Indigenous Australians continue to live in socioeconomically disadvantaged conditions, reflected in a life expectancy 17 years less than for non-Indigenous Australians.1 In the Northern Territory, the main contributors to this shorter life expectancy are now reported to be non-communicable diseases, such as cardiovascular disease, diabetes and malignancy, rather than infectious diseases.2
However, regional differences in incidence and prevalence rates of infectious diseases suggest this might not be true for all areas of the NT. In Central Australia, rates of invasive pneumococcal disease3 and of chronic hepatitis B infection in some desert communities4 are far higher than elsewhere, and rates of childhood bronchiectasis are among the highest in the world.5 Bronchiectasis is associated with infection by the human T-cell lymphotropic virus type 1 (HTLV-1),6 which is endemic to Central Australia but rare in the tropical north of the NT.7 In resource-poor regions, HTLV-1 infection predisposes to scabies8 and Strongyloides hyperinfection,9 both of which may lead to life-threatening bacterial sepsis. The contribution of infectious diseases to Indigenous mortality in Central Australia might therefore be obscured by the inclusion of data from the more populous north.
The Central Australian Human Research Ethics Committee approved the study.
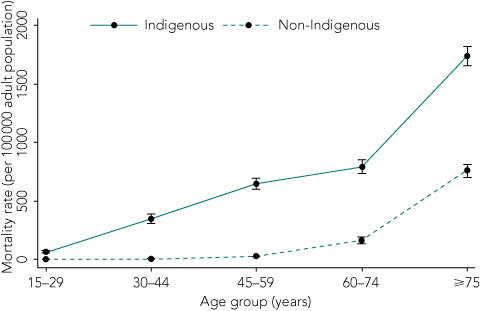
Sixty per cent of deaths among Indigenous patients (209/351) were associated with an infection, compared with 25% among non-Indigenous patients (41/162) (P < 0.001). Death was related to malignancy for only 9% of Indigenous patients (31/351), compared with 39% of non-Indigenous patients (63/162) (P < 0.001). Infection-related mortality rates are shown in Box 1 and Box 2; characteristics and comorbidities of these patients are shown in Box 3. The overall population-based infection-related MRR for Indigenous versus non-Indigenous patients was 9.9 (95% CI, 7.1–13.9; P < 0.001) (Box 1). After adjusting for age and year of death in the Poisson regression model, the MRR increased to 11.3 (95% CI, 8.0–15.8; P < 0.001). The overall MRR for patients aged < 60 years was 31.5 (95% CI, 16.1–61.8), with no change after adjustment for year of death.
Excluding chronic viral infections, a pathogen was identified for 58% (122/209) of Indigenous and 37% (15/41) of non-Indigenous patients with infection-related deaths (Box 4). For Indigenous patients, bacterial pathogens were isolated from any site in 56% (117/209), and from blood in 38% (79/209). Half of the bacterial pathogens (58/117) were enteric organisms.
No non-bacterial infections occurred in non-Indigenous patients (Box 4). In Indigenous patients, non-bacterial infections identified were the parasitic infection strongyloidiasis (2), invasive fungal infections (3) and chronic hepatitis B viral infection (10). HTLV-1 serological status was determined for only one patient with proven strongyloidiasis. This patient was seropositive, as were all those with invasive fungal infections. Of 64 Indigenous patients whose HTLV-1 serological status was determined, 30 were seropositive.
A further 28% (59/209) of Indigenous and 34% (14/41) of non-Indigenous patients had radiological evidence of pneumonia, but no pathogen was isolated. The respiratory tract was therefore the main focus of infection for both groups (Box 5). Microbiology and imaging tests failed to identify a focus of infection for the remaining 9% (19/209) of Indigenous and 37% (15/41) of non-Indigenous patients.
At least one recognised risk factor for bacterial sepsis was recorded for 145 Indigenous (69%) and 23 non-Indigenous (56%) patients. For Indigenous patients, this was most frequently diabetes (40%), excess alcohol consumption (38%) or end-stage kidney disease (ESKD) (17%) (Box 3). Nearly a third of patients had no risk factors, and alcohol was the only identified risk factor for 28% of Indigenous patients.
Parasitic infestation, invasive fungal infections and chronic hepatitis B infection are also likely to have contributed to adverse outcomes for Indigenous patients. Internationally, HTLV-1 infection is a major risk factor for Strongyloides hyperinfection9 and in our recent audit of complicated strongyloidiasis in the Central Australian Indigenous population, seven of 11 patients tested were HTLV-1 seropositive.10 In this study, nearly 50% of Indigenous patients tested were HTLV-1 seropositive, compared with background HTLV-1 seroprevalence rates of 7.2%11–13.9%.7 This difference is unlikely to result solely from a predisposition to strongyloidiasis, and most bacterial pathogens isolated were not those typically associated with scabies. Whether HTLV-1 infection increases the risk of bacterial sepsis or worsens its outcomes in some other way warrants further study.
In this study, 10 Indigenous patients with chronic hepatitis B infection died with complications of end-stage liver disease (ESLD). Prevalence rates of chronic hepatitis B infection have previously been reported to be as high as 26% in some Australian desert communities.4 While incidence rates are now low since the introduction of vaccination, a cohort of older patients continues to progress to ESLD.
Possible limitations of this study are the accuracy of ICD-10-AM coding at ASH and an inability to establish the actual cause of death, as discharge coding is based on reasons for admission, not death. Nevertheless, microbiology and imaging tests independently confirmed the coded reason for admission for 91% of the Indigenous patients with infection-related deaths. Moreover, the infection-related mortality rate for the non-Indigenous patient group (0.35 per 1000 population) is consistent with estimated rates for white people in the United States (0.41–1.0 per 1000 population)12-14 and rates of severe sepsis in major Australian centres (0.77 per 1000 population).15 The contribution of infectious diseases to other deaths that were coded as being unrelated to infection, that occurred in other units or that followed transfer to tertiary referral centres was not studied. The observations reported here are therefore likely to underestimate the actual number of infection-related deaths at ASH.
Although reported rates of diabetes (31%)14 and ESKD (13%)16 for African Americans who died with sepsis approach those reported here, the major contributor to racial disparities in infection-related mortality in the US is HIV infection,17 which remains rare in Central Australia. Nevertheless, racial differences in sepsis-related mortality rates between adult white and African Americans are far less than those we found, with a twofold difference consistently reported for hospital-based incidence and mortality rates in the US.13,14,16 In contrast, Indigenous Australian adults were 11 times more likely overall, and those aged less than 60 were 31.5 times more likely, than non-Indigenous adults to suffer an inhospital infection-related death.
Infection-related mortality rates for Indigenous Australian patients are more comparable to those from other resource-poor regions. The proportions of infection-related adult deaths and admission-based mortality rates at ASH are similar to those in African hospitals before the current HIV pandemic (13.3%–34% and 1.45 per 100 admissions, respectively).18,19 However, population-based infection-related mortality rates at ASH are considerably worse than those for some African hospitals before the arrival of HIV (0.56 and 1.1 per 1000 adults in rural South Africa20 and Kenya,18 respectively).
The relative increase in risk of death due to sepsis associated with diabetes21 is reportedly 1.3, for alcohol dependence,22 1.46, and for established cirrhosis,23 2.0. Thus, these comorbid conditions are unlikely to fully account for the racial disparities in infection-related deaths at ASH. However, infection-related mortality rates at ASH are consistent with the known high incidence and prevalence rates of infectious diseases and their sequelae in Central Australia.3-5,7 Very high background rates of infectious diseases in the Indigenous population are therefore likely to be the major contributor to racial differences in infection-related mortality. Inadequate housing, overcrowding and poor sanitation inevitably expose susceptible individuals to Strongyloides stercoralis and virulent bacterial pathogens. In some communities, the median number of people living in a house is still as high as 17,24 and nearly 50% of houses are without functioning sanitation facilities.25
These data support the need for urgent intervention to improve housing infrastructure, sanitation and education, and reiterate the importance of regional clinical research to guide public health initiatives. Particularly important are prospective studies to determine whether infectious diseases, rather than chronic diseases,2 are indeed the major cause of mortality in Central Australia. Studies are also needed to elucidate the clinical significance of HTLV-1 in the region and to define risk factors for bacterial infection, especially with the enteric pathogens associated with many deaths in this study. Such knowledge may allow Indigenous people to develop locally appropriate interventions that could reduce these alarming infection-related mortality rates.
1 Infection-related mortality rates for Indigenous and non-Indigenous adults, Alice Springs Hospital, 2000–2005
2 Population-based infection-related mortality rates among Indigenous and non-Indigenous adults by age, Alice Springs Hospital, 2000–2005
 | |||||||||||||||
3 Characteristics and comorbidities of patients whose deaths were associated with an infection at Alice Springs Hospital, 2000–2005
- Lloyd J Einsiedel1
- Liselle A Fernandes2
- Richard J Woodman3
- 1 Department of Medicine, Alice Springs Hospital, Alice Springs, NT.
- 2 Clinical Research, Northern Territory Rural Clinical School, Alice Springs, NT.
- 3 Department of General Practice, Flinders University, Adelaide, SA.
We wish to thank Ms Jill Burgoyne for all data obtained from CARESYS and Dr Yuejen Zhao for providing regional population data. This study received funding from the Northern Territory Rural Clinical School, which is an initiative of the Australian Government Department of Health and Ageing.
None identified.
- 1. Australian Bureau of Statistics. The health and welfare of Australia’s Aboriginal and Torres Strait Islander peoples, 2005. Canberra: ABS, 2005. (ABS Cat. No. 4704.0.)
- 2. Zhao Y, Dempsey K. Causes of inequality in life expectancy between Indigenous and non-Indigenous people in the Northern Territory, 1981-2000: a decomposition study. Med J Aust 2006; 184: 490-494. <MJA full text>
- 3. Krause VL, Reid SJC, Merianos A. Invasive pneumococcal disease in the Northern Territory of Australia,1994-1998. Med J Aust 2000; 173 (7 Suppl): S27-S31.
- 4. Burrell CJ, Cameron AS, Hart G, et al. Hepatitis B reservoirs and attack rates in an Australian community: a basis for vaccination and cross-infection policies. Med J Aust 1983; 2: 492-496.
- 5. Chang AB, Masel JP, Boyce NC, et al. Non-CF bronchiectasis: clinical and HRCT evaluation. Pediatr Pulmonol 2003; 35: 477-483.
- 6. Steinfort DP, Brady S, Weisinger HS, Einsiedel L. Bronchiectasis in Central Australia: a young face to an old disease. Respir Med 2008; 102: 574-578.
- 7. Bastian I, Hinuma Y, Doherty RR. HTLV-1 among Northern Territory Aborigines. Med J Aust 1993; 159: 12-16.
- 8. Mollison LC, Lo ST, Marning G. HTLV-1 and scabies in Australian aborigines. Lancet 1993; 341: 1281-1282.
- 9. Gotuzzo E, Terashima A, Alvarez H, et al. Strongyloides stercoralis hyperinfection associated with human T cell lymphotropic virus type-1 in Peru. Am J Trop Med Hyg 1999; 60: 146-149.
- 10. Einsiedel L, Fernandes L. Strongyloides stercoralis: a cause of morbidity and mortality for Indigenous people in central Australia. Intern Med J 2008; in press.
- 11. Bastian I. HTLV-1 studies in the Northern Territory [PhD thesis]. Darwin: Menzies School of Health Research, University of Sydney, 1996.
- 12. Simonsen L, Conn LA, Pinner RW, Teutsch S. Trends in infectious disease hospitalizations in the United States, 1980-1994. Arch Intern Med 1998; 158: 1923-1928.
- 13. Martin GS, Mannino DM, Eaton S, Moss M. The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med 2003; 348: 1546-1554.
- 14. Dombrovskiy VY, Martin AA, Sunderram J, Paz HL. Occurrence and outcomes of sepsis: influence of race. Crit Care Med 2007; 35: 763-768.
- 15. Finfer S, Bellomo R, Lipman J, et al. Adult-population incidence of severe sepsis in Australian and New Zealand intensive care units. Intensive Care Med 2004; 30: 589-596.
- 16. Esper AM, Moss M, Lewis CA, et al. The role of infection and comorbidity: factors that influence disparities in sepsis. Crit Care Med 2006; 34: 2576-2582.
- 17. Wong MD, Shapiro MF, Boscardin WJ, Ettner SL. Contribution of major diseases to disparities in mortality. N Engl J Med 2002; 347: 1585-1592.
- 18. Petit PLC, van Ginneken JK. Analysis of hospital records in four African countries, 1975-1990, with emphasis on infectious diseases. J Trop Med Hyg 1995; 98: 217-227.
- 19. Couper I, Walker ARP. Causes of death in a rural hospital in South Africa. Cent Afr J Med 1997; 43: 219-222.
- 20. Bac DJ. Causes of death at the Donald Fraser Hospital, Venda. South Afr J Epidemiol Infect 1989; 4(2): 25-27.
- 21. Joshi N, Caputo GM, Weitekamp MR, Karchmer AW. Infections in patients with diabetes mellitus. N Engl J Med 1999; 341: 1906-1912.
- 22. O’Brien JM Jr, Lu B, Ali NA, et al. Alcohol dependence is independently associated with sepsis, septic shock, and hospital mortality among adult intensive care unit patients. Crit Care Med 2007; 35: 345-350.
- 23. Foreman MG, Mannino DM, Moss M. Cirrhosis as a risk factor for sepsis and death: analysis of the National Hospital Discharge Survey. Chest 2003; 124: 1016-1020.
- 24. McDonald MI, Towers RJ, Andrews RM, et al. Low rates of streptococcal pharyngitis and high rates of pyoderma in Australian aboriginal communities where acute rheumatic fever is hyperendemic. Clin Infect Dis 2006; 43: 683-689.
- 25. Bailie RS, Stevens MR, McDonald E, et al. Skin infection, housing and social circumstances in children living in remote Indigenous communities: testing conceptual and methodological approaches. BMC Public Health 2005; 5: 128.





Abstract
Objective: To compare infection-related mortality rates and pathogens isolated for Indigenous and non-Indigenous adult patients at Alice Springs Hospital (ASH).
Design, participants and setting: Retrospective study of inhospital deaths of adults (patients aged ≥ 15 years) associated with an infection during a medical or renal admission to ASH between 1 January 2000 and 31 December 2005.
Main outcome measures: Admission- and population-based infection-related mortality rates and mortality rate ratios (MRRs) for Indigenous versus non-Indigenous adults.
Results: There were 513 deaths, of 351 Indigenous and 162 non-Indigenous patients. For Indigenous patients, 60% of deaths were infection-related, compared with 25% for non-Indigenous patients (P < 0.001). The admission-based infection-related MRR for Indigenous versus non-Indigenous adults was 2.2 (95% CI, 1.6–3.1) (15.3 v 6.8 deaths per 1000 admissions; P < 0.001). After adjusting for age and year of death, the population-based infection-related MRR was 11.3 (95% CI, 8.0–15.8) overall (351 v 35 deaths per 100 000 population; P < 0.001) and 31.5 (95% CI, 16.1–61.8) for patients aged < 60 years. The median age of patients who died with an infection was 49 (interquartile range [IQR], 38–67) years for Indigenous and 73 (IQR, 58–80) years for non-Indigenous patients (P < 0.001). For Indigenous patients, 56% of infection-related deaths were associated with bacterial sepsis, with half of these due to enteric organisms. Other deaths followed chronic hepatitis B infection, invasive fungal infections and complications of strongyloidiasis.
Conclusion: Indigenous patients at ASH are 11 times more likely than non-Indigenous patients to die with an infectious disease. This racial disparity reflects the ongoing socioeconomic disadvantage experienced by Indigenous Australians.