Persistent allergic rhinitis (PAR) affects a substantial number of people worldwide,1 including 16% of the population of Australia (over 3.2 million people).2 The condition involves an inflammatory response to allergens such as house dust mites and pet dander.3,4 The primary symptoms are nasal obstruction, sneezing, rhinorrhoea and nasal itch, which, by definition, are present more than 4 days a week, and for more than 4 weeks (in contrast to intermittent allergic rhinitis).5 In addition, patients with PAR often present with headaches, impaired hearing, postnasal drip, decreased taste and smell, earache and symptoms of sleep apnoea.4,6 Allergic rhinitis is also associated with a higher prevalence of asthma.7 Although PAR is not life-threatening, it affects quality of life and has substantial economic and social impact.8 Pharmacotherapy provides symptomatic relief of PAR.9 However, most medications have side effects, such as nose bleeds and septal perforation, and need to be taken for prolonged periods.5 Increasingly, sufferers are seeking alternative therapies for PAR and other allergic diseases.10 For example, Chinese herbal medicine has been shown to be beneficial in the treatment of seasonal allergic rhinitis (SAR)11 and PAR.12 Acupuncture was found to be effective for adult patients with SAR13 and for children with PAR.14 We undertook a study to evaluate the effectiveness of individualised acupuncture treatment in adults with PAR.
The study was a randomised, single-blind, sham-controlled trial. Randomisation, by a researcher who did not have direct contact with participants, used a computer-generated random number in a sealed envelope. Participants randomly selected an envelope before beginning treatment. Participants, assessors and the Chinese medicine practitioner who performed the Chinese medicine diagnosis were blinded to the treatment allocation. The acupuncturist was not blinded, but was instructed not to communicate with the participants about treatment procedures and responses. The trial involved a 1-week baseline period, an 8-week treatment period and a 12-week follow-up period (Box 1).
Three key acupoints and one supplementary acupoint were used for each participant. The key acupoints were Yingxiang (LI 20), Yintang (Extra point), and Fengchi (GB 20).13 The supplementary acupoint was determined individually on the basis of Chinese medicine syndrome differentiation, being Hegu (LI 4) for lung qi deficiency syndrome, Zusanli (ST 36) for spleen qi deficiency syndrome, or Qihai (CV 6) for kidney qi deficiency syndrome.13,15
For sham acupuncture, the insertion sites were 1–1.5 cm from the acupoints used for real treatment, and shorter needles (diameter, 0.25 mm; length, 13 mm; Suzhou Medical Appliance Factory) were applied in a shallow needling technique (3–5 mm).13
Needling was carried out with participants in the supine position. Needling sites were swabbed with 70% isopropyl alcohol before insertion. On needle withdrawal, dry sterilised cotton balls were firmly applied to insertion points.13
Throughout the study, four nasal symptoms (nasal obstruction, sneezing, rhinorrhoea and nasal itch) were self-assessed daily and recorded in a diary by participants, using a five-point scale (0 = no symptom; 1 = mild; 2 = moderate; 3 = severe; and 4 = very severe).16 Seven-day individual and total nasal symptom scores (TNSS) were determined from the daily symptom scores.
Participants recorded in a diary their use of PAR relief medications (short-acting antihistamines, sympathomimetic nasal decongestants and intranasal steroids) daily through-out the trial. Each dose of oral, nasal or ocular administration of a specific relief medication was scored as one.17 Seven-day relief medication scores were calculated from the daily scores.
The sample size was determined on the basis of a 70% reduction in TNSS with real acupuncture and a 30% reduction with sham acupuncture.13 Thus, a sample size of 36 for each group would provide 80% power with a type I error rate of 5% (two-tailed). For participants who withdrew, intention-to-treat analysis was applied to outcome data (nasal symptom scores and relief medication scores) using the last recorded data.
There were no significant differences between the real and sham acupuncture groups with respect to age, number of years of PAR symptoms, sex ratio, or number with a family history of PAR (Box 2). There were also no significant differences between the two groups in the proportion with positive results for any of the six skin-prick allergen tests (P > 0.4 for each allergen), nor in the proportions allocated treatment with each of the three supplementary acupoints (P = 0.78).
There were no significant differences between the two treatment groups in mean 7-day score for individual nasal symptoms or TNSS in the baseline period (Box 3). There was also no significant difference (P = 0.11) between the groups in mean relief medication scores. Eight participants in the real acupuncture group and six in the sham group were using nasal corticosteroids as relief medication.
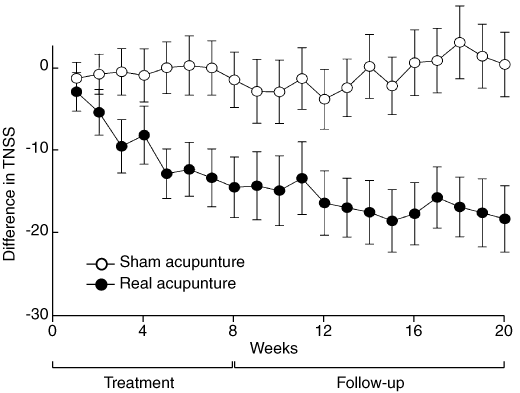
After 8 weeks of treatment, there was a significantly greater reduction from baseline in the 7-day TNSS in the real acupuncture group than in the sham treatment group (Box 4 and Box 5). The only individual symptom for which the mean reduction in the score from baseline was significantly greater with real than sham acupuncture was rhinorrhoea (Box 4).
Twelve weeks after treatment ended, the mean reduction from baseline in the 7-day TNSS remained greater for the real acupuncture group than for the sham treatment group (Box 4 and Box 5). Moreover, the mean reductions from baseline in the 7-day scores for all four individual nasal symptoms were each greater in the real treatment group than in the sham treatment group (Box 4).
PAR is characterised by chronic nasal symptoms: sneezing, nasal itch, nasal obstruction and rhinorrhoea.5 After 8 weeks of treatment, we observed a greater decrease with real acupuncture than with sham treatment in only one of these symptoms — rhinorrhoea. However, the decrease in TNSS (total score for the four symptoms) was greater with real acupuncture. Twelve weeks after the end of treatment, the decreases from baseline in the TNSS and all four individual symptom scores were greater in the real acupuncture group than in the sham group. The reduction in the TNSS with treatment, and the persistence of the effect, appear to be the most clinically significant findings of the study. Consistent with our findings, acupuncture has been reported to be beneficial in a number of previous studies on allergic rhinitis,13,14,17 including a study on children with PAR.14
In our study, participants were permitted to take PAR symptomatic relief medication when they considered it necessary. We assessed the use of relief medication as a secondary outcome measure, but the findings were difficult to interpret. Use of relief medication was reduced, compared with baseline, immediately after 8 weeks of real acupuncture treatment, and the reduction was still apparent 12 weeks later. There were no such changes in the sham-treated group. However, comparison of medication use between the groups showed no significant differences. The inconsistency in results of the two analyses may have resulted from the numerical, although statistically insignificant, differences in use of relief medication between the groups at baseline. Thus, we cannot draw a definitive conclusion about the effects of acupuncture treatment on use of conventional pharmacotherapy.13,14
To our knowledge, no other randomised controlled trial of acupuncture in adults with PAR has been reported in the English medical literature. However, a recent study on acupuncture in childhood PAR reported a positive outcome.14 Unlike that study, our study included individualised selection of acupoints, guided by Chinese medicine syndrome classification, a procedure we used in an earlier trial on acupuncture for SAR, which also demonstrated a positive outcome.13 Given the similarities between the two types of allergic rhinitis,5 it is perhaps not surprising that we have found that acupuncture is also effective in the symptomatic treatment of PAR.
Although several studies have demonstrated positive outcomes of acupuncture in various allergic conditions,13,14,17 the mechanisms of action of acupuncture are yet to be elucidated. Acupuncture has been reported to inhibit the synthesis of cytokines, such as interleukin-6 and interleukin-10, in patients with asthma,18 and interleukin-10 in patients with allergic rhinitis.19 Acupuncture also affects cellular immunity by regulating CD3 and CD4 T cells,18 as well as stimulating the release of neuropeptides involved in neurogenic inflammation, such as β-endorphin.20 The relevance to our findings is open to conjecture.
Deciding on an appropriate control procedure for clinical studies on acupuncture is a particular challenge.21 One approach has been to use non-penetrating needles,22 but such a procedure has been reported to produce more than placebo effects.23 A recent systematic review concluded that a technique in which needles are inserted shallowly at locations 1–2 cm removed from defined acupoints is a common and appropriate sham control for clinical trials on acupuncture.24 This was the procedure used for sham treatment in our study.
Consistent with the findings of previous acupuncture studies for allergic rhinitis,13,14,17 and a specific acupuncture safety study,25 we found that acupuncture was well tolerated, with only minor and minimal adverse events, none of which were serious enough to result in participant withdrawal from the trial.
2 Participant characteristics compared between treatment groups
* Real acupuncture group compared with sham acupuncture group. PAR = persistent allergic rhinitis. |
|||||||||||||||
3 Baseline 7-day nasal symptom scores by treatment group (mean, 95% CI)
* Real versus sham acupuncture. TNSS = total nasal symptom score. |
|||||||||||||||
Received 12 January 2007, accepted 25 June 2007
- Charlie C L Xue1
- Xuedong An1
- Thomas P Cheung1
- Cliff Da Costa2
- George B Lenon1
- Frank C Thien3
- David F Story4
- 1 Division of Chinese Medicine, School of Health Sciences, World Health Organization Collaborating Centre for Traditional Medicine, RMIT University, Melbourne, VIC.
- 2 School of Mathematical and Geospatial Sciences, RMIT University, Melbourne, VIC.
- 3 Department of Allergy, Immunology and Respiratory Medicine, The Alfred Hospital and Monash University, Melbourne, VIC.
- 4 School of Health Sciences, RMIT University, Melbourne, VIC.
We gratefully acknowledge RMIT University for financial support of the study, Dr Vincent Dinh for medical assessment of participants, Professor Xun Chuan Ji for specialist nasal assessments, staff of the RMIT Chinese Medicine Research Group for their assistance in the conduct of this trial, and Dr Lin Zhang for assistance in proof reading and constructive comments during preparation of the manuscript.
None identified.
- 1. Bauchau V, Durham SR. Epidemiological characterization of the intermittent and persistent types of allergic rhinitis. Allergy 2005; 60: 350-353.
- 2. Australian Bureau of Statistics. National Health Survey: summary of results 2004–2005. Canberra: ABS, 2006. (ABS Cat. No. 4364.0.)
- 3. Douglass JA, O’Hehir RE. 1. Diagnosis, treatment and prevention of allergic disease: the basics. Med J Aust 2006; 185: 228-233. <MJA full text>
- 4. Plaut M, Valentine MD. Clinical practice. Allergic rhinitis. N Engl J Med 2005; 353: 1934-1944.
- 5. Bousquet J, Van Cauwenberge P, Khaltaev N. Allergic rhinitis and its impact on asthma. J Allergy Clin Immunol 2001; 108 (5 Suppl): S147-S334.
- 6. Leger D, Annesi-Maesano I, Carat F, et al. Allergic rhinitis and its consequences on quality of sleep: an unexplored area. Arch Intern Med 2006; 166: 1744-1748.
- 7. Leynaert B, Neukirch C, Kony S, et al. Association between asthma and rhinitis according to atopic sensitization in a population-based study. J Allergy Clin Immunol 2004; 113: 86-93.
- 8. Fineman SM. The burden of allergic rhinitis: beyond dollars and cents. Ann Allergy Asthma Immunol 2002; 88 (4 Suppl 1): 2-7.
- 9. Walls RS, Heddle RJ, Tang ML, et al. Optimising the management of allergic rhinitis: an Australian perspective. Med J Aust 2005; 182: 28-33. <MJA full text>
- 10. Schafer T, Riehle A, Wichmann HE, Ring J. Alternative medicine in allergies — prevalence, patterns of use, and costs. Allergy 2002; 57: 694-700.
- 11. Xue CC, Thien FC, Zhang JJ, et al. Treatment for seasonal allergic rhinitis by Chinese herbal medicine: a randomized placebo controlled trial. Altern Ther Health Med 2003; 9: 80-87.
- 12. Hu G, Walls RS, Bass D, et al. The Chinese herbal formulation biminne in management of perennial allergic rhinitis: a randomized, double-blind, placebo-controlled, 12-week clinical trial. Ann Allergy Asthma Immunol 2002; 88: 478-487.
- 13. Xue CC, English R, Zhang JJ, et al. Effect of acupuncture in the treatment of seasonal allergic rhinitis: a randomized controlled clinical trial. Am J Chin Med 2002; 30: 1-11.
- 14. Ng DK, Chow PY, Ming SP, et al. A double-blind, randomized, placebo-controlled trial of acupuncture for the treatment of childhood persistent allergic rhinitis. Pediatrics 2004; 114: 1242-1247.
- 15. Qiu ML. Chinese acupuncture and moxibustion. London: Longman Group UK, 1993.
- 16. Prenner BM, Chervinsky P, Hampel FC Jr, et al. Double-strength beclomethasone dipropionate (84 micrograms/spray) aqueous nasal spray in the treatment of seasonal allergic rhinitis. J Allergy Clin Immunol 1996; 98: 302-308.
- 17. Brinkhaus B, Hummelsberger J, Kohnen R, et al. Acupuncture and Chinese herbal medicine in the treatment of patients with seasonal allergic rhinitis: a randomized-controlled clinical trial. Allergy 2004; 59: 953-960.
- 18. Joos S, Schott C, Zou H, et al. Immunomodulatory effects of acupuncture in the treatment of allergic asthma: a randomized controlled study. J Altern Complement Med 2000; 6: 519-525.
- 19. Petti FB, Liguori A, Ippoliti F. Study on cytokines IL-2, IL-6, IL-10 in patients of chronic allergic rhinitis treated with acupuncture. J Tradit Chin Med 2002; 22: 104-111.
- 20. Dawidson I, Angmar-Mansson B, Blom M, et al. Sensory stimulation (acupuncture) increases the release of calcitonin gene-related peptide in the saliva of xerostomia sufferers. Neuropeptides 1999; 33: 244-250.
- 21. Birch S. Clinical research on acupuncture. Part 2. Controlled clinical trials, an overview of their methods. J Altern Complement Med 2004; 10: 481-498.
- 22. Streitberger K, Kleinhenz J. Introducing a placebo needle into acupuncture research. Lancet 1998; 352: 364-365.
- 23. Kaptchuk TJ, Stason WB, Davis RB, et al. Sham device v inert pill: randomised controlled trial of two placebo treatments. BMJ 2006; 332: 391-397.
- 24. Dincer F, Linde K. Sham interventions in randomized clinical trials of acupuncture — a review. Complement Ther Med 2003; 11: 235-242.
- 25. MacPherson H, Thomas K, Walters S, Fitter M. The York acupuncture safety study: prospective survey of 34 000 treatments by traditional acupuncturists. BMJ 2001; 323: 486-487.





Abstract
Objective: To investigate the effectiveness and safety of acupuncture in persistent allergic rhinitis (PAR)
Design: Randomised, single-blind, sham-controlled trial conducted from May 2004 to February 2005.
Participants and intervention: 80 patients with PAR (age, 16–70 years) were randomly assigned to receive real or sham acupuncture. After a 1-week baseline period, participants were treated twice weekly for 8 weeks and followed up for another 12 weeks.
Main outcome measures: Nasal obstruction, sneezing, rhinorrhoea and nasal itch were each self-assessed daily on a 5-point scale, and scores were aggregated weekly. The sum of the symptom scores (total nasal symptom score, TNSS) was also determined. A secondary outcome was use of PAR relief medication.
Results: After 8 weeks’ treatment, the weekly mean difference in TNSS from baseline was greater with real (−17.2; 95% CI, −24.6 to −9.8) than with sham acupuncture (−4.2; 95% CI, −11.0 to 2.7) (P = 0.01). The decrease in individual symptom score was also greater with real acupuncture for rhinorrhoea (P < 0.01) but not the other symptoms. At the end of follow-up, the greater difference in TNSS from baseline in the real acupuncture group was still apparent: real, −21.0 (95% CI, −29.1 to −12.9) versus sham, − 2.3 (95% CI, −10.2 to 5.6) (P = 0.001). Moreover, the differences from baseline in all four individual symptom scores were greater for the real than for the sham group (P < 0.05). Real and sham acupuncture were both well tolerated.
Conclusion: Our findings suggest that acupuncture is effective in the symptomatic treatment of PAR.
Trial registration: Australian Government Therapeutic Goods Administration CTN 034/2004.