Over a 6-month period, two patients presented to a community hospital emergency department with perforated gastric ulcers as the result of recreational misuse of over-the-counter ibuprofen–codeine preparations. Misuse of these medications appears to be an emerging cause of significant morbidity in patients with codeine addiction.
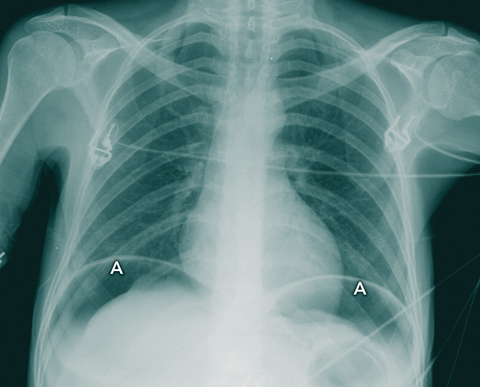
An erect chest x-ray confirmed the presence of gas under the diaphragm (Box 1). An urgent laparotomy revealed a perforated anterior gastric antrum ulcer and 2.6 L of green turbid fluid in the peritoneal cavity. The patient was given an additional four units of packed red cells intraoperatively, and her ulcer was oversewn. Postoperatively, she was transferred to an intensive care unit at another hospital.
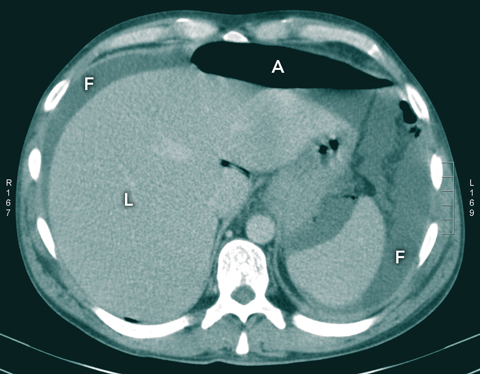
Intravenous contrast computed tomography revealed the presence of free fluid and gas in the peritoneal cavity (Box 2). An urgent laparotomy revealed a 1.5 cm gastric antrum ulcer with gross peritoneal contamination. Postoperatively, the patient was offered care at an inpatient drug and alcohol service, but absconded before transfer could be arranged.
Ibuprofen–codeine preparations first entered the Australian over-the-counter (OTC) market in October 2002. The leading product in the market, Nurofen Plus, contains 200 mg ibuprofen and 12.8 mg codeine phosphate in each tablet,1 making it the strongest codeine tablet available in Australia without prescription.
Several cases of severe hypokalaemia secondary to ibuprofen-induced renal tubule acidosis after Nurofen Plus misuse have been reported in the literature.2-4 One study showed that patients with acute upper gastrointestinal presentations were 5.2 times more likely to have consumed high-dose OTC non-aspirin non-steroidal anti-inflammatory drugs within the previous week, and the increased risk was dose-dependent.5
- 1. New drugs. Ibuprofen/codeine phosphate. Aust Prescriber 2002; 25: 94-99.
- 2. Lambert AP, Close C. Life-threatening hypokalaemia from abuse of Nurofen Plus. J R Soc Med 2005; 98: 21.
- 3. Dyer BT, Martin JL, Mitchell JL, et al. Hypokalaemia in ibuprofen and codeine phosphate abuse. Int J Clin Pract 2004; 58: 1061-1062.
- 4. Chetty R, Baoku Y, Mildner R, et al. Severe hypokalaemia and weakness due to Nurofen misuse. Ann Clin Biochem 2003; 40: 422-423.
- 5. Lewis JD, Kimmel SE, Localio AR, et al. Risk of serious upper gastrointestinal toxicity with over-the-counter nonaspirin nonsteroidal anti-inflammatory drugs. Gastroenterology 2005; 129: 1865-1874.





None identified.