nevertheless, as the vast majority of young adults visit a GP each year, consideration could be given to the possibility of incentive payments to GPs to increase chlamydia screening.
Australian general practice has achieved near universal computerisation in the past 10 years and many practices have set up systems in their clinical software to recall patients for routine tests.32 There are no readily available data showing whether this has had an impact on patient health, but the possibility of using clinical software to allow GPs to set up a system of chlamydia test recall in their practice could be further investigated.
Other possible options include chlamydia screening outside a general practice setting — for example, school-based,17 sports club-based33 and/or postal-based34,35 testing. Such programs may be more effective in reaching young adults, who may not attend a GP or who may not have a Medicare card. While sports club-based testing has been shown to be highly acceptable to young adults, school-based and postal-based testing have suffered from low participation rates when investigated, and postal testing programs have tended to attract better educated people in older age groups.34 The federal government, in common with many overseas screening programs, is currently focusing on chlamydia screening within general practice, but it may need to consider other options if general practice-based programs fail.
- Jane S Hocking1
- Jennifer Walker1
- David Regan2
- Marcus Y Chen1,3
- Christopher K Fairley3
- 1 School of Population Health, University of Melbourne, Melbourne, VIC.
- 2 National Centre in HIV Epidemiology and Clinical Research, Sydney, NSW.
- 3 Melbourne Sexual Health Centre, Melbourne, VIC.
Jane Hocking and Marcus Chen are supported by National Health and Medical Research Council (NHMRC) postdoctoral research fellowships. David Regan is supported by an NHMRC Capacity Building Grant in Population Health.
None identified.
- 1. Australian Government Department of Health and Ageing. National Notifiable Diseases Surveillance System. Number of notifications of chlamydial infection, Australia, 2005 by age group and sex. Canberra: DoHA, 2005. http://www9.health.gov.au/cda/Source/Rpt_5.cfm (accessed Jul 2007).
- 2. Peipert JF. Genital chlamydial infections. N Engl J Med 2003; 349: 2424-2430.
- 3. Sutton T, Martinko T, Hale S, Fairchok MP. Prevalence and high rate of asymptomatic infection of Chlamydia trachomatis in male college reserve officer training corps cadets. Sex Transm Dis 2003; 30: 901-904.
- 4. Morré SA, van Valkengoed IGM, de Jong A, et al. Mailed, home-obtained urine specimens: a reliable screening approach for detecting asymptomatic Chlamydia trachomatis infections. J Clin Microbiol 1999; 37: 976-980.
- 5. Magid D, Douglas JM, Schwartz JS. Doxycycline compared with azithromycin for treating women with genital Chlamydia trachomatis infections: an incremental cost-effectiveness analysis. Ann Intern Med 1996; 124: 389-399.
- 6. Australian Government Department of Health and Ageing. National Sexually Transmissible Infections Strategy 2005–2008. Canberra: DoHA, 2005. http://www.health.gov.au/internet/wcms/publishing.nsf/Content/phd-sti-strategy-cnt.htm/$FILE/sti_strategy.pdf (accessed Jul 2007).
- 7. Abbott T. Pilot testing program for chlamydia. Media release ABB078/05, 27 Jun 2005. Canberra: Australian Government Department of Health and Ageing, 2005. http://www.health.gov.au/internet/ministers/publishing.nsf/Content/health-mediarel-yr2005-ta-abb078.htm?OpenDocument&yr=2005&mth=6 (accessed Jul 2007).
- 8. Abbott T. Chlamydia projects to target high-risk groups. Media release ABB115/06, 8 Aug 2006. Canberra: Australian Government Department of Health and Ageing, 2006. http://www.health.gov.au/internet/ministers/publishing.nsf/Content/health-mediarel-yr2006-ta-abb115.htm?OpenDocument&yr=2006&mth=8 (accessed Jul 2007).
- 9. Australian Government Department of Health and Ageing. The design, modelling and evaluation of the Chlamydia Pilot in General Practice. Request for tender. Canberra: DoHA, 2007. https://www.tenders.gov.au/?event=public.advert.showClosed&AdvertUUID=0F156190-F5E5-8ADC-9AC38D8C78C25DAE (accessed Nov 2007).
- 10. Low N. Current status of chlamydia screening in Europe. Eurosurveillance 2004; 9(10-12): 68-70. http://www.eurosurveillance.org/eq/2004/04-04/pdf/eq_10_2004_68-70.pdf (accessed Nov 2007).
- 11. Hillis SD, Nakashima A, Amsterdam L, et al. The impact of a comprehensive chlamydia prevention program in Wisconsin. Fam Plann Perspect 1995; 27: 108-111.
- 12. Herrmann B, Egger M. Genital Chlamydia trachomatis infections in Uppsala County, Sweden, 1985–1993: declining rates for how much longer? Sex Transm Dis 1995; 22: 253-260.
- 13. Smittskyddsinstitutet [Swedish Institute for Infectious Disease Control]. Epidemiologisk årsrapport 2005. Solna: Smittskyddsinstitutet, 2006. http://www.smittskyddsinstitutet.se/upload/Publikationer/SMI-epidemiologisk-arsrapport-2005.pdf (accessed Nov 2007).
- 14. Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2004 supplement. Chlamydia prevalence monitoring project annual report 2004. Atlanta, Ga: Department of Health and Human Services, Centres for Disease Control and Prevention, 2005. http://www.cdc.gov/std/Chlamydia2004/ctsupplement_2004FINAL.pdf (accessed Jul 2007).
- 15. Statens Serum Institut [State Serum Institute]. Epi-News: national surveillance of communicable diseases. Copenhagen: Statens Serum Institut, 2005. http://www.ssi.dk/sw34485.asp (accessed Jul 2007).
- 16. Scholes D, Stergachis A, Heidrich FE, et al. Prevention of pelvic inflammatory disease by screening for cervical chlamydial infection. N Engl J Med 1996; 334: 1362-1366.
- 17. Ostergaard L, Andersen B, Møller JK, Olesen F. Home sampling versus conventional swab sampling for screening of Chlamydia trachomatis in women: a cluster-randomised 1-year follow-up study. Clin Infect Dis 2000; 31: 951-957.
- 18. Low N, Egger M. What should we do about screening for genital chlamydia? Int J Epidemiol 2002; 31: 891-893.
- 19. Egger M, Low N, Smith GD, et al. Screening for chlamydia infections and the risk of ectopic pregnancy in a county in Sweden: ecological analysis. BMJ 1998; 316: 1776-1780.
- 20. Kretzschmar K, Welte R, van den Hoek A, Postma MJ. Comparative model-based analysis of screening programs for Chlamydia trachomatis infections. Am J Epidemiol 2001; 153: 90-101.
- 21. Turner K, Adams E, LaMontagne D, et al. Modelling the effectiveness of chlamydia screening in England. Sex Transm Infect 2006; 82: 496-502.
- 22. Götz H, Lindbäck J, Ripa T, et al. Is the increase in notifications of Chlamydia trachomatis infections in Sweden the result of changes in prevalence, sampling frequency or diagnostic methods? Scand J Infect Dis 2002; 34: 28-34.
- 23. Nicoll A, Hughes G, Connelly M, et al. Assessing the impact of national anti-HIV sexual health campaigns: trends in the transmission of HIV and other sexually transmitted infections in England. Sex Transm Infect 2001; 77: 242-247.
- 24. Johnson AM, Mercer CH, Erens B, et al. Sexual behaviour in Britain: partnerships, practices and HIV risk behaviours. Lancet 2001; 358: 1835-1842.
- 25. Rissel CE, Richters J, Grulich AE, et al. Sex in Australia: first experiences of vaginal intercourse and oral sex among a representative sample of adults. Aust N Z J Public Health 2003; 27: 131-137.
- 26. Mitchell H. Cervical screening in Australia — changing from opportunistic to organised. Health Care Inform Rev Online [Internet] 2000; 4 (7). http://hcro.enigma.co.nz/website/index.cfm?fuseaction=articledisplay&FeatureID=112 (accessed Jul 2007).
- 27. Wain GV. Cervical cancer prevention: the saga goes on, but so much has changed [editorial]! Med J Aust 2006; 185: 476-477. <MJA full text>
- 28. Bowden FJ, Currie MJ, Toyne H, et al. Screening for Chlamydia trachomatis at the time of routine Pap smear in general practice: a cluster randomised controlled trial. Med J Aust 2008; 188: 76-80.
- 29. Medicare Australia. General Practice Immunisation Incentives (GPII) Scheme. http://www.medicareaustralia.gov.au/providers/incentives_allowances/gpii_scheme.shtml (accessed Nov 2007).
- 30. Health Insurance Commission. General practice immunisation incentives — 2002/2003 statistical report. Canberra: HIC, 2003.
- 31. Health Insurance Commission. Practice Incentive Program (PIP). http://www.medicareaustralia.gov.au/providers/incentives_allowances/pip.htm (accessed Apr 2007).
- 32. McInnes DK, Saltman DC, Kidd MR. General practitioners’ use of computers for prescribing and electronic health records: results from a national survey. Med J Aust 2006; 185: 88-91. <MJA full text>
- 33. Gold J, Hocking J, Hellard M. The feasibility of recruiting young men in rural areas from community football clubs for STI screening. Aust N Z J Public Health 2007; 31: 243-246.
- 34. Hocking J, Willis J, Tabrizi S, et al. A chlamydia prevalence survey of young women living in Melbourne, Victoria. Sex Health 2006; 3: 235-240.
- 35. Macleod J, Salisbury C, Low N, et al. Coverage and uptake of systematic postal screening for genital Chlamydia trachomatis and prevalence of infection in the United Kingdom general population: cross sectional study. BMJ 2005; 330: 940-943.





Abstract
Chlamydia screening programs overseas have failed to reduce chlamydia prevalence despite screening 20%–30% of young sexually active women.
The Australian federal government announced in 2005 that it would provide $12.5 million for chlamydia control. Policymakers must look to chlamydia screening programs in other countries to learn from their experience.
Australia has an excellent primary health care system and a strong track record in establishing highly successful public health programs. This experience places it in a strong position to design and implement an innovative chlamydia screening program to reduce chlamydia prevalence.